Advanced Ferrocenyl Methyl-1,2,3-Triazole Aromatic Amines for High-Energy Solid Propellants
Advanced Ferrocenyl Methyl-1,2,3-Triazole Aromatic Amines for High-Energy Solid Propellants
In the rapidly evolving landscape of aerospace propulsion and energetic materials, the demand for high-performance burning rate catalysts that offer superior thermal stability and reduced migration is paramount. Patent CN110294780B introduces a groundbreaking class of aromatic amine burning rate catalysts containing ferrocenyl methyl-1,2,3-triazole groups, representing a significant leap forward in solid propellant technology. This innovation addresses the critical limitations of traditional ferrocene derivatives by integrating nitrogen-rich heterocyclic structures that enhance energy density while simultaneously mitigating volatility issues through robust hydrogen bonding networks. For R&D directors and procurement specialists in the defense and aerospace sectors, understanding the mechanistic advantages and synthetic accessibility of these compounds is essential for securing a reliable supply chain of next-generation energetic additives.
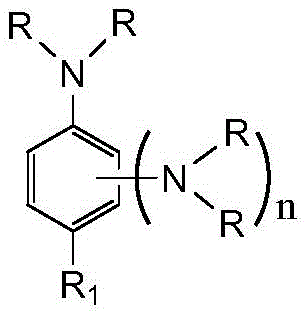
The strategic incorporation of the 1,2,3-triazole moiety into the ferrocene architecture creates a multifunctional molecule that serves both as a combustion catalyst and an energetic binder component. Unlike conventional ferrocene burn rate catalysts which often suffer from sublimation and migration within the propellant matrix over time, these novel derivatives exhibit exceptional resistance to volatilization due to the strong intermolecular forces generated by the triazole and aromatic amine nitrogen atoms. This structural integrity ensures consistent ballistic performance throughout the shelf life of missile propellant charges, thereby reducing the invisible costs associated with national defense basic reserves and enhancing the operational reliability of weapon systems in diverse environmental conditions.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the solid propellant industry has relied heavily on ferrocene and its simple derivatives, such as n-butyl ferrocene and catocene, to regulate burning rates and reduce pressure indices. However, these legacy compounds are plagued by inherent physicochemical deficiencies, specifically their tendency to migrate and volatilize easily under natural storage conditions. This migration leads to non-uniform distribution within the propellant grain, causing unpredictable burning behaviors and potentially catastrophic failures in rocket engine performance. Furthermore, the synthesis of many high-performance ferrocene derivatives has traditionally involved complex multi-step processes requiring harsh reaction conditions, expensive precursors, and difficult purification protocols, which collectively drive up the manufacturing cost and limit the scalability of these critical materials for large-scale aerospace applications.
The Novel Approach
The methodology disclosed in the patent offers a transformative solution by utilizing a modular synthetic strategy that combines the catalytic power of ferrocene with the energetic benefits of nitrogen-rich triazoles. By employing a copper-catalyzed azide-alkyne cycloaddition (CuAAC), often referred to as click chemistry, the process allows for the efficient coupling of azidomethylferrocene with various poly-alkynyl aromatic amines under mild conditions. This approach not only simplifies the operational workflow by eliminating the need for extreme temperatures or pressures but also yields products with precisely defined structures that maximize hydrogen bonding potential. The resulting catalysts demonstrate superior thermal stability, with weight loss initial temperatures exceeding 300°C, and significantly lower migration distances compared to commercial standards, marking a distinct advancement in propellant additive technology.
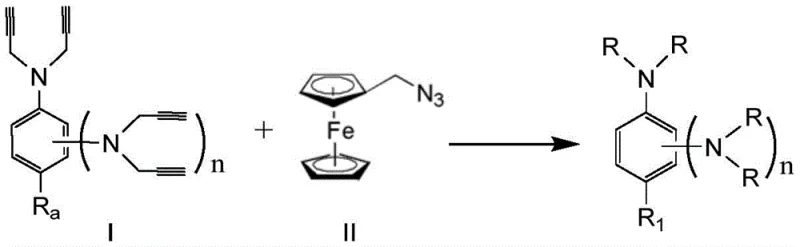
Mechanistic Insights into CuAAC Catalyzed Triazole Formation
The core of this technological breakthrough lies in the efficiency and atom economy of the copper(I)-catalyzed azide-alkyne cycloaddition reaction, which facilitates the formation of the 1,2,3-triazole ring with high regioselectivity. In this mechanism, the terminal alkyne groups on the aromatic amine precursors react with the azide functionality on the ferrocene derivative in the presence of a copper catalyst system generated in situ from copper sulfate pentahydrate and sodium ascorbate. This redox system maintains the active copper(I) species necessary for the cycloaddition to proceed smoothly at room temperature in methanol, ensuring that the sensitive ferrocene moiety remains intact while forming the robust triazole linkage. The reaction kinetics are favorable, typically reaching completion within 20 to 24 hours, which allows for high throughput in a manufacturing setting without compromising the yield or purity of the final energetic material.
Beyond the synthesis mechanics, the functional performance of these catalysts is driven by the unique electronic and steric properties of the triazole-ferrocene hybrid structure. The nitrogen-rich triazole ring possesses a positive heat of formation, which contributes additional energy to the propellant system upon decomposition, effectively boosting the specific impulse of the rocket motor. Simultaneously, the lone pair electrons on the triazole nitrogens and the aromatic amine nitrogens engage in extensive hydrogen bonding interactions, creating a supramolecular network that physically anchors the ferrocene units within the polymer matrix. This anchoring effect is the key to suppressing migration and volatility, ensuring that the catalyst remains homogeneously distributed to provide consistent catalytic activity on the decomposition of ammonium perchlorate and hexogen throughout the operational life of the propellant.
How to Synthesize Ferrocenyl Triazole Catalysts Efficiently
The synthesis of these high-value burning rate catalysts follows a streamlined two-stage protocol that begins with the preparation of the poly-alkynyl aromatic amine precursors followed by the click chemistry coupling step. The initial stage involves the nucleophilic substitution of aromatic amines, such as aniline or phenylenediamines, with 3-bromopropyne in a polar aprotic solvent like DMF, utilizing potassium carbonate as a base to scavenge the generated acid. Once the alkyne-functionalized amine is secured, it is subjected to the cycloaddition reaction with azidomethylferrocene under inert atmosphere conditions to prevent oxidation of the sensitive reagents. The detailed standardized synthesis steps, including specific molar ratios, solvent volumes, and purification techniques via column chromatography, are outlined in the technical guide below for immediate implementation by process chemists.
- Prepare the alkyne precursor by reacting aromatic amines (such as aniline or phenylenediamine) with 3-bromopropyne in DMF using potassium carbonate as a base at 60°C.
- Mix the alkyne precursor with azidomethylferrocene in methanol under a nitrogen atmosphere to ensure an inert reaction environment.
- Add catalytic amounts of copper sulfate pentahydrate and sodium ascorbate, stir at room temperature for 20-24 hours, then filter and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel catalyst technology presents a compelling value proposition centered on cost optimization and supply security. The synthetic route described in the patent utilizes commodity chemicals such as aniline derivatives, propargyl bromide, and simple copper salts, all of which are readily available in the global chemical market at competitive price points. By avoiding the use of precious metal catalysts like palladium or rhodium, which are subject to extreme price volatility and supply chain bottlenecks, manufacturers can achieve significant cost reduction in burning rate catalyst manufacturing while maintaining a resilient supply chain that is less susceptible to geopolitical disruptions affecting rare earth or precious metal markets.
- Cost Reduction in Manufacturing: The operational simplicity of the synthesis process directly translates to lower capital expenditure and operating costs for production facilities. Since the reaction proceeds efficiently at room temperature without the need for specialized high-pressure reactors or cryogenic cooling systems, the energy consumption per kilogram of product is drastically minimized. Furthermore, the high yields reported in the patent examples, often exceeding 80%, indicate a material-efficient process that reduces waste generation and raw material consumption, thereby enhancing the overall economic viability of producing these advanced energetic additives on a commercial scale.
- Enhanced Supply Chain Reliability: The reliance on robust and well-established chemical transformations ensures that the production of these catalysts can be scaled up rapidly to meet surging demand from the aerospace and defense sectors. The starting materials, including various isomers of phenylenediamine and ferrocene derivatives, are produced by multiple suppliers globally, mitigating the risk of single-source dependency. This diversification of the raw material base allows for greater flexibility in sourcing strategies, ensuring continuous production schedules and reliable delivery timelines for critical propellant formulations even during periods of global supply chain stress.
- Scalability and Environmental Compliance: From an environmental and regulatory perspective, the aqueous workup and standard organic solvent usage align well with existing waste management infrastructure in fine chemical plants. The absence of toxic heavy metals in the final product, combined with the high thermal stability that reduces the risk of accidental decomposition during transport and storage, simplifies compliance with hazardous material regulations. This ease of handling and disposal makes the technology highly attractive for large-scale industrial adoption, facilitating the seamless transition from laboratory benchtop synthesis to multi-ton annual production capacities required for national defense programs.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of ferrocenyl triazole catalysts in solid propellant formulations. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a factual basis for decision-making.
Q: How does the triazole group improve the stability of ferrocene catalysts?
A: The introduction of the 1,2,3-triazole group and the nitrogen atoms in the aromatic amine backbone facilitates the formation of intermolecular hydrogen bonds. This structural feature significantly reduces migration and volatility under natural conditions compared to traditional ferrocene derivatives.
Q: What is the primary advantage of these catalysts for solid propellants?
A: These catalysts not only provide the necessary iron content for combustion catalysis but also introduce nitrogen-rich heterocyclic groups with positive formation enthalpy. This dual functionality improves the energy level of the propellant while effectively lowering the decomposition temperature of ammonium perchlorate.
Q: Is the synthesis process scalable for industrial production?
A: Yes, the preparation method utilizes simple operational steps involving room temperature stirring and standard filtration. The use of readily available reagents like copper sulfate and sodium ascorbate suggests a pathway that can be adapted for commercial scale-up without requiring exotic high-pressure equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ferrocenyl Triazole Catalyst Supplier
As the global demand for high-energy, low-signature propellants continues to rise, partnering with an experienced chemical manufacturer is crucial for translating patented innovations into reality. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to deliver these complex ferrocene derivatives with unmatched consistency. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced analytical instrumentation to ensure that every batch meets stringent purity specifications, guaranteeing the ballistic performance and safety of your final energetic formulations.
We invite you to collaborate with our technical team to explore how these advanced burning rate catalysts can optimize your specific propellant architectures. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits of switching to our triazole-ferrocene hybrids. We encourage you to contact our technical procurement team today to obtain specific COA data and comprehensive route feasibility assessments tailored to your project requirements, ensuring a seamless integration of these high-performance materials into your supply chain.
