Scalable Synthesis of Anidulafungin Side Chain via Advanced Suzuki Coupling Technology
The pharmaceutical industry continuously seeks robust manufacturing routes for complex antifungal agents, and the synthesis of the anidulafungin side chain intermediate, p-pentoxy terphenyl formic acid, represents a critical bottleneck in this value chain. Patent CN109574811B discloses a transformative preparation method that fundamentally shifts the paradigm from hazardous organometallic reagents to a safer, palladium-catalyzed Suzuki coupling strategy. This innovation addresses the long-standing challenges of moisture sensitivity and thermal instability associated with traditional lithiation or Grignard pathways, offering a viable solution for reliable pharmaceutical intermediates supplier networks globally. By utilizing tetrahydroxydiboron as a stable boron source and optimizing ligand systems, the process achieves high conversion rates while maintaining stringent safety standards required for modern chemical plants. The technical breakthrough lies not only in the reaction chemistry but in the holistic design of the workflow, which simplifies downstream processing and minimizes the generation of hazardous waste streams. For R&D directors and procurement leaders, this patent data signifies a mature technology ready for immediate technology transfer and commercial deployment without the need for extensive re-engineering of existing infrastructure.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the terphenyl backbone for echinocandin antifungals relied heavily on the use of n-butyllithium or Grignard reagents to generate the necessary aryl metal species for carbon-carbon bond formation. These conventional methodologies impose severe operational constraints, primarily due to the extreme pyrophoric nature of n-butyllithium and the rigorous moisture exclusion required for Grignard reactions, often necessitating specialized glove box environments for reagent preparation. The handling of such hazardous materials increases the risk of fire and explosion, leading to elevated insurance costs and complex safety protocols that can hinder cost reduction in pharmaceutical intermediates manufacturing. Furthermore, the quenching of these reactive species often generates significant amounts of inorganic salt waste and requires low-temperature conditions that demand energy-intensive cooling systems. The filtration of magnesium chips from Grignard reactions adds another layer of mechanical complexity, potentially introducing metal particulates that are difficult to remove in later purification stages. These factors collectively render traditional routes less attractive for large-scale production where safety, efficiency, and environmental compliance are paramount concerns for supply chain heads.
The Novel Approach
The novel approach detailed in the patent data leverages the versatility of the Suzuki-Miyaura coupling reaction to construct the terphenyl skeleton under significantly milder and safer conditions. By replacing unstable organolithium or organomagnesium species with stable aryl boronic acids generated in situ from tetrahydroxydiboron, the process eliminates the need for cryogenic temperatures and strict anhydrous environments during the critical bond-forming steps. This shift allows for the use of standard stainless steel reactors and simplifies the operational procedure, directly contributing to enhanced supply chain reliability and reduced capital expenditure on specialized equipment. The method employs a dual-coupling strategy where the boronic acid intermediate is coupled with an aryl halide, facilitated by specific palladium catalysts and phosphine ligands that maximize yield while minimizing homocoupling byproducts. The subsequent hydrolysis step utilizes phase transfer catalysis to ensure complete conversion of the ester to the carboxylic acid, streamlining the final isolation process. This comprehensive redesign of the synthetic route demonstrates a clear commitment to process intensification and safety, making it an ideal candidate for the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Pd-Catalyzed Suzuki Coupling
The core of this synthetic innovation lies in the precise control of the palladium catalytic cycle during the formation of the biaryl and terphenyl bonds. In the second step, the oxidative addition of the aryl bromide to the palladium(0) species, generated from palladium acetate and tris(o-methylphenyl)phosphine, is the rate-determining step that dictates the overall efficiency of the boronic acid formation. The choice of the bulky and electron-rich tris(o-methylphenyl)phosphine ligand is critical, as it stabilizes the active palladium center and facilitates the transmetallation with the tetrahydroxydiboron species activated by potassium acetate. This specific ligand environment suppresses the formation of palladium black and prevents catalyst deactivation, ensuring a consistent turnover number throughout the reaction batch. The use of a mixed solvent system involving tetrahydrofuran and methanol further enhances the solubility of the inorganic base and the boron reagent, promoting a homogeneous reaction phase that improves mass transfer kinetics. Understanding these mechanistic nuances is essential for R&D teams aiming to replicate the high yields reported in the patent examples, as slight deviations in ligand-to-metal ratios can significantly impact the impurity profile of the intermediate.
Impurity control is rigorously managed through the selection of reaction conditions that favor the desired cross-coupling over competing homocoupling or protodeboronation pathways. The third step involves a second Suzuki coupling between the newly formed boronic acid and methyl 4-iodobenzoate, where the reactivity of the aryl iodide ensures rapid oxidative addition even at moderate temperatures of 85°C. The use of sodium carbonate as the base in a toluene and n-propanol mixture provides an optimal pH environment that activates the boron species without promoting the hydrolysis of the ester functionality prematurely. This careful balancing of chemical reactivity ensures that the terphenyl ester is formed with high selectivity, minimizing the presence of unreacted starting materials or mono-coupled byproducts that would be difficult to separate later. The final hydrolysis step employs cetyltrimethylammonium bromide as a phase transfer catalyst, which facilitates the transport of hydroxide ions into the organic phase to attack the ester carbonyl efficiently. This mechanism ensures complete conversion to the carboxylic acid, resulting in a final product that meets the stringent purity specifications required for API synthesis.
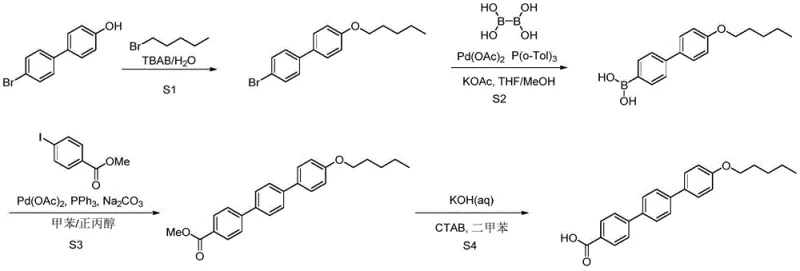
How to Synthesize p-pentoxy terphenyl formic acid Efficiently
The synthesis of this high-value antifungal intermediate is structured around a logical four-step sequence that prioritizes safety and yield at every stage. The process begins with a straightforward nucleophilic substitution to install the pentyloxy chain, followed by two sequential palladium-catalyzed cross-coupling reactions that build the aromatic core, and concludes with a basic hydrolysis to reveal the final carboxylic acid functionality. Each step has been optimized to use commercially available raw materials and standard laboratory equipment, removing barriers to entry for manufacturing partners. The detailed standardized synthesis steps see the guide below.
- Perform nucleophilic substitution of 4-hydroxy-4'-bromobiphenyl with 1-bromopentane using TBAB catalyst and NaOH base at 60-95°C.
- Execute the first Suzuki coupling with tetrahydroxydiboron using Pd(OAc)2 and P(o-Tol)3 ligand under nitrogen protection at 0-40°C.
- Complete the second Suzuki coupling with methyl 4-iodobenzoate using Pd(OAc)2 and PPh3, followed by alkaline hydrolysis to yield the final acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this Suzuki-based methodology offers tangible benefits that extend beyond simple chemical yield improvements. The elimination of hazardous reagents like n-butyllithium drastically reduces the safety risks associated with raw material storage and handling, leading to lower insurance premiums and simplified regulatory compliance documentation. The process operates under mild thermal conditions, avoiding the need for energy-intensive cryogenic cooling systems, which translates into significant operational cost savings over the lifetime of the production campaign. Furthermore, the use of stable boron reagents and robust palladium catalysts ensures consistent batch-to-batch quality, reducing the risk of production delays caused by failed reactions or out-of-specification results. This reliability is crucial for maintaining the continuity of supply for downstream API manufacturers who depend on timely delivery of key intermediates to meet their own production schedules. The simplified workup procedures, which rely on standard filtration and washing techniques rather than complex distillations or chromatography, further enhance the throughput capacity of the manufacturing facility.
- Cost Reduction in Manufacturing: The replacement of expensive and hazardous organometallic reagents with stable and cost-effective tetrahydroxydiboron significantly lowers the raw material cost per kilogram of the final product. By avoiding the need for specialized equipment to handle pyrophoric materials, capital expenditure is reduced, and the overall process becomes more economically viable for large-scale production. The high yields achieved in each step, particularly the near-quantitative conversion in the final hydrolysis step, minimize material loss and maximize the output from a fixed amount of starting material. Additionally, the ability to recover and recycle solvents like toluene and xylene further contributes to the overall cost efficiency of the manufacturing process. These factors combine to create a highly competitive cost structure that allows suppliers to offer better pricing without compromising on quality or safety standards.
- Enhanced Supply Chain Reliability: The reliance on commercially available and stable reagents ensures that the supply chain is less vulnerable to disruptions caused by the scarcity of specialized chemicals. Unlike n-butyllithium, which requires careful temperature control during transport and storage, the reagents used in this process can be handled under ambient conditions, simplifying logistics and warehousing requirements. The robustness of the reaction conditions means that production can be maintained even in facilities with varying levels of infrastructure, providing flexibility in sourcing and manufacturing locations. This stability is essential for building long-term partnerships with pharmaceutical companies that require guaranteed supply volumes to support their clinical and commercial programs. The reduced risk of safety incidents also means fewer unplanned shutdowns, ensuring a consistent flow of materials to the market.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing reaction conditions that are easily transferable from laboratory scale to multi-ton industrial reactors. The absence of highly toxic or persistent organic pollutants in the waste stream simplifies wastewater treatment and reduces the environmental footprint of the manufacturing operation. The use of aqueous workups and the ability to filter solid byproducts directly from the reaction mixture minimize the generation of liquid waste, aligning with modern green chemistry principles. This environmental compatibility is increasingly important for meeting the stringent sustainability goals of global pharmaceutical companies and regulatory bodies. The straightforward scale-up path ensures that production capacity can be increased rapidly to meet surges in demand without the need for extensive process re-validation or equipment modification.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this intermediate, based on the specific advantages outlined in the patent literature. These answers are derived from the detailed experimental data and comparative analysis provided in the invention disclosure, offering clarity on process capabilities and limitations. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing supply chains. The information provided here serves as a preliminary guide for further discussions with our technical sales team.
Q: Why is the Suzuki coupling method preferred over Grignard reagents for this intermediate?
A: The Suzuki coupling method avoids the use of highly moisture-sensitive and flammable reagents like n-butyllithium or Grignard reagents, significantly reducing safety risks and operational complexity in large-scale manufacturing environments.
Q: What are the key purity control measures in this synthesis route?
A: Purity is controlled through specific ligand selection such as tri(o-methylphenyl)phosphine which reduces byproducts, and rigorous aqueous workup procedures including thermal pulping and specific solvent washing steps to remove palladium residues and inorganic salts.
Q: Is this process suitable for commercial scale-up production?
A: Yes, the process operates under mild conditions (0-95°C) without requiring cryogenic temperatures or inert atmosphere glove boxes for reagent preparation, making it highly adaptable for multi-ton commercial production facilities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable p-pentoxy terphenyl formic acid Supplier
NINGBO INNO PHARMCHEM stands at the forefront of fine chemical manufacturing, leveraging advanced synthetic methodologies like the Suzuki coupling process to deliver high-quality intermediates for the global pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of p-pentoxy terphenyl formic acid meets the exacting standards required for antifungal API synthesis. Our commitment to safety and environmental stewardship aligns perfectly with the benefits of this patent-protected route, offering our partners a sustainable and reliable source of critical materials. By choosing us as your partner, you gain access to a wealth of technical expertise and a robust supply chain infrastructure designed to support your long-term growth.
We invite you to collaborate with us to optimize your supply chain and reduce your overall manufacturing costs through the adoption of this superior synthetic route. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage you to contact us to request specific COA data and route feasibility assessments that demonstrate the tangible benefits of switching to this safer and more efficient process. Let us help you secure a stable supply of high-purity intermediates that will drive the success of your antifungal drug development programs. Together, we can achieve new heights of efficiency and reliability in the production of life-saving medicines.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
