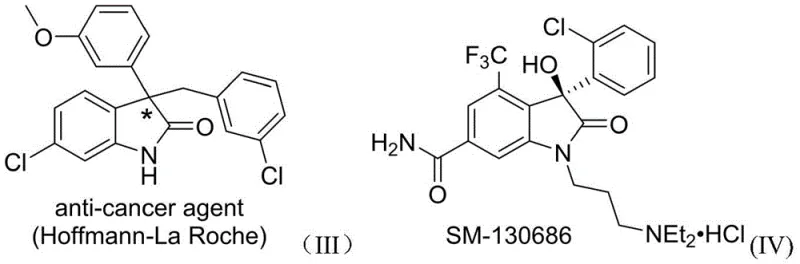
Revolutionizing Complex Heterocycle Production: One-Step Synthesis of 3-Aryl-3'-Amino Bis-Oxoindoles for Pharma Applications
The pharmaceutical industry is constantly seeking efficient routes to access complex heterocyclic scaffolds that serve as the core structures for next-generation therapeutics. A groundbreaking development in this field is detailed in Chinese Patent CN109776384B, which discloses a novel synthesis method for 3-aryl-3'-amino bis-quaternary carbon bis-oxoindole compounds. This specific structural framework is of immense interest because it combines two biologically active motifs: the 3-aryloxindole structure found in anticancer agents and the 3-aminooxindole structure present in gastrin receptor antagonists. The patent highlights a transformative approach that utilizes a rhodium-catalyzed multicomponent reaction to construct these sterically crowded molecules in a single operational step. By leveraging diazo compounds, N,N-disubstituted anilines, and isatin imine compounds as raw materials, this method overcomes the historical synthetic difficulties associated with forming continuous quaternary carbon centers. For R&D directors and procurement managers alike, this represents a significant leap forward in accessing high-value pharmaceutical intermediates with superior efficiency and reduced environmental impact compared to legacy methodologies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the 3,3'-disubstituted oxindole skeleton has been a formidable challenge in organic synthesis due to severe steric hindrance and the presence of two contiguous quaternary carbon centers. Traditional synthetic strategies often involve linear sequences exceeding five distinct steps, leading to a cumulative loss of material at each stage. Consequently, the total reaction yield for these conventional routes frequently falls below 30%, rendering them economically unviable for large-scale manufacturing. Furthermore, these multi-step processes generate substantial amounts of chemical waste and require prolonged process times, which complicates supply chain logistics and increases the overall carbon footprint of the manufacturing operation. The structural complexity, characterized by highly functionalized molecules and all-carbon quaternary centers, often necessitates harsh reaction conditions or expensive protecting group strategies that further erode process efficiency and increase the cost of goods sold for the final active pharmaceutical ingredient.
The Novel Approach
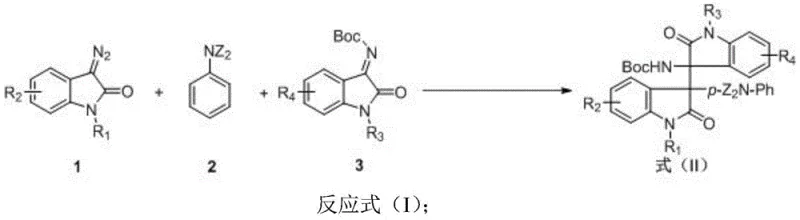
In stark contrast to these cumbersome traditional routes, the method disclosed in Patent CN109776384B offers a streamlined, one-step solution that dramatically simplifies the production landscape. This novel approach employs a rhodium acetate catalyst to facilitate a three-component coupling reaction at room temperature, eliminating the need for energy-intensive heating or cooling cycles. The reaction exhibits exceptional atom economy, as the only byproduct released is nitrogen gas, meaning virtually all atoms from the starting materials are incorporated into the final product structure. With reported isolated yields reaching as high as 99% for specific derivatives and excellent diastereoselectivity (dr > 20:1), this method provides a robust platform for generating diverse libraries of bis-oxoindole compounds. This shift from a multi-step linear synthesis to a convergent one-step process fundamentally alters the economic model of producing these complex intermediates, offering substantial potential for cost reduction in API manufacturing.
Mechanistic Insights into Rhodium-Catalyzed Multicomponent Coupling
The core of this technological breakthrough lies in the sophisticated use of rhodium carbenoid chemistry to trap ion-pair intermediates. The reaction initiates with the decomposition of the diazo compound by the dirhodium tetraacetate catalyst (Rh2(OAc)4), generating a highly reactive metal-carbene species. This electrophilic intermediate is then intercepted by the nucleophilic N,N-disubstituted aniline, forming a transient ammonium ylide or similar ion-pair intermediate. Crucially, the presence of the isatin imine compound allows for an immediate trapping of this reactive species, facilitating the formation of the new carbon-carbon and carbon-nitrogen bonds that define the bis-quaternary center. The use of 4A molecular sieves as an additive plays a critical role in maintaining anhydrous conditions, which prevents the hydrolysis of the sensitive diazo or imine components and ensures the catalyst remains active throughout the transformation. This mechanistic pathway allows for the precise construction of the sterically demanding 3-aryl-3'-amino architecture with high regioselectivity, avoiding the formation of unwanted regioisomers that often plague similar cyclization reactions.
From an impurity control perspective, the high selectivity of this catalytic system is a major advantage for pharmaceutical manufacturing. The reaction conditions are mild enough to prevent the decomposition of sensitive functional groups often present on the aromatic rings, such as halogens or alkoxy groups, which are common in drug candidates. The patent data indicates that the diastereoselectivity is consistently greater than 20:1, which significantly reduces the burden on downstream purification processes. In traditional syntheses, separating diastereomers often requires costly preparative HPLC or multiple recrystallizations, but the inherent selectivity of this rhodium-catalyzed route minimizes these challenges. This level of control over the stereochemical outcome ensures that the resulting high-purity oxindole derivatives meet the stringent quality specifications required for clinical trial materials and eventual commercial drug production, thereby reducing the risk of batch failures.
How to Synthesize 3-Aryl-3'-Amino Bis-Oxoindole Efficiently
The practical implementation of this synthesis is remarkably straightforward, designed to be accessible for both laboratory-scale discovery and pilot-plant operations. The protocol involves dissolving the isatin imine, the aniline derivative, and the rhodium catalyst in a common organic solvent like dichloromethane, followed by the controlled addition of the diazo component. The use of a peristaltic pump for the slow addition of the diazo compound is a key operational detail that helps manage the exotherm and control the concentration of the reactive carbene species, ensuring safety and reproducibility. Following the reaction, the workup is simplified to standard column chromatography, avoiding complex extraction or distillation procedures. For a detailed, step-by-step guide on executing this reaction with specific molar ratios and purification parameters, please refer to the standardized synthesis instructions provided below.
- Dissolve isatin imine compound, rhodium acetate catalyst (Rh2(OAc)4), N,N-disubstituted aniline, and 4A molecular sieves in an organic solvent such as dichloromethane.
- Slowly add the diazo compound solution to the reaction mixture at room temperature (25°C) over a period of one hour using a peristaltic pump.
- Stir the reaction mixture for two hours after addition is complete, then purify the crude product via column chromatography using ethyl acetate and petroleum ether.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers compelling strategic advantages that extend beyond simple yield improvements. The transition from a lengthy multi-step sequence to a single-step catalytic process fundamentally de-risks the supply chain by reducing the number of unit operations and intermediate handling stages. Fewer steps mean fewer opportunities for human error, equipment failure, or quality deviations, leading to a more reliable supply of critical pharmaceutical intermediates. Additionally, the use of readily available starting materials such as substituted anilines and isatin derivatives ensures that the raw material supply base is robust and less susceptible to geopolitical or logistical disruptions. This stability is crucial for maintaining continuous production schedules for downstream API manufacturing.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the drastic reduction in processing time and material consumption. By eliminating the need for multiple isolation and purification steps associated with traditional five-step syntheses, manufacturers can significantly lower labor costs and solvent usage. The high atom economy ensures that expensive starting materials are converted efficiently into the desired product rather than waste, directly improving the margin profile of the intermediate. Furthermore, the low catalyst loading and the ability to operate at room temperature reduce energy consumption and the cost associated with specialized reactor equipment, contributing to substantial overall cost savings without compromising quality.
- Enhanced Supply Chain Reliability: The simplicity of the one-pot reaction design enhances supply chain resilience by shortening the lead time required to produce batches of the intermediate. With a reaction time of only a few hours and a simple workup procedure, facilities can achieve faster turnaround times, allowing for more responsive inventory management. The robustness of the reaction across a wide range of substrates, including those with electron-withdrawing or electron-donating groups, means that a single manufacturing line can be adapted to produce various analogues needed for structure-activity relationship (SAR) studies or different drug candidates, providing flexibility to the supply chain.
- Scalability and Environmental Compliance: From an environmental and regulatory standpoint, this method aligns well with green chemistry principles. The generation of nitrogen gas as the sole byproduct minimizes the volume of hazardous waste requiring treatment and disposal. The avoidance of harsh reagents and extreme conditions simplifies the safety profile of the manufacturing process, reducing the regulatory burden related to process safety management. As the industry moves towards more sustainable practices, adopting a high-efficiency catalytic process like this positions the supply chain to meet increasingly stringent environmental compliance standards while maintaining high production volumes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the feasibility and advantages of the method for potential partners and licensees. Understanding these details is essential for evaluating the integration of this technology into existing manufacturing workflows.
Q: What is the primary advantage of this Rh-catalyzed method over traditional synthesis?
A: The primary advantage is step economy and yield. Traditional methods often require more than five steps with total yields below 30%, whereas this patented one-step process achieves yields up to 99% with excellent diastereoselectivity (dr > 20:1).
Q: What catalyst loading is required for this transformation?
A: The process utilizes a low loading of rhodium acetate (Rh2(OAc)4), typically around 0.05 mmol relative to 0.3 mmol of substrate (approx. 16 mol%), which helps minimize heavy metal residue and cost.
Q: Is this method scalable for industrial production?
A: Yes, the reaction operates under mild conditions (room temperature) and uses common solvents like dichloromethane, making it highly suitable for commercial scale-up without requiring extreme pressure or cryogenic temperatures.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Aryl-3'-Amino Bis-Oxoindole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the 3-aryl-3'-amino bis-quaternary carbon bis-oxoindole scaffold in modern drug discovery, particularly for oncology and metabolic disorders. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to clinical supply is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying the stringent purity specifications required for these complex heterocycles, ensuring that every batch meets the highest international standards for pharmaceutical intermediates.
We invite you to collaborate with us to leverage this advanced rhodium-catalyzed technology for your specific project needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your target molecule, demonstrating how this efficient route can optimize your budget. Please contact our technical procurement team today to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your proprietary candidates, and let us help you accelerate your path to market.
