Advanced Manufacturing of Tetrahydropyran Ketone Intermediates for Global Pharma Supply Chains
Advanced Manufacturing of Tetrahydropyran Ketone Intermediates for Global Pharma Supply Chains
The landscape of fine chemical manufacturing is constantly evolving, driven by the need for more efficient, safer, and cost-effective synthetic routes for complex molecular scaffolds. A pivotal advancement in this domain is detailed in patent CN1898228A, which discloses a novel preparation method for 3-(4-tetrahydropyran)-3-oxopropionic acid alkyl ester compounds and their precursors, 4-acyl tetrahydropyrans. These tetrahydropyran derivatives serve as critical building blocks in the synthesis of various active pharmaceutical ingredients (APIs) and agrochemical agents, offering unique structural rigidity and metabolic stability. The technical breakthrough described in this intellectual property lies in its ability to construct these valuable intermediates under significantly milder conditions compared to historical precedents, utilizing a direct acylation strategy that bypasses the need for hazardous organometallic reagents. For R&D directors and procurement specialists alike, understanding the nuances of this chemistry is essential for optimizing supply chains and reducing the overall cost of goods sold (COGS) in drug development.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 4-acyl tetrahydropyrans and related beta-keto esters has been plagued by operational complexity and safety concerns inherent to traditional organometallic chemistry. As noted in the background art of the patent, previous methods often relied on the use of Grignard reagents, which require strictly anhydrous conditions, cryogenic temperatures, and meticulous handling to prevent runaway exotherms or decomposition. Furthermore, the conventional pathways frequently involved multi-step sequences, such as the synthesis of 4-cyanotetrahydropyran-4-carboxylic acid ethyl ester followed by hydrolysis and high-temperature decarboxylation, before finally reacting with Grignard reagents to install the acyl group. This convoluted approach not only results in cumulative yield losses at each stage but also generates significant amounts of chemical waste, including magnesium salts and halogenated by-products, which complicate downstream purification and environmental compliance. The reliance on such aggressive reagents inherently limits the scalability of the process and increases the barrier to entry for reliable suppliers who must invest heavily in specialized containment and safety infrastructure.
The Novel Approach
In stark contrast to these cumbersome legacy methods, the invention disclosed in CN1898228A introduces a streamlined, base-catalyzed condensation reaction that directly couples a 4-acyltetrahydropyran with a carbonic acid diester. This innovative route allows for the formation of the target 3-(4-tetrahydropyran)-3-oxopropionate structure in a single operational step, dramatically simplifying the process flow. By employing readily available bases such as sodium hydride or alkali metal alcoholates, the reaction proceeds efficiently at moderate temperatures ranging from 35°C to 130°C, eliminating the need for cryogenic cooling. The strategic removal of by-product alcohols via distillation during the reaction drives the equilibrium towards the desired product, ensuring high conversion rates without the need for excessive reagent loading. This methodology not only enhances the safety profile of the manufacturing process by avoiding pyrophoric reagents but also significantly improves the atom economy, making it an ideal candidate for green chemistry initiatives in modern pharmaceutical manufacturing.
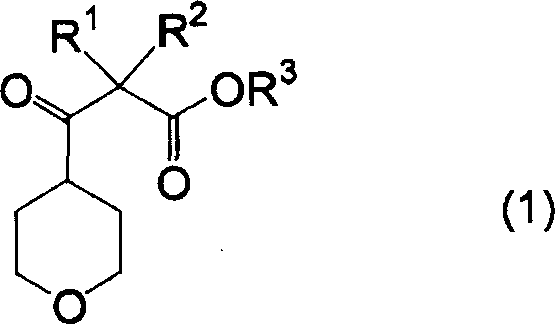
Mechanistic Insights into Base-Catalyzed Acylation
The core of this technological advancement relies on a classic yet optimized Claisen-type condensation mechanism, adapted specifically for the sterically hindered environment of the tetrahydropyran ring. The reaction initiates with the deprotonation of the alpha-carbon adjacent to the carbonyl group of the 4-acyltetrahydropyran substrate by a strong base, generating a resonance-stabilized enolate intermediate. This nucleophilic species then attacks the electrophilic carbonyl carbon of the carbonic acid diester, leading to the formation of a tetrahedral intermediate. Subsequent elimination of an alkoxide group restores the carbonyl functionality, yielding the beta-keto ester product. The choice of base is critical; the patent highlights the efficacy of sodium hydride and sodium methylate, which provide the necessary basicity to drive enolization without inducing unwanted side reactions such as ring-opening of the tetrahydropyran moiety. This selectivity is paramount for maintaining the integrity of the heterocyclic scaffold, which is often sensitive to harsh acidic or nucleophilic conditions.
Furthermore, the patent describes a complementary process for preparing the requisite 4-acyl tetrahydropyran starting materials via an acid-catalyzed decarboxylation of 4-acyl-4-alkoxycarbonyl tetrahydropyrans. This upstream transformation involves heating the precursor in the presence of inorganic acids like sulfuric acid or hydrochloric acid, facilitating the loss of carbon dioxide and an alcohol molecule to reveal the ketone functionality. Understanding this mechanistic pathway is crucial for quality control, as incomplete decarboxylation can lead to impurities that persist through the subsequent acylation step. By optimizing the acid concentration and temperature—typically between 70°C and 150°C—manufacturers can ensure near-quantitative conversion, thereby minimizing the burden on purification units and ensuring a consistent supply of high-purity intermediates for downstream coupling reactions.
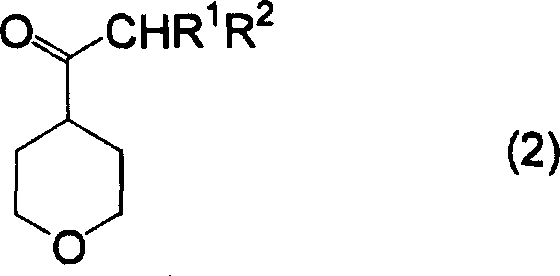
How to Synthesize 3-(4-Tetrahydropyran)-3-Oxopropionate Efficiently
Implementing this synthesis route requires careful attention to reaction parameters to maximize yield and minimize impurity formation. The process begins with the precise charging of the 4-acyltetrahydropyran substrate and the carbonic acid diester into a reactor equipped with a distillation column to facilitate the continuous removal of volatile alcohols. The addition of the base catalyst must be controlled to manage the initial exotherm, after which the reaction mixture is heated to the optimal range of 80°C to 85°C. Detailed standard operating procedures regarding stoichiometry, solvent selection, and workup protocols are essential for reproducibility on a commercial scale. For a comprehensive guide on the specific experimental conditions and isolation techniques validated in the patent examples, please refer to the standardized synthesis steps outlined below.
- Prepare the reaction vessel with 4-acyltetrahydropyran and a suitable solvent such as toluene or DMF.
- Add carbonic acid diester (e.g., dimethyl carbonate) and a base catalyst like sodium methylate or sodium hydride.
- Heat the mixture to 80-130°C while distilling off by-product alcohols to drive the equilibrium forward.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, the adoption of this patented methodology offers substantial benefits that extend beyond mere technical feasibility. By transitioning away from Grignard-based chemistries, organizations can realize significant cost reductions in API manufacturing through the elimination of expensive, moisture-sensitive reagents and the associated infrastructure required for their safe handling. The simplified workflow reduces the number of unit operations, which directly correlates to lower labor costs, reduced energy consumption, and decreased equipment occupancy time. Moreover, the use of commodity chemicals such as dimethyl carbonate and sodium methylate ensures a robust and resilient supply chain, mitigating the risks associated with the sourcing of specialized organometallics that are often subject to market volatility and logistical constraints.
- Cost Reduction in Manufacturing: The economic impact of this process is driven primarily by the drastic simplification of the synthetic route. By consolidating multiple reaction steps into a single pot operation and utilizing inexpensive, bulk-available reagents, the overall variable cost of production is significantly lowered. The avoidance of heavy metal catalysts or pyrophoric reagents also removes the need for costly scavenging steps and specialized waste disposal protocols, further enhancing the financial viability of the process. Additionally, the high yields reported in the patent examples suggest that raw material utilization is optimized, reducing the cost per kilogram of the final active intermediate and improving the overall margin profile for the finished drug product.
- Enhanced Supply Chain Reliability: Supply continuity is a critical metric for procurement managers, and this method excels by relying on a portfolio of stable, non-hazardous starting materials. Unlike processes dependent on custom-synthesized organolithium or Grignard reagents, which can suffer from long lead times and strict transportation regulations, the reagents used here are globally sourced commodities. This accessibility ensures that production schedules are less likely to be disrupted by raw material shortages. Furthermore, the mild reaction conditions reduce the risk of batch failures due to thermal runaways or sensitivity to ambient moisture, leading to more predictable manufacturing cycles and reliable delivery timelines for downstream customers.
- Scalability and Environmental Compliance: The transition from laboratory to commercial scale is seamless with this technology due to its inherent safety and simplicity. The absence of cryogenic requirements and the use of standard solvents like toluene or ethanol allow the process to be easily scaled from pilot plants to multi-ton reactors without significant engineering hurdles. From an environmental standpoint, the process aligns with green chemistry principles by minimizing waste generation and avoiding the use of persistent organic pollutants or toxic heavy metals. This compliance not only reduces the environmental footprint but also simplifies regulatory filings and audits, accelerating the time-to-market for new pharmaceutical products that utilize these intermediates.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of this technology for their specific applications, we have compiled a set of frequently asked questions based on the detailed disclosures within the patent literature. These inquiries address common concerns regarding reaction scope, impurity profiles, and scalability, providing a clear overview of what can be expected when implementing this synthesis route. The answers are derived directly from the experimental data and technical specifications provided in the source documentation, ensuring accuracy and relevance for process development scientists.
Q: What are the advantages of this new synthesis method over traditional Grignard routes?
A: This method eliminates the need for hazardous Grignard reagents and complex multi-step sequences, operating under milder conditions with simpler workup procedures.
Q: What purity levels can be achieved with this decarboxylation process?
A: The patent data indicates that high purity levels, such as 99% by liquid chromatography, are achievable through standard distillation and extraction techniques.
Q: Is this process scalable for industrial production of API intermediates?
A: Yes, the use of common solvents like toluene and stable reagents like sodium methylate makes the process highly amenable to large-scale commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-(4-Tetrahydropyran)-3-Oxopropionate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of your drug development programs. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistency and precision. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. By leveraging advanced synthetic methodologies like the one described in CN1898228A, we are able to offer cost-effective solutions without compromising on the quality or safety of the materials we supply to the global marketplace.
We invite you to collaborate with us to explore how this innovative chemistry can benefit your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume and purity requirements. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise in fine chemical manufacturing can become a strategic asset to your supply chain.
