Advanced Copper-Catalyzed Synthesis of Polychlorinated Alkynes for Industrial Scale-Up
The landscape of organic synthesis for functionalized alkynes is undergoing a significant transformation, driven by the urgent need for greener, more efficient manufacturing processes in the pharmaceutical and agrochemical sectors. Patent CN114956952A introduces a groundbreaking methodology for the synthesis of copper-catalyzed polychlorinated alkyne compounds, addressing critical bottlenecks in current production capabilities. This innovative protocol utilizes a redox-neutral, multi-component reaction strategy that couples polyhalogenated alkane reagents with aromatic olefins and aromatic alkynes. By leveraging a cost-effective copper salt catalyst and a specialized terpyridine ligand system, this technology enables the construction of complex molecular architectures under exceptionally mild conditions. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediate supplier, this patent represents a pivotal shift away from energy-intensive processes toward sustainable, room-temperature catalysis that maintains high purity standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of polychlorinated compounds has been fraught with significant operational challenges that hinder large-scale commercial viability. Traditional strategies predominantly rely on the intramolecular cyclization of polychlorinated alkane reagents with functionalized alkenes or intermolecular 1,2-addition reactions. These legacy methods invariably demand the use of stoichiometric amounts of strong oxidants, which not only increases raw material costs but also generates substantial hazardous waste streams that require complex disposal protocols. Furthermore, these reactions typically necessitate harsh thermal conditions, often requiring heating up to 100°C to overcome high activation energy barriers. Such elevated temperatures pose safety risks in industrial settings and can lead to the decomposition of thermally sensitive functional groups, thereby limiting the substrate scope and resulting in lower overall yields and difficult purification profiles for high-purity pharmaceutical intermediates.
The Novel Approach
In stark contrast, the methodology disclosed in CN114956952A offers a streamlined, redox-neutral pathway that eliminates the need for external oxidants and excessive heating. The process employs a catalytic system comprising a copper salt, specifically Copper(II) triflate, coordinated with an alpha,alpha,alpha-terpyridine ligand. This catalyst system facilitates a three-component coupling reaction in a mixed solvent of methanol and acetonitrile at room temperature (20-30°C). The elimination of stoichiometric oxidants and the reduction of reaction temperature to ambient levels drastically simplify the engineering controls required for production. This approach not only enhances safety but also improves the economic feasibility of producing complex polychlorinated alkynes, making it an ideal candidate for cost reduction in pharmaceutical intermediates manufacturing while ensuring robust functional group compatibility.
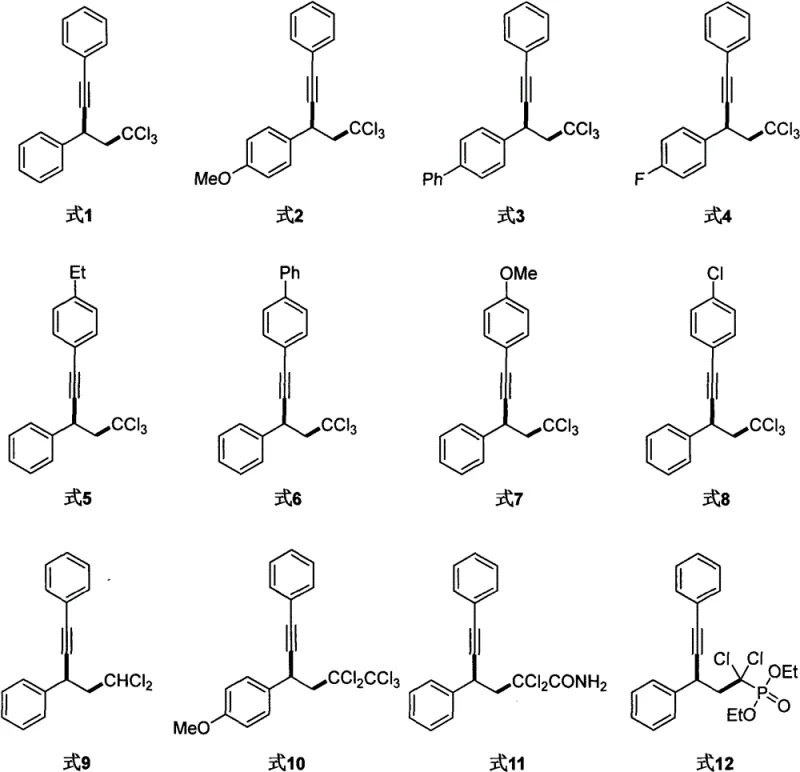
The versatility of this synthetic route is further evidenced by its broad substrate scope, accommodating a wide array of electronic and steric environments. As demonstrated in the experimental data, the reaction tolerates various substituents on the aromatic rings, including electron-donating groups like methoxy and ethyl, as well as electron-withdrawing groups such as fluoro and chloro. This flexibility is crucial for medicinal chemists who require diverse building blocks for drug discovery. The ability to synthesize these structures without protecting group manipulation or harsh conditions underscores the method's potential for accelerating the development of new active pharmaceutical ingredients (APIs) and advanced materials.
Mechanistic Insights into Copper-Catalyzed Radical Coupling
The mechanistic foundation of this transformation lies in a copper-mediated radical pathway that proceeds under redox-neutral conditions. The copper catalyst, stabilized by the tridentate terpyridine ligand, likely facilitates the generation of radical species from the polyhalogenated alkane reagent through a single-electron transfer (SET) process. This radical intermediate then adds across the double bond of the aromatic olefin, followed by interaction with the aromatic alkyne to form the final polychlorinated alkyne product. The choice of the terpyridine ligand is critical, as it modulates the redox potential of the copper center, ensuring efficient turnover while preventing catalyst deactivation. Understanding this mechanism allows process chemists to fine-tune reaction parameters, such as the molar ratios of reactants (optimized at 1:1.5:1.5 for olefin:alkyne:reagent), to maximize conversion rates and minimize byproduct formation.
From an impurity control perspective, the mild nature of this radical mechanism offers distinct advantages over ionic pathways that might suffer from rearrangement or elimination side reactions. The room-temperature operation prevents thermal degradation of the polychlorinated moieties, which are often susceptible to dehydrohalogenation under basic or heated conditions. Consequently, the crude reaction mixtures obtained via this method exhibit cleaner profiles, reducing the burden on downstream purification units. This results in higher isolated yields, as seen in examples where yields reach up to 90% for specific substrates, ensuring that the final high-purity OLED material or pharmaceutical precursor meets stringent quality specifications with minimal processing steps.
How to Synthesize Polychlorinated Alkyne Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires strict adherence to the optimized protocol to ensure reproducibility and safety. The process involves the sequential addition of reagents under an inert nitrogen atmosphere to prevent oxidation of the catalyst or radical intermediates. The specific ratio of methanol to acetonitrile (1:1) is vital for solubilizing both the organic substrates and the inorganic base, potassium carbonate, creating a homogeneous reaction environment that promotes efficient mass transfer. While the general procedure is robust, attention to detail regarding the order of addition and stirring efficiency is paramount for achieving the reported yields.
- Prepare the reaction vessel under inert atmosphere and add copper catalyst, terpyridine ligand, aromatic olefin, aromatic alkyne, and polychlorinated alkane reagent.
- Introduce the base (potassium carbonate) and the mixed solvent system of methanol and acetonitrile to initiate the reaction at room temperature.
- Stir the mixture for 24 hours, filter through celite, concentrate the organic phase, and purify the crude product via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this copper-catalyzed technology translates into tangible strategic benefits that extend beyond simple yield improvements. The shift from stoichiometric oxidants to a catalytic cycle fundamentally alters the cost structure of production by reducing the volume of raw materials required per kilogram of product. Additionally, the use of commodity chemicals like styrene derivatives and phenylacetylene ensures a stable and resilient supply chain, mitigating the risks associated with sourcing exotic or regulated reagents. This stability is essential for maintaining continuous production schedules and meeting the demanding delivery timelines of global pharmaceutical clients.
- Cost Reduction in Manufacturing: The elimination of expensive stoichiometric oxidants and the ability to run reactions at room temperature significantly lowers the operational expenditure (OPEX) associated with energy consumption and waste treatment. By removing the need for heating mantles or oil baths to reach 100°C, facilities can reduce their carbon footprint and utility costs. Furthermore, the simplified workup procedure, which involves basic filtration and evaporation rather than complex quenching of oxidants, reduces labor hours and solvent usage, leading to substantial cost savings in the overall manufacturing process.
- Enhanced Supply Chain Reliability: The reliance on readily available starting materials such as substituted styrenes and terminal alkynes ensures that production is not bottlenecked by the availability of specialized reagents. The robustness of the catalyst system, which tolerates moisture and air to a reasonable extent during setup, further enhances process reliability. This resilience allows for flexible scheduling and rapid scale-up capabilities, ensuring that customers receive their orders of commercial scale-up of complex polymer additives or intermediates without unexpected delays caused by raw material shortages or process failures.
- Scalability and Environmental Compliance: Operating at ambient temperature inherently improves the safety profile of the reaction, making it easier to scale from gram to ton quantities without encountering exothermic runaway risks. The redox-neutral nature of the reaction minimizes the generation of heavy metal waste and halogenated byproducts, aligning with increasingly strict environmental regulations. This compliance reduces the regulatory burden on manufacturing sites and facilitates smoother audits, positioning the production facility as a preferred partner for environmentally conscious multinational corporations seeking sustainable supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this copper-catalyzed synthesis. These insights are derived directly from the experimental data and comparative studies presented in the patent documentation, providing clarity on catalyst selection, solvent effects, and substrate compatibility. Understanding these nuances is critical for process engineers aiming to integrate this technology into existing production lines.
Q: What are the primary advantages of this copper-catalyzed method over traditional synthesis?
A: Unlike conventional methods that require stoichiometric oxidants and high temperatures (100°C), this novel approach operates at room temperature under redox-neutral conditions, significantly reducing energy consumption and waste generation.
Q: Is this synthesis method compatible with sensitive functional groups?
A: Yes, the mild reaction conditions demonstrate excellent functional group tolerance, successfully accommodating substituents such as methoxy, fluoro, chloro, and phenyl groups without degradation.
Q: What represents the optimal catalyst and ligand system for this transformation?
A: Experimental data indicates that Copper(II) triflate (Cu(OTf)2) combined with an alpha,alpha,alpha-terpyridine ligand provides the highest efficiency and yield for generating polychlorinated alkyne compounds.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polychlorinated Alkyne Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this copper-catalyzed methodology for the next generation of fine chemical synthesis. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab bench to market is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of polychlorinated alkyne intermediate meets the exacting standards required for pharmaceutical and electronic applications.
We invite you to collaborate with our technical team to explore how this innovative route can optimize your specific supply chain requirements. Please contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your project needs. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our expertise can drive value and efficiency in your manufacturing operations.
