Advanced Purification Technology for 9-Phenylacridine Enabling Commercial Scale-Up of Complex Electronic Chemicals
Advanced Purification Technology for 9-Phenylacridine Enabling Commercial Scale-Up of Complex Electronic Chemicals
The rapid evolution of the semiconductor and optoelectronic industries demands increasingly stringent specifications for raw materials, particularly photoinitiators used in advanced photoresist formulations. Patent CN112125845B introduces a groundbreaking purification methodology specifically designed for 9-phenylacridine, a critical intermediate in the synthesis of high-performance photocuring materials. This technology addresses the persistent challenge of removing trace diphenylamine, a stubborn impurity that significantly degrades the performance of integrated circuit products. By leveraging a sophisticated organic acid extraction technique, this innovation ensures that the final product meets the rigorous purity standards required for modern lithography applications. For global procurement teams and R&D directors, understanding the nuances of this patent is essential for securing a reliable 9-phenylacridine supplier capable of delivering consistent quality.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of 9-phenylacridine has relied on synthesis routes that generate significant quantities of unreacted diphenylamine, which is notoriously difficult to separate due to its structural similarity and solubility characteristics. Traditional purification protocols, such as those disclosed in earlier patents like CN101525392A, often employ harsh inorganic acids like sulfuric acid followed by neutralization with ammonia water. These legacy methods frequently result in the formation of black, sticky byproducts that complicate filtration and reduce overall yield. Furthermore, the use of strong inorganic reagents necessitates extensive wastewater treatment to handle heavy metal salts and acidic effluents, creating substantial environmental compliance burdens. The inability of these conventional techniques to reduce diphenylamine content below critical thresholds often renders the material unsuitable for high-end electronic applications, forcing manufacturers to seek costly alternatives or accept suboptimal performance in their final photoresist products.
The Novel Approach
In stark contrast to these archaic methods, the novel approach detailed in the patent utilizes a mild yet highly effective organic acid washing strategy that selectively targets the basic impurity without compromising the integrity of the target molecule. By employing aqueous solutions of organic acids such as acetic, citric, or glycolic acid, the process protonates the residual diphenylamine, converting it into a water-soluble salt that partitions cleanly into the aqueous phase. This selective extraction mechanism allows the 9-phenylacridine to remain in the organic solvent layer, facilitating a straightforward phase separation that eliminates the need for complex crystallization steps or activated carbon decolorization. The result is a streamlined workflow that not only enhances product purity to over 99% but also drastically simplifies the operational complexity, making it an ideal candidate for cost reduction in electronic chemical manufacturing where margin pressures are intense.
Mechanistic Insights into Organic Acid Extraction Purification
The core of this technological breakthrough lies in the precise manipulation of acid-base equilibria within a biphasic solvent system. 9-Phenylacridine is synthesized via the cyclization of diphenylamine and benzoic acid, typically catalyzed by zinc chloride, as illustrated in the reaction scheme below. However, the equilibrium nature of this condensation reaction inevitably leaves behind unreacted diphenylamine. The purification method exploits the fact that diphenylamine is a secondary amine with basic properties, whereas 9-phenylacridine is significantly less basic due to the delocalization of the nitrogen lone pair into the aromatic acridine system.
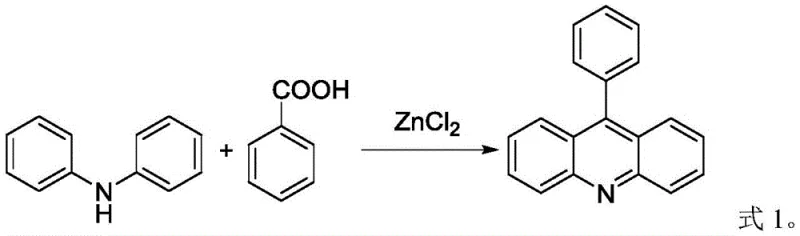
When the crude organic solution containing both species is contacted with a dilute organic acid solution, the diphenylamine readily accepts a proton to form a cationic ammonium species. This charged species exhibits high affinity for the polar aqueous environment and low affinity for the non-polar toluene phase, driving its migration out of the product stream. Conversely, the 9-phenylacridine remains largely unprotonated under these mild acidic conditions, retaining its lipophilicity and staying dissolved in the toluene. This differential partitioning is the key to achieving the ultra-low impurity levels of less than 0.05% diphenylamine. Furthermore, because organic acids are volatile or biodegradable, any trace carryover into the final product can be easily removed during the solvent stripping phase, ensuring no residual contamination affects the photochemical properties of the photoinitiator.
How to Synthesize 9-Phenylacridine Efficiently
The implementation of this purification protocol requires careful control of temperature and phase ratios to maximize extraction efficiency while minimizing solvent consumption. The process begins with a pre-wash to remove inorganic salts, followed by the critical acid extraction step, and concludes with a water wash to neutralize the organic phase. Detailed operational parameters, including specific mass ratios and thermal profiles, are critical for reproducibility on a manufacturing scale. For process engineers looking to implement this technology, the standardized synthetic steps are outlined in the guide below.
- Dissolve crude 9-phenylacridine in toluene at 75-90°C and wash with water to remove water-soluble inorganic impurities.
- Treat the organic phase with a 2-8% aqueous organic acid solution (e.g., acetic or citric acid) to extract residual diphenylamine.
- Wash the separated organic phase with water to remove residual acid, followed by distillation and crystallization to obtain high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders and procurement managers, the adoption of this purification technology translates directly into enhanced operational resilience and financial efficiency. The shift from hazardous inorganic reagents to benign organic acids mitigates regulatory risks associated with the transport and storage of corrosive materials, thereby reducing insurance premiums and safety compliance costs. Additionally, the simplicity of the liquid-liquid separation process reduces the dependency on specialized filtration equipment and skilled labor, allowing for faster batch turnover times. This operational agility is crucial for maintaining supply continuity in the volatile electronics market, where demand spikes can occur with little notice. By partnering with a manufacturer utilizing this advanced route, buyers can secure a more stable supply of high-purity intermediates without the risk of production bottlenecks caused by complex purification failures.
- Cost Reduction in Manufacturing: The elimination of expensive activated carbon treatments and the reduction in solvent usage due to higher extraction efficiency lead to substantial cost savings in the overall production budget. By avoiding the generation of heavy metal-laden sludge, the facility also saves significantly on hazardous waste disposal fees, which are a major cost driver in fine chemical manufacturing. The higher yield obtained through this gentle purification method means that less raw material is wasted, further improving the cost-per-kilogram metric for the final API or electronic chemical. These cumulative efficiencies allow suppliers to offer more competitive pricing structures without compromising on quality margins.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals like toluene and acetic acid, which are widely available in the global market, ensures that production is not held hostage by the scarcity of exotic reagents. This raw material accessibility guarantees that manufacturing schedules can be maintained even during periods of regional supply chain disruption. The robustness of the process against minor variations in feedstock quality further enhances reliability, as the extraction step is forgiving and consistently delivers the required purity specifications. Consequently, customers can rely on shorter lead times and more predictable delivery windows for their high-purity 9-phenylacridine orders.
- Scalability and Environmental Compliance: The process is inherently scalable, moving seamlessly from laboratory benchtop experiments to multi-ton reactor systems without the need for fundamental process redesign. The absence of persistent organic pollutants and heavy metals in the waste stream simplifies the environmental permitting process and aligns with increasingly strict global green chemistry initiatives. This environmental compatibility future-proofs the supply chain against tightening regulations, ensuring long-term viability for the manufacturing site. Companies prioritizing sustainability in their vendor selection criteria will find this method particularly attractive, as it demonstrates a commitment to responsible chemical stewardship.
Frequently Asked Questions (FAQ)
To assist technical decision-makers in evaluating the feasibility of this purification route for their specific applications, we have compiled answers to common inquiries regarding the process capabilities and output quality. These insights are derived directly from the experimental data and technical disclosures within the patent documentation, providing a transparent view of the technology's performance. Understanding these details is vital for assessing whether this method aligns with your specific purity requirements and processing constraints.
Q: What is the final purity of 9-phenylacridine achieved by this method?
A: The patented purification method consistently achieves a product purity exceeding 99%, with residual diphenylamine content reduced to below 0.05%.
Q: Which organic acids are suitable for the extraction process?
A: The process is compatible with various organic acids including acetic acid, glycolic acid, citric acid, propionic acid, and malonic acid.
Q: Does this method introduce metal ion contamination?
A: No, unlike traditional inorganic acid/base methods, this protocol utilizes organic acids which avoid the introduction of metal ions, simplifying downstream waste treatment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 9-Phenylacridine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent theory to commercial reality requires deep technical expertise and robust infrastructure. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of this advanced purification method are fully realized at an industrial level. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of 9-phenylacridine meets the exacting standards required for photoresist and optoelectronic applications. We are committed to delivering not just a chemical product, but a comprehensive solution that enhances your downstream manufacturing efficiency.
We invite you to engage with our technical procurement team to discuss how this innovative purification technology can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic benefits of switching to this superior grade of material. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project needs, ensuring a seamless partnership that drives mutual growth and technological advancement.
