Advanced Synthesis of Heat-Resistant FMNTZ for Next-Generation Energetic Materials
Introduction to High-Density Energetic Material Synthesis
The pursuit of high-energy-density materials that maintain thermal stability under extreme conditions is a critical frontier in the development of next-generation propulsion and explosive systems. Patent CN113912557A introduces a breakthrough methodology for synthesizing 1-difluoromethyl-3,5-dinitro-1,2,4-triazole (FMNTZ), a novel compound that effectively balances high crystal density with exceptional heat resistance. By incorporating a difluoromethyl group into the triazole skeleton, this innovation addresses the traditional bottleneck where increasing nitro groups often compromises stability. The resulting material boasts a calculated density of up to 1.92 g/cm³ and a melting point of 307.7°C, positioning it as a superior alternative to conventional benchmarks like HMX.
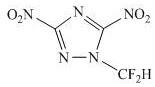
This patent outlines a robust two-step synthetic route starting from the readily available 3,5-diamino-1,2,4-triazole (DAT). The process leverages difluoromethylation followed by a diazotization reaction to install the energetic nitro groups efficiently. For R&D directors and procurement specialists, this represents a significant opportunity to access high-performance energetic intermediates with a streamlined supply chain. The method avoids the use of exotic catalysts, relying instead on standard inorganic bases and common organic solvents, which simplifies the path to commercial scale-up and ensures consistent quality control for high-purity energetic material manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional energetic materials, primarily based on C, H, O, and N frameworks, face a fundamental physical limitation regarding crystal density, which typically caps around 2.2 g/cm³. To achieve higher energy output, chemists historically increased the number of nitro groups; however, this approach invariably leads to decreased thermal stability and increased sensitivity, creating safety hazards during storage and transport. Furthermore, many existing synthesis routes for fluorine-containing energetic compounds involve complex multi-step sequences or require harsh reaction conditions that degrade yield and purity. The reliance on expensive transition metal catalysts or difficult-to-remove impurities often inflates production costs and complicates the waste treatment process, making large-scale manufacturing economically unviable for many specialized applications.
The Novel Approach
The methodology described in CN113912557A overcomes these hurdles by strategically introducing fluorine atoms, which possess high electronegativity and oxidizing potential, to enhance both density and stability without sacrificing safety. The use of ethyl bromodifluoroacetate as a difluoromethylating agent allows for the direct functionalization of the triazole ring under relatively mild conditions (90-110°C). This novel approach eliminates the need for complex protection-deprotection strategies often seen in heterocyclic chemistry. By utilizing a straightforward diazotization step to convert amino groups to nitro groups, the process achieves a theoretical detonation velocity of 9546.9 m/s, significantly outperforming HMX. This efficiency translates directly into reduced processing time and lower operational expenditures for manufacturers seeking cost reduction in advanced materials manufacturing.
Mechanistic Insights into Difluoromethylation and Diazotization
The core of this synthesis lies in the precise control of the difluoromethylation reaction, where 3,5-diamino-1,2,4-triazole reacts with ethyl bromodifluoroacetate in the presence of sodium carbonate. The inorganic base facilitates the deprotonation of the triazole nitrogen, enabling a nucleophilic attack on the bromodifluoroacetate ester. This step is critical for establishing the carbon-fluine bonds that contribute to the molecule's high density and thermal resilience. The reaction is conducted in acetone, a solvent that ensures good solubility of reactants while allowing for easy removal post-reaction. The subsequent purification via DMF dissolution and filtration removes inorganic salts effectively, ensuring the intermediate 1-difluoromethyl-3,5-diamino-1,2,4-triazole is obtained with high purity before the final energetic functionalization.
The second stage involves a classic diazotization mechanism adapted for energetic synthesis. The intermediate is dissolved in sulfuric acid and added to an aqueous sodium nitrite solution. Under controlled heating (50-70°C), the amino groups are converted into diazonium species which subsequently transform into nitro groups, releasing nitrogen gas and water as byproducts. This transformation is exothermic and requires careful temperature management to prevent decomposition, yet the patent demonstrates that the fluorine substituents stabilize the triazole ring sufficiently to withstand these conditions. Theoretical calculations using isodesmic reactions, as illustrated in the patent's computational analysis, confirm the thermodynamic stability of the final product.
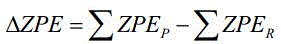
Understanding the electronic effects of the difluoromethyl group is essential for optimizing the reaction yield. The strong electron-withdrawing nature of the fluorine atoms influences the acidity of the triazole protons and the nucleophilicity of the ring nitrogens. This electronic modulation not only aids in the initial alkylation but also enhances the overall oxygen balance of the final molecule, contributing to its superior detonation pressure of 42.03 GPa. For technical teams, mastering these mechanistic nuances allows for fine-tuning of stoichiometry and temperature profiles to maximize the 81% yield observed in the second step, ensuring that the commercial scale-up of complex energetic compounds remains efficient and reproducible.
How to Synthesize 1-difluoromethyl-3,5-dinitro-1,2,4-triazole Efficiently
The synthesis protocol detailed in the patent provides a clear roadmap for laboratory and pilot-scale production, emphasizing safety and simplicity. The process begins with the preparation of the difluoromethylated intermediate, followed by the critical nitration step. Operators must adhere to strict temperature controls during the diazotization phase to manage the evolution of brown nitrogen oxide gases safely. The workup procedure involves neutralization and solvent evaporation, which are standard unit operations in fine chemical plants, minimizing the need for specialized equipment. Detailed standardized synthesis steps for optimizing yield and purity are provided in the guide below.
- React 3,5-diamino-1,2,4-triazole with ethyl bromodifluoroacetate and sodium carbonate in acetone at 90-110°C for 24 hours to form the intermediate.
- Purify the intermediate 1-difluoromethyl-3,5-diamino-1,2,4-triazole by dissolution in DMF and solvent evaporation.
- Perform diazotization by adding the intermediate sulfuric acid solution to aqueous sodium nitrite at 50-70°C to yield the final dinitro product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis route offers distinct logistical and economic benefits derived from its chemical simplicity. The reliance on commodity chemicals such as sodium carbonate, acetone, and sulfuric acid means that raw material sourcing is robust and less susceptible to the volatility associated with specialty catalysts. The elimination of transition metals from the process flow removes the need for expensive and time-consuming heavy metal scavenging steps, which are often a bottleneck in pharmaceutical and fine chemical manufacturing. This streamlining results in substantial cost savings and a significantly reduced environmental footprint, aligning with modern green chemistry initiatives.
- Cost Reduction in Manufacturing: The process utilizes inexpensive inorganic bases and common organic solvents, avoiding the high costs associated with noble metal catalysts or exotic reagents. The straightforward filtration and evaporation workup reduces energy consumption and labor hours compared to complex chromatographic purifications. Furthermore, the high yield in the second step (approximately 81%) minimizes raw material waste, leading to a more favorable cost-per-kilogram profile for the final energetic material.
- Enhanced Supply Chain Reliability: Starting materials like 3,5-diamino-1,2,4-triazole and ethyl bromodifluoroacetate are commercially available from multiple global suppliers, reducing the risk of single-source dependency. The robustness of the reaction conditions, which tolerate standard industrial heating and stirring equipment, ensures that production can be scaled rapidly to meet demand surges without requiring bespoke reactor modifications. This reliability is crucial for maintaining continuous supply lines for defense and aerospace applications.
- Scalability and Environmental Compliance: The synthesis generates minimal hazardous waste, primarily consisting of inorganic salts and aqueous acidic streams that can be neutralized and treated using standard effluent protocols. The absence of chlorinated solvents or persistent organic pollutants simplifies regulatory compliance and waste disposal logistics. The high thermal stability of the product also reduces risks during storage and transportation, lowering insurance and handling costs associated with sensitive energetic materials.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of FMNTZ. These insights are derived directly from the experimental data and theoretical calculations presented in the patent documentation. Understanding these factors is essential for evaluating the feasibility of integrating this material into existing formulation pipelines.
Q: What are the thermal stability characteristics of FMNTZ compared to HMX?
A: FMNTZ exhibits a melting point of 307.7°C, which is significantly higher than HMX (278°C), offering superior heat resistance for high-temperature applications.
Q: What is the theoretical detonation performance of this new compound?
A: Calculations predict a detonation velocity of approximately 9546.9 m/s and a detonation pressure of 42.03 GPa, surpassing standard HMX metrics.
Q: Is the synthesis process scalable for industrial production?
A: Yes, the method utilizes common solvents like acetone and DMF with straightforward filtration and evaporation steps, facilitating easy scale-up without complex purification requirements.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-difluoromethyl-3,5-dinitro-1,2,4-triazole Supplier
As the demand for high-performance energetic materials grows, partnering with an experienced CDMO is vital for translating patent innovations into commercial reality. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our rigorous QC labs and stringent purity specifications guarantee that every batch of 1-difluoromethyl-3,5-dinitro-1,2,4-triazole meets the exacting standards required for advanced propulsion and explosive applications, providing you with a secure and high-quality supply chain.
We invite you to collaborate with our technical team to explore the full potential of this fluorine-containing triazole derivative. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to support your R&D and manufacturing goals, ensuring a seamless transition from laboratory discovery to industrial deployment.
