Advanced Magnesium Catalysis for Efficient Carboxylic Acid Deoxygenation and Boronate Synthesis
Advanced Magnesium Catalysis for Efficient Carboxylic Acid Deoxygenation and Boronate Synthesis
The landscape of organoboron synthesis is undergoing a significant transformation driven by the urgent need for greener, more sustainable, and cost-effective manufacturing processes. A pivotal advancement in this field is documented in patent CN108948059B, which introduces a novel carboxylic acid deoxyboration reaction method. This technology leverages a specifically designed, large sterically hindered amino ligand-stabilized magnesium alkyl compound to catalyze the reduction of carboxylic acids with boranes. Unlike traditional methods that rely heavily on toxic and expensive transition metals, this innovation utilizes earth-abundant magnesium to achieve high catalytic activity under mild, solvent-free conditions. For R&D directors and procurement specialists in the fine chemical and pharmaceutical sectors, this represents a paradigm shift towards safer, more scalable, and economically viable production of valuable organoborate intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of organoborate compounds from carboxylic acids has been dominated by the use of transition metal catalysts containing elements such as Ruthenium (Ru), Rhodium (Rh), Iridium (Ir), Cobalt (Co), Iron (Fe), and Copper (Cu). While effective, these conventional methodologies present substantial drawbacks for large-scale industrial application. The primary concern is the inherent toxicity associated with heavy metal residues, which necessitates complex and costly purification steps to meet stringent regulatory standards for pharmaceutical intermediates. Furthermore, the reliance on precious metals introduces significant supply chain volatility and cost fluctuations. These traditional processes often require harsh reaction conditions or specific solvents that contribute to environmental burdens through volatile organic compound (VOC) emissions and hazardous waste generation, making them increasingly unsustainable in the context of modern green chemistry mandates.
The Novel Approach
In stark contrast, the methodology outlined in patent CN108948059B offers a robust alternative by employing a main group metal catalyst. This novel approach utilizes a magnesium alkyl compound stabilized by a bulky amino ligand, which facilitates the deoxyboration reaction with exceptional efficiency. The process operates under anhydrous and oxygen-free conditions but critically eliminates the need for reaction solvents, thereby simplifying the workup procedure and reducing waste. The substrate scope is remarkably broad, accommodating a wide variety of carboxylic acids. As illustrated below, the method is applicable to diverse aromatic carboxylic acids where R can be hydrogen, alkyl, alkoxy, halogen, ester, or acid groups.
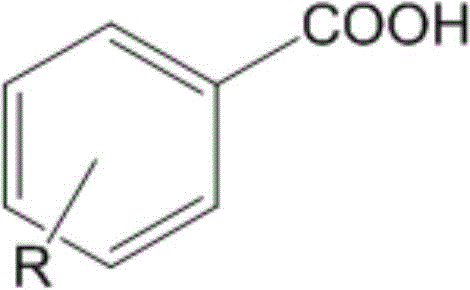
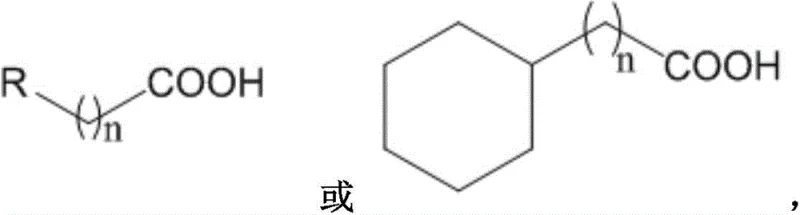
Furthermore, the versatility extends to aliphatic systems, including linear and cyclic fatty acids, as shown in the general structure below where R represents hydrogen, halogen, or carboxyl groups. This universality makes the technology highly attractive for a reliable pharmaceutical intermediate supplier looking to diversify their portfolio of boronic acid building blocks without the baggage of heavy metal contamination.
Mechanistic Insights into Magnesium-Catalyzed Deoxyboration
The core of this technological breakthrough lies in the unique structure of the catalyst itself. The magnesium center is coordinated by a large sterically hindered amino ligand, which plays a dual role in stabilizing the reactive metal-alkyl bond and modulating the electronic environment to facilitate catalysis. The steric bulk prevents the formation of inactive oligomeric species, ensuring that the magnesium remains monomeric and highly reactive towards the borane reagent. This structural integrity is crucial for maintaining high turnover numbers and preventing catalyst deactivation during the reaction cycle. The specific architecture of the catalyst, featuring bulky phenyl and silyl groups around the magnesium center, creates a protected pocket that allows for selective activation of the carboxylic acid functionality while tolerating other sensitive functional groups present on the substrate.
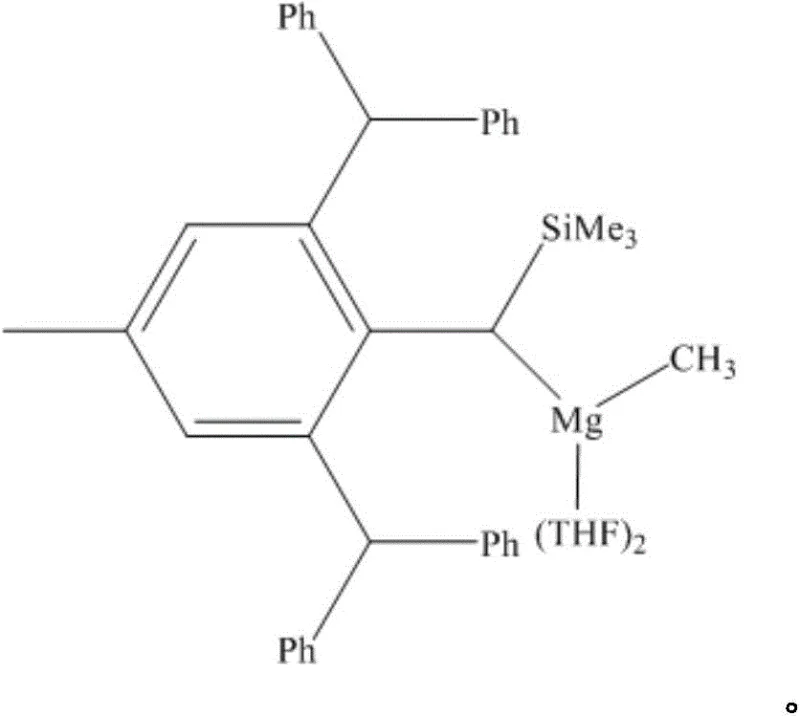
From an impurity control perspective, the mechanism offers distinct advantages over transition metal catalysis. Since magnesium is a main group metal, the resulting byproducts are generally inorganic salts or benign organic magnesium species that are significantly easier to remove via aqueous workup compared to trace transition metals which often require specialized scavengers or chromatography. The reaction proceeds through a concerted pathway where the magnesium hydride or alkyl species likely activates the B-H bond of the pinacolborane, followed by insertion of the carboxylic acid carbonyl. This pathway avoids the formation of radical intermediates that could lead to side reactions, thereby ensuring a clean impurity profile. The ability to achieve near-quantitative conversion with only 0.1 mol% catalyst loading underscores the high efficiency of this mechanistic pathway, making it ideal for the commercial scale-up of complex fine chemical intermediates.
How to Synthesize Organoborate Compounds Efficiently
Implementing this synthesis route requires strict adherence to inert atmosphere techniques to preserve the activity of the magnesium catalyst. The process is designed to be operationally simple, eliminating the need for solvent drying and recovery systems. The standardized protocol involves mixing the carboxylic acid and pinacolborane directly, followed by the introduction of the catalyst under nitrogen protection. Detailed standard operating procedures regarding stoichiometry, temperature control, and quenching methods are essential for reproducibility. For a comprehensive guide on executing this transformation in a pilot or production setting, please refer to the step-by-step synthesis guide below.
- Under anhydrous and oxygen-free conditions, add the carboxylic acid substrate to a reaction vessel equipped with stirring.
- Introduce pinacolborane to the mixture and ensure uniform blending without the use of additional solvents.
- Add the bulky amino ligand-stabilized magnesium alkyl catalyst (0.1 mol%) and maintain the reaction at 60°C for approximately 1 hour.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this magnesium-catalyzed technology translates into tangible strategic benefits beyond mere technical feasibility. The elimination of organic solvents represents a direct reduction in raw material costs and waste disposal fees, contributing to a leaner manufacturing budget. Moreover, the shift away from precious transition metals mitigates the risk associated with the price volatility of commodities like rhodium or ruthenium. The simplicity of the reaction setup, requiring only moderate heating and standard inert gas protection, lowers the barrier for equipment investment and facilitates faster technology transfer from lab to plant. These factors collectively enhance the reliability of the supply chain for high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The economic impact of this process is driven primarily by the drastic simplification of the reaction medium. By operating under solvent-free conditions, manufacturers eliminate the capital and operational expenditures associated with solvent storage, recycling distillation columns, and VOC abatement systems. Additionally, the catalyst loading is exceptionally low at 0.1 mol%, and since magnesium is vastly cheaper than noble metals, the direct material cost per kilogram of product is significantly reduced. The absence of heavy metal residues also removes the need for expensive metal scavenger resins or extensive chromatographic purification, further streamlining the cost structure and improving overall margin potential for bulk chemical production.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of earth-abundant magnesium rather than geographically concentrated precious metals. The raw materials, including pinacolborane and various carboxylic acids, are commodity chemicals with stable global availability. The robustness of the catalyst system, which tolerates a wide range of functional groups without degradation, ensures consistent batch-to-batch quality even with slight variations in feedstock purity. This reliability reduces the risk of production delays caused by catalyst shortages or failed batches due to metal sensitivity, ensuring a steady flow of critical building blocks for downstream drug synthesis and agrochemical manufacturing.
- Scalability and Environmental Compliance: Scaling this reaction is inherently safer and more compliant with environmental regulations. The absence of flammable organic solvents reduces the fire hazard rating of the production facility, potentially lowering insurance premiums and safety infrastructure costs. The "green" nature of the process, characterized by atom economy and lack of toxic metal waste, aligns perfectly with increasingly stringent environmental, social, and governance (ESG) goals of multinational corporations. This compliance advantage facilitates smoother regulatory approvals and enhances the marketability of the final products to eco-conscious clients in the European and North American markets who prioritize sustainable sourcing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this magnesium-catalyzed deoxyboration technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a factual basis for decision-making. Understanding these nuances is critical for R&D teams evaluating process compatibility and for procurement officers assessing vendor capabilities. The answers reflect the specific advantages of using bulky amino ligand-stabilized magnesium catalysts over traditional methods.
Q: What are the advantages of using magnesium catalysts over transition metals?
A: Magnesium catalysts offer significant advantages including non-toxicity, lower cost compared to precious metals like Ru or Rh, and easier removal of metal residues from the final product, which is critical for pharmaceutical applications.
Q: Does this method require organic solvents?
A: No, the patented process operates under solvent-free conditions, which drastically reduces waste generation and simplifies the downstream purification process, aligning with green chemistry principles.
Q: What types of carboxylic acids are compatible with this reaction?
A: The method demonstrates broad substrate scope, successfully converting both aromatic carboxylic acids (such as benzoic acid derivatives) and aliphatic carboxylic acids (including fatty acids and cyclic acids) into their corresponding boronic esters.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Organoborate Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of main group metal catalysis in modernizing chemical synthesis. Our technical team has extensively analyzed the feasibility of scaling the magnesium-catalyzed deoxyboration pathway described in CN108948059B. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. Our facilities are equipped with rigorous QC labs capable of detecting trace impurities, guaranteeing that our products meet stringent purity specifications required by top-tier pharmaceutical clients. We are committed to delivering high-quality organoborate intermediates that leverage this advanced, green chemistry technology.
We invite you to collaborate with us to optimize your supply chain for boronic acid derivatives. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and detailed route feasibility assessments to demonstrate how our adoption of this solvent-free, magnesium-catalyzed process can drive value and sustainability in your manufacturing operations.
