Revolutionizing Energetic Materials: Commercial Scale-Up of High-Density ZXC-17 Fusion-Cast Explosives
Revolutionizing Energetic Materials: Commercial Scale-Up of High-Density ZXC-17 Fusion-Cast Explosives
The global demand for high-performance energetic materials that balance extreme power with enhanced safety profiles has driven significant innovation in the field of fusion-cast explosives. Patent CN109810006B introduces a breakthrough compound, 3,4,5-trifluoro-2,6-dinitroaniline (designated as ZXC-17), which represents a paradigm shift away from traditional carriers like TNT. This novel fluorinated nitroaniline derivative addresses critical limitations in modern munitions, offering a melting point of 92.20 °C and an exceptional single crystal density of 1.972 g/cm³. For R&D directors and procurement specialists seeking reliable energetic material suppliers, ZXC-17 presents a compelling alternative that combines superior detonation performance with reduced mechanical sensitivity. The structural integrity and electronic properties of this molecule, as depicted below, are engineered to maximize packing density while maintaining thermal stability during the casting process.
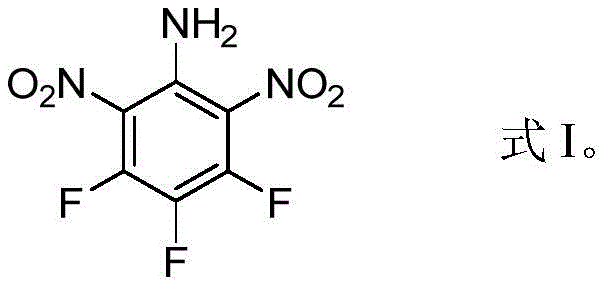
Unlike conventional explosives that often suffer from volume changes upon cooling or excessive sensitivity to impact, ZXC-17 demonstrates an impact sensitivity of 62J, which is markedly safer than the 29J observed in TNT. This improvement is crucial for supply chain heads concerned with the safe transport and storage of hazardous materials. The synthesis pathway described in the patent utilizes a straightforward nitration strategy that avoids complex transition metal catalysis, thereby simplifying the purification process and reducing the environmental footprint associated with heavy metal waste. As the industry moves towards greener synthesis methods, the ability to produce high-energy insensitive explosives using standard nitrating agents positions ZXC-17 as a key candidate for next-generation military and civil explosion applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, 2,4,6-trinitrotoluene (TNT) has served as the primary liquid-phase carrier for fusion-cast explosives, dominating the market throughout the 20th century due to its favorable melting characteristics. However, the reliance on TNT-based formulations has introduced significant operational and environmental challenges that modern defense contractors can no longer ignore. TNT production generates harmful waste streams that pose serious health risks to workers and require costly remediation protocols. Furthermore, TNT-based cast explosives are prone to physical defects such as oil leakage, shrinkage, cavitation, and embrittlement during the cooling phase, which can compromise the structural integrity of the munition and affect its sensitivity and vulnerability. Earlier attempts to replace TNT with compounds like 2,3,4-trifluoro-1,5-dinitrobenzene (ZXC-16) offered improvements in density and detonation velocity, yet they still fell short of the theoretical performance limits required for advanced warhead designs. The mechanical sensitivity of many high-energy alternatives also remains a critical bottleneck, as materials that are too sensitive pose unacceptable risks during the high-throughput manufacturing processes required for mass production.
The Novel Approach
The introduction of 3,4,5-trifluoro-2,6-dinitroaniline (ZXC-17) via the methodology in CN109810006B effectively resolves these contradictions by leveraging the unique electronic effects of fluorine substitution on the aromatic ring. This novel approach yields a compound with a density of 1.972 g/cm³, which is approximately 0.332 g/cm³ higher than that of TNT, directly translating to higher detonation pressures and velocities essential for penetrating hard targets. The strategic placement of fluorine atoms not only enhances the density but also modulates the intermolecular forces to achieve a melting point of 92.20 °C, ensuring excellent fluidity for casting without the risk of premature decomposition. Moreover, the synthesis route is designed for industrial scalability, utilizing robust reaction conditions that tolerate minor variations in temperature and stoichiometry without compromising yield. By shifting the molecular architecture to a trifluoro-dinitroaniline scaffold, manufacturers can achieve a substantial cost reduction in energetic material manufacturing through improved yield efficiency and simplified downstream processing, eliminating the need for extensive recrystallization steps often required for less pure intermediates.
Mechanistic Insights into Nitration of Fluorinated Aniline Derivatives
The core chemical transformation driving the production of ZXC-17 is an electrophilic aromatic substitution, specifically a nitration reaction performed on a protected aniline precursor. To prevent the oxidation of the amino group and to control the regioselectivity of the nitration, the synthesis begins with the acylation of 3,4,5-trifluoroaniline. This protection step converts the highly reactive amine into a more stable amide or carbamate derivative, referred to as Raw Material 1 in the patent documentation. The choice of acylating agent, such as acetyl chloride, acetic anhydride, or chloroformates, allows for fine-tuning of the intermediate's solubility and reactivity profile. Once protected, the aromatic ring undergoes nitration in a highly acidic medium, typically consisting of 95-98% concentrated sulfuric acid combined with a nitrate source like potassium nitrate or ammonium nitrate. The strong electron-withdrawing nature of the fluorine atoms and the protecting group deactivates the ring, necessitating vigorous conditions to introduce the two nitro groups at the 2 and 6 positions ortho to the nitrogen substituent.
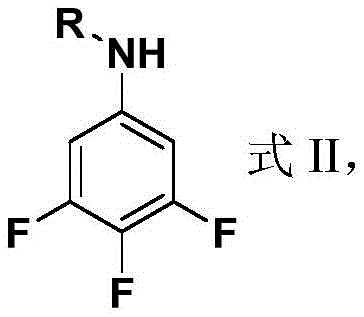
Following the nitration, the reaction mixture is quenched into ice water, causing the precipitation of the target compound. Interestingly, the patent indicates that the final product retains the amino functionality, suggesting that the acyl protecting group is hydrolyzed during the acidic workup or that the nitration conditions are sufficiently robust to proceed despite the steric bulk. The mechanism relies on the generation of the nitronium ion (NO₂⁺) in situ, which attacks the electron-deficient aromatic ring. The presence of three fluorine atoms significantly alters the electron density distribution, making the positions ortho to the amino group the most viable sites for electrophilic attack despite the overall deactivation of the ring. This precise control over impurity formation is vital for R&D teams, as side reactions such as ipso-substitution of fluorine or over-nitration can lead to unstable byproducts. The process described ensures high purity by leveraging the low solubility of the final dinitroaniline in aqueous media, allowing for easy separation from acidic residues and unreacted starting materials through simple filtration.
How to Synthesize 3,4,5-Trifluoro-2,6-Dinitroaniline Efficiently
The synthesis of ZXC-17 is optimized for both laboratory precision and industrial throughput, utilizing a two-stage protocol that prioritizes safety and yield consistency. The initial stage involves the preparation of the protected intermediate under mild conditions to ensure complete conversion of the trifluoroaniline starting material. The subsequent nitration stage is carefully controlled using an ice-water bath to manage the exothermic nature of the reaction, preventing thermal runaway while ensuring complete dissolution of the nitrate salts in the sulfuric acid matrix. Detailed standardized operating procedures regarding stoichiometry, temperature ramping, and quenching rates are critical for reproducing the high yields reported in the patent examples. For process engineers looking to implement this technology, the following guide outlines the critical operational parameters derived from the intellectual property.
- Prepare the protected intermediate (Raw Material 1) by reacting 3,4,5-trifluoroaniline with an acylating agent such as acetyl chloride or methyl chloroformate in acetonitrile with triethylamine.
- Perform the nitration reaction by adding the protected intermediate to a mixture of concentrated sulfuric acid (95-98%) and a nitrate source (e.g., potassium nitrate) under ice-water bath conditions.
- Quench the reaction mixture into ice water to precipitate the crude product, followed by filtration, washing, and drying to obtain the final orange-yellow solid ZXC-17.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of ZXC-17 technology offers distinct strategic advantages that extend beyond mere performance metrics. The synthesis route described in the patent eliminates the need for expensive and scarce transition metal catalysts, relying instead on commodity chemicals like sulfuric acid, potassium nitrate, and acetonitrile. This reliance on widely available feedstocks significantly mitigates supply chain risks associated with geopolitical instability or raw material shortages. Furthermore, the simplified purification process, which relies on precipitation and filtration rather than complex chromatography or distillation, drastically reduces the energy consumption and solvent usage per kilogram of product. These factors combine to create a manufacturing profile that supports substantial cost savings in energetic material manufacturing, allowing defense contractors to optimize their budgets without sacrificing the quality or reliability of their munitions. The robustness of the process also implies a lower rate of batch failures, ensuring a more consistent and predictable supply of critical energetic components.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts removes the necessity for expensive heavy metal scavenging and removal steps, which are often regulatory bottlenecks in pharmaceutical and fine chemical production. By utilizing simple inorganic nitrates and sulfuric acid, the variable cost of goods sold is significantly lowered, and the capital expenditure required for specialized reactor linings is minimized. The high yield reported in the patent examples, often exceeding 85%, further amplifies these economic benefits by maximizing the output from each batch of raw materials. Additionally, the ease of recrystallization and purification reduces the labor hours and utility costs associated with downstream processing, contributing to a leaner and more efficient production line.
- Enhanced Supply Chain Reliability: The raw materials required for ZXC-17 synthesis, including trifluoroaniline derivatives and common acylating agents, are produced by a broad network of global chemical suppliers, reducing dependency on single-source vendors. This diversification of the supply base enhances resilience against market fluctuations and logistical disruptions. The stability of the intermediate and final products also allows for more flexible inventory management, as the materials do not require extreme storage conditions or have excessively short shelf lives. For supply chain heads, this translates to reduced lead times for high-purity energetic intermediates and the ability to scale production up or down in response to fluctuating demand without incurring prohibitive costs or delays.
- Scalability and Environmental Compliance: The process is inherently scalable, moving seamlessly from gram-scale laboratory synthesis to multi-ton industrial production without fundamental changes to the reaction chemistry. The use of aqueous workups and the absence of volatile organic solvents in the final isolation steps simplify waste treatment and align with increasingly stringent environmental regulations. The insolubility of the product in water facilitates efficient solid-liquid separation, minimizing the volume of contaminated wastewater generated. This green chemistry profile not only reduces disposal costs but also enhances the corporate social responsibility standing of the manufacturer, making it an attractive partner for government contracts that prioritize sustainable sourcing and production practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of ZXC-17 technology. These insights are derived directly from the experimental data and beneficial effects outlined in the patent literature, providing a factual basis for decision-making. Understanding the specific performance parameters and synthesis constraints is essential for integrating this new energetic material into existing formulation pipelines. We encourage technical teams to review these details closely to assess compatibility with current manufacturing infrastructure.
Q: Why is ZXC-17 considered superior to TNT for fusion-cast applications?
A: ZXC-17 offers a significantly higher single crystal density of 1.972 g/cm³ compared to TNT's 1.64 g/cm³, resulting in superior detonation velocity and pressure. Furthermore, its impact sensitivity of 62J indicates much lower mechanical sensitivity than TNT (29J), enhancing safety during handling and casting.
Q: What is the optimal melting point range for this fusion-cast explosive carrier?
A: The compound exhibits a melting point of 92.20 °C, which falls ideally within the 70-120 °C range required for fusion-cast explosives. This ensures sufficient fluidity for casting without premature thermal decomposition, addressing common issues like shrinkage and cavitation found in traditional carriers.
Q: Are the raw materials for ZXC-17 synthesis readily available for industrial scaling?
A: Yes, the synthesis utilizes commercially accessible reagents including 3,4,5-trifluoroaniline, common acylating agents, and standard nitrating salts like potassium nitrate or ammonium nitrate. The process avoids exotic catalysts, facilitating straightforward supply chain management and cost reduction.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,4,5-Trifluoro-2,6-Dinitroaniline Supplier
As the global landscape for energetic materials evolves, partnering with a CDMO expert who understands the nuances of high-energy synthesis is paramount for success. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory curiosity to deployed capability is seamless. Our facility is equipped with rigorous QC labs and adheres to stringent purity specifications, guaranteeing that every batch of 3,4,5-trifluoro-2,6-dinitroaniline meets the exacting standards required for fusion-cast explosive applications. We recognize that the margin for error in energetic materials is non-existent, which is why our quality assurance protocols are designed to detect and eliminate even trace impurities that could affect sensitivity or stability.
We invite you to engage with our technical procurement team to discuss how ZXC-17 can enhance your current product portfolio. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic benefits of switching to this superior carrier. We are prepared to provide specific COA data and route feasibility assessments tailored to your volume requirements, ensuring that you have all the necessary information to make an informed sourcing decision. Contact us today to secure a reliable supply of next-generation energetic intermediates that deliver unmatched performance and safety.
