Advanced Synthesis of 4-Oxoquinoline Compounds for High-Purity HIV Integrase Inhibitors
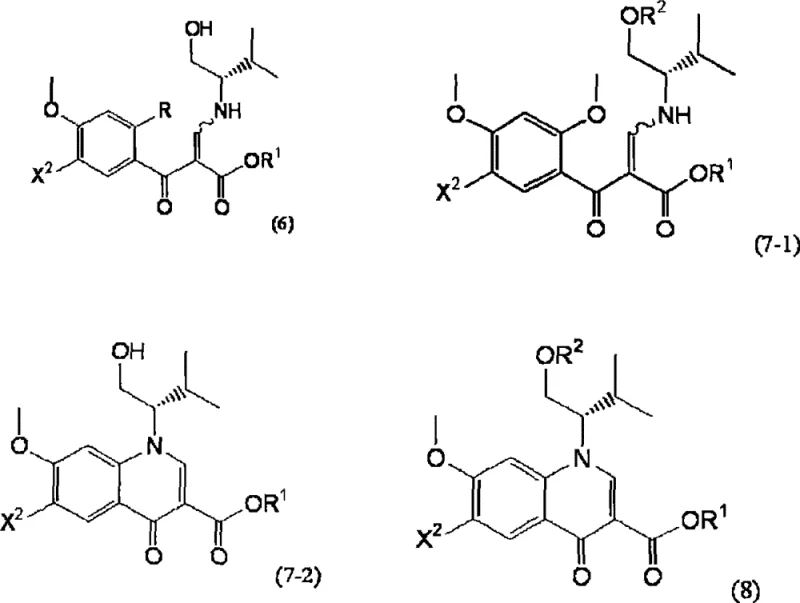
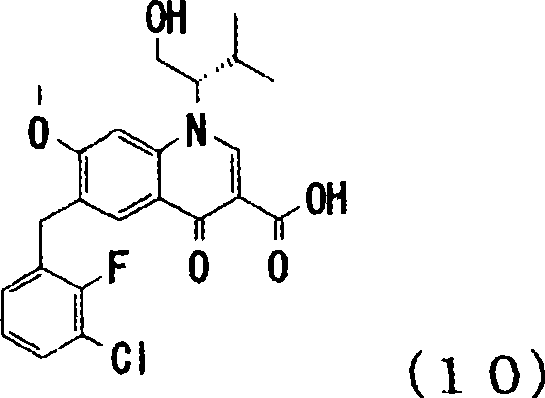
The pharmaceutical industry continuously seeks robust synthetic pathways for antiretroviral therapies, particularly for HIV integrase inhibitors which represent a cornerstone of modern treatment regimens. Patent CN101437801B presents a significant technological advancement by disclosing novel intermediates, specifically compounds represented by general formulae (6), (7-1), (7-2), and (8), which serve as precursors for high-value 4-oxoquinoline derivatives. These compounds are engineered to overcome the longstanding limitations associated with traditional synthesis routes, offering a streamlined approach to constructing the complex quinoline core essential for integrase inhibition. By utilizing highly assignable starting materials and optimizing protection group strategies, this methodology ensures a reliable supply chain for critical pharmaceutical intermediates. The technical breakthrough lies not just in the final product but in the stability and manageability of the intermediates throughout the multi-step sequence. For procurement specialists and R&D directors, understanding these structural nuances is vital for securing a consistent source of high-purity materials.
Historically, the manufacturing of 4-oxoquinoline compounds has been plagued by significant process inefficiencies and safety hazards that complicate commercial scale-up. Conventional methods, such as those referenced in prior art documents like WO04/046115, often rely on late-stage introduction of substituents, which leads to the formation of difficult-to-remove byproducts and corrosive waste streams. Specifically, the generation of hydrofluoric acid (HF) during cyclization or deprotection steps poses a severe threat to production facility integrity, requiring specialized corrosion-resistant equipment and complex neutralization protocols. Furthermore, the formation of sodium fluoride byproducts during alkoxylation steps necessitates additional purification stages that drastically reduce overall yield and increase processing time. These legacy processes also struggle with the stability of intermediates, where unprotected hydroxyl groups can lead to degradation or side reactions during storage and transport. The reliance on such volatile chemistry creates bottlenecks in the supply chain, making it difficult to guarantee the continuity of supply required for large-scale API production.
In stark contrast, the novel approach detailed in the patent data fundamentally restructures the synthetic timeline to mitigate these risks through the use of pre-functionalized and protected intermediates. By introducing the methoxy group early in the synthesis via compounds like (1-A) or (2-A), the process completely avoids the problematic late-stage alkoxylation that generates sodium fluoride waste. The strategic application of hydroxyl protecting groups, such as t-butyldimethylsilyl (TBDMS), on intermediates like compound (7-1) ensures that the sensitive alcohol functionalities remain inert during harsh cyclization conditions. This protection strategy allows the use of standard reaction vessels without the fear of HF corrosion, thereby simplifying the engineering requirements for manufacturing plants. Moreover, the stability of these protected intermediates facilitates easier handling and longer shelf life, reducing the risk of batch failure due to raw material degradation. This shift from reactive, unstable species to robust, protected scaffolds represents a paradigm shift in cost reduction in pharmaceutical intermediate manufacturing by minimizing waste and maximizing throughput.
Mechanistic Insights into Palladium-Catalyzed Coupling and Cyclization
The core of this synthetic innovation relies on a sophisticated sequence of cyclization and cross-coupling reactions that demand precise control over reaction parameters to ensure high fidelity. The formation of the quinoline ring system typically involves an intramolecular cyclization of a beta-ketoester derivative, such as compound (7-1), using bases like potassium carbonate in high-boiling solvents like toluene at temperatures ranging from 90°C to 120°C. This step is critical for establishing the rigid planar structure required for biological activity, and the presence of the protecting group prevents unwanted nucleophilic attacks on the side chain. Following ring closure, the introduction of the benzyl moiety is achieved through a palladium-catalyzed coupling reaction, often utilizing reagents like tris(dibenzylideneacetone)dipalladium(0) and triphenylphosphine. The use of organozinc reagents, such as 3-chloro-2-fluorobenzyl zinc bromide, generated in situ from zinc powder and the corresponding benzyl halide, allows for mild reaction conditions that preserve the stereochemical integrity of the chiral centers. This mechanistic pathway is designed to minimize the formation of homocoupling byproducts and ensures that the final product retains the necessary optical purity for therapeutic efficacy.
Impurity control is another pivotal aspect of this mechanism, particularly regarding the removal of heavy metal catalysts and inorganic salts post-reaction. The patent specifies rigorous workup procedures involving aqueous ammonium chloride and ethylenediamine solutions to effectively chelate and remove residual palladium species, which is essential for meeting regulatory limits on heavy metals in pharmaceutical products. Additionally, the process incorporates specific crystallization steps using solvent systems like methyl isobutyl ketone and heptane to selectively precipitate the desired product while leaving dimeric impurities in the solution. This level of purification is crucial because even trace amounts of structurally related impurities can affect the safety profile of the final HIV medication. The ability to reduce impurities to levels below 0.1% demonstrates the robustness of the purification protocol and highlights the suitability of this route for producing clinical-grade materials. Understanding these mechanistic details allows R&D teams to troubleshoot potential deviations and maintain strict quality control standards throughout the production lifecycle.
How to Synthesize 4-Oxoquinoline Intermediates Efficiently
Implementing this synthesis requires a disciplined approach to reaction monitoring and parameter control to replicate the high yields reported in the patent examples. The process begins with the halogenation of dimethoxybenzoic acid, followed by conversion to the acid chloride and subsequent condensation with malonic ester potassium to form the beta-ketoester backbone. Detailed standard operating procedures for each transformation, including specific temperature ramps and addition rates, are essential to prevent exothermic runaways and ensure consistent particle size distribution in the final crystals. Operators must be trained to handle air-sensitive reagents like organozinc compounds under inert atmospheres to prevent oxidation and loss of reactivity. The following guide outlines the critical operational milestones required to achieve successful scale-up.
- Perform halogenation of dimethoxybenzoic acid using bromine in acetic acid at 25°C to form 5-bromo-2,4-dimethoxybenzoic acid.
- Convert the acid to acid chloride using thionyl chloride, then react with malonic ester potassium to form the beta-ketoester intermediate.
- Execute cyclization using potassium carbonate in toluene at 110°C, followed by palladium-catalyzed benzylation to yield the final quinoline carboxylic acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthetic route offers tangible benefits that extend beyond simple chemical yield improvements. By eliminating the need for corrosion-resistant reactors required for handling hydrofluoric acid, manufacturers can utilize standard glass-lined or stainless steel equipment, which significantly lowers capital expenditure and maintenance costs. The stability of the protected intermediates reduces the need for expedited shipping and specialized cold-chain logistics, allowing for more flexible inventory management and reduced warehousing expenses. Furthermore, the simplified purification steps mean less solvent consumption and lower waste disposal fees, contributing to a more sustainable and cost-effective manufacturing footprint. These operational efficiencies translate directly into a more resilient supply chain capable of withstanding market fluctuations and raw material shortages. Ultimately, partnering with a supplier who utilizes this advanced technology ensures a steady flow of high-quality intermediates without the disruptions common to older, hazard-prone processes.
- Cost Reduction in Manufacturing: The elimination of hazardous reagents like hydrofluoric acid removes the necessity for expensive specialty equipment and complex safety protocols, leading to substantial operational savings. Additionally, the avoidance of late-stage functionalization reduces the number of purification steps required, minimizing solvent usage and energy consumption per kilogram of product. The higher stability of intermediates also decreases the rate of batch rejection due to degradation, further optimizing the cost of goods sold. These factors combine to create a leaner manufacturing process that is economically superior to conventional methods relying on unstable precursors.
- Enhanced Supply Chain Reliability: The use of robust, protected intermediates ensures that materials can be stored for extended periods without significant loss of potency, providing a buffer against supply chain disruptions. The reliance on commercially available starting materials and standard reagents reduces the risk of bottlenecks associated with sourcing exotic or custom-synthesized chemicals. This reliability is critical for maintaining continuous production schedules for life-saving HIV medications, where interruptions can have severe consequences for patient care. A stable supply of high-purity intermediates allows pharmaceutical companies to plan their production cycles with greater confidence and accuracy.
- Scalability and Environmental Compliance: The process is designed with industrial scalability in mind, utilizing reaction conditions that are easily transferable from laboratory to pilot and commercial scales. The reduction in hazardous waste generation, particularly the absence of fluoride-containing effluents, simplifies environmental compliance and reduces the burden on wastewater treatment facilities. This alignment with green chemistry principles not only mitigates regulatory risk but also enhances the corporate social responsibility profile of the manufacturing operation. Scalable processes that minimize environmental impact are increasingly preferred by global pharmaceutical partners seeking sustainable supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of these 4-oxoquinoline intermediates. These answers are derived directly from the technical specifications and beneficial effects outlined in the patent documentation to provide clarity on process capabilities. Understanding these details helps stakeholders make informed decisions regarding technology transfer and procurement strategies. The focus is on practical implementation and the tangible advantages offered by this specific synthetic methodology.
Q: Why is avoiding hydrofluoric acid (HF) generation critical in this synthesis?
A: Conventional methods often generate HF during the cyclization or deprotection steps, which causes severe corrosion to production facilities and necessitates complex removal operations. The novel process described in CN101437801B utilizes pre-methoxylated intermediates to bypass these hazardous conditions, significantly enhancing equipment longevity and operational safety.
Q: How does the new method improve the stability of synthetic intermediates?
A: The invention introduces specific hydroxyl protecting groups, such as t-butyldimethylsilyl, on key intermediates like compound (7-1) and (8). These protected forms exhibit superior stability under severe reaction conditions and during long-term storage, ensuring consistent quality for subsequent palladium-catalyzed coupling reactions.
Q: What are the purity specifications achievable with this process?
A: Through optimized crystallization steps using solvents like methyl isobutyl ketone and heptane, impurities such as dimeric byproducts can be reduced to levels below 0.1%. This rigorous purification protocol ensures the final 4-oxoquinoline compounds meet the stringent purity requirements necessary for active pharmaceutical ingredient (API) synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Oxoquinoline Compounds Supplier
As the demand for effective antiretroviral therapies continues to grow, securing a dependable source of high-quality intermediates is paramount for pharmaceutical developers. NINGBO INNO PHARMCHEM stands at the forefront of this sector, leveraging extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to meet global needs. Our commitment to excellence is underpinned by stringent purity specifications and rigorous QC labs that ensure every batch meets the highest international standards. We understand the critical nature of these supply chains and are dedicated to providing uninterrupted support for your API development projects. Our technical team is well-versed in the nuances of complex heterocyclic synthesis and ready to assist with any customization requirements.
We invite you to engage with our technical procurement team to discuss how our advanced manufacturing capabilities can support your specific project goals. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our optimized processes can reduce your overall production costs. We encourage potential partners to reach out for specific COA data and route feasibility assessments to verify our capacity to deliver on your quality and volume expectations. Together, we can accelerate the development of next-generation HIV treatments and improve patient outcomes worldwide.
