Advanced Catalytic Hydrogenation Strategy for Manufacturing High-Purity 3,5-Dichloro-2,4-Difluoroaniline
Advanced Catalytic Hydrogenation Strategy for Manufacturing High-Purity 3,5-Dichloro-2,4-Difluoroaniline
The global demand for high-performance benzoylurea insecticides continues to drive innovation in the synthesis of their critical precursors. Specifically, the production of 3,5-dichloro-2,4-difluoroaniline serves as a pivotal bottleneck in the supply chain for Teflubenzuron, a potent chitin synthesis inhibitor widely used in modern crop protection. Recent intellectual property developments, notably patent CN109369414B, have introduced a transformative approach to synthesizing this key intermediate. This patent discloses a refined catalytic hydrogenation process that utilizes a platinum on carbon (Pt/C) catalyst in conjunction with a specific dehalogenation inhibitor. By addressing the longstanding challenges of selectivity and waste generation, this technology offers a robust pathway for reliable agrochemical intermediate suppliers seeking to enhance their manufacturing capabilities. The method not only ensures exceptional product purity but also aligns with increasingly stringent environmental regulations regarding industrial effluent and solid waste management.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of 3,5-dichloro-2,4-difluoroaniline has relied heavily on reduction methods utilizing iron powder under acidic conditions. While these traditional routes are chemically feasible, they suffer from profound operational and environmental drawbacks that hinder modern scalable production. The process typically involves heating the nitro precursor with iron powder in a mixed solvent system, followed by neutralization and steam distillation. This generates copious amounts of iron sludge, a hazardous solid waste that is difficult and costly to treat. Furthermore, the separation of the product often requires organic solvent extraction or energy-intensive steam distillation, which can lead to product loss through tar formation and significant thermal degradation. The resulting crude product frequently exhibits poor color and requires extensive purification steps, such as activated carbon decolorization and recrystallization, further eroding overall yield and increasing the carbon footprint of the manufacturing process.
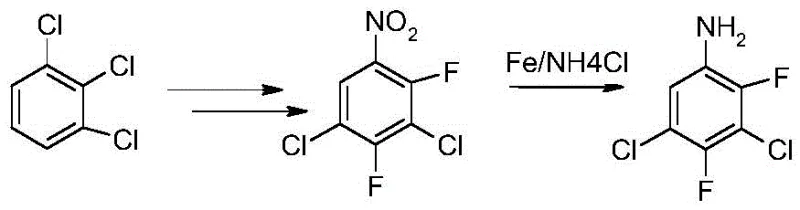
The Novel Approach
In stark contrast to the archaic iron powder reduction, the methodology outlined in patent CN109369414B leverages advanced catalytic hydrogenation to achieve a cleaner and more efficient transformation. The core innovation lies in the direct reduction of 3,5-dichloro-2,4-difluoronitrobenzene using molecular hydrogen in the presence of a 3% Pt/C catalyst. Crucially, the process incorporates a dehalogenation inhibitor, specifically phosphoric acid, which plays a vital role in preserving the integrity of the halogenated aromatic ring. This strategic addition prevents the unwanted removal of chlorine and fluorine atoms, a common side reaction in catalytic hydrogenation of polyhalogenated nitro compounds. The reaction proceeds under mild conditions, typically between 75-80°C and moderate hydrogen pressures of 1.0-1.5 MPa. The outcome is a high-purity product obtained as a white to off-white solid, eliminating the need for complex downstream purification and significantly reducing the generation of three wastes (wastewater, waste gas, and solid waste).
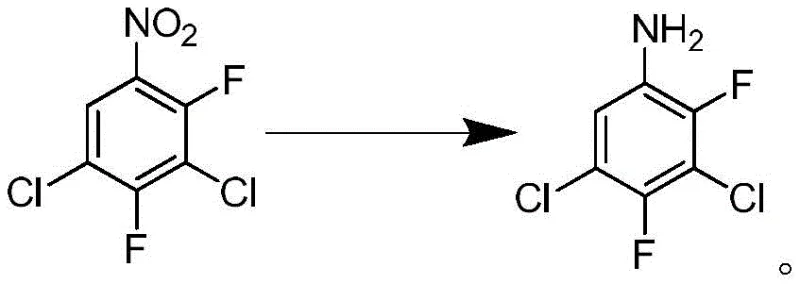
Mechanistic Insights into Pt/C-Catalyzed Hydrogenation with Dehalogenation Inhibition
The success of this synthetic route hinges on the delicate balance between reducing the nitro group and preserving the carbon-halogen bonds. In standard catalytic hydrogenation, noble metal catalysts like palladium or platinum are highly active and can inadvertently catalyze hydrodehalogenation, where hydrogen atoms replace halogen substituents on the aromatic ring. This leads to a complex mixture of dechlorinated and defluorinated byproducts, severely compromising the purity of the final aniline derivative. The introduction of phosphoric acid as an inhibitor modifies the electronic environment of the catalyst surface or protonates intermediate species in a way that disfavors the cleavage of C-Cl and C-F bonds. By carefully controlling the molar ratio of the inhibitor to the substrate (optimized between 1:0.05 to 0.1), the process achieves remarkable selectivity. The acid effectively passivates the sites on the platinum surface responsible for dehalogenation while leaving the sites active for nitro group reduction fully functional. This mechanistic nuance allows for the conversion of the nitro group to an amine with minimal structural degradation of the sensitive fluorinated and chlorinated scaffold.
Furthermore, the choice of solvent and reaction parameters contributes significantly to the control of the impurity profile. The use of lower alcohols such as methanol or ethanol provides a polar medium that facilitates hydrogen solubility and mass transfer without promoting side reactions. The moderate temperature range of 75-80°C is sufficient to drive the kinetics of the reduction forward without providing enough thermal energy to overcome the activation barrier for dehalogenation, especially in the presence of the inhibitor. Post-reaction workup is streamlined; the catalyst is simply filtered off and can be recycled, while the product crystallizes out upon the addition of water to the concentrated filtrate. This simplicity in isolation is a direct result of the high selectivity achieved during the reaction phase, demonstrating how mechanistic control translates directly into process efficiency and product quality.
How to Synthesize 3,5-Dichloro-2,4-Difluoroaniline Efficiently
Implementing this advanced hydrogenation protocol requires precise adherence to the optimized reaction conditions to maximize yield and purity. The process begins with the careful charging of the autoclave with the nitro precursor, the alcohol solvent, the platinum catalyst, and the phosphoric acid inhibitor. Maintaining the correct stoichiometry is essential, particularly the ratio of the inhibitor, to ensure complete suppression of dehalogenation byproducts. Once the system is pressurized with hydrogen, the temperature must be strictly controlled within the specified window to balance reaction rate and selectivity. Monitoring the reaction progress via HPLC is recommended to determine the exact endpoint, ensuring complete consumption of the starting material without over-reaction. Following the reaction, the recovery of the catalyst and the crystallization steps are critical for obtaining the final product in its characteristic white powdery form. For detailed operational parameters and safety guidelines, please refer to the standardized synthesis steps provided below.
- Charge the autoclave with 3,5-dichloro-2,4-difluoronitrobenzene, alcohol solvent, 3% Pt/C catalyst, and phosphoric acid inhibitor.
- Pressurize with hydrogen to 1.0-1.5 MPa and maintain temperature at 75-80°C until reaction completion monitored by HPLC.
- Filter catalyst, recover solvent, add water for crystallization, and dry to obtain white powdery 3,5-dichloro-2,4-difluoroaniline.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition from traditional iron powder reduction to this catalytic hydrogenation method represents a significant opportunity for cost reduction in agrochemical intermediates manufacturing. The elimination of iron sludge removes a major cost center associated with hazardous waste disposal and environmental compliance. Traditional methods generate tons of solid waste per ton of product, requiring expensive treatment protocols. By switching to a clean catalytic process, facilities can drastically reduce their environmental liability and operational overhead. Additionally, the superior quality of the crude product means that fewer resources are spent on purification steps like decolorization and recrystallization. This streamlining of the production flow not only lowers variable costs but also shortens the overall cycle time, allowing for faster turnaround and improved responsiveness to market demand fluctuations.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the removal of the iron powder reduction step, which is inherently wasteful. By utilizing a heterogeneous catalyst that can be recovered and reused, the consumption of consumable reagents is minimized. The high selectivity of the reaction reduces the formation of byproducts, which in turn increases the effective yield of the desired intermediate. Higher yields mean that less raw material is required to produce the same amount of finished goods, directly impacting the cost of goods sold (COGS). Furthermore, the simplified workup procedure reduces energy consumption associated with steam distillation and solvent recovery, contributing to substantial cost savings in utility expenses.
- Enhanced Supply Chain Reliability: Adopting a cleaner and more robust synthesis route enhances the reliability of the supply chain for high-purity agrochemical intermediates. The traditional iron powder method is prone to batch-to-batch variability due to the difficulty in controlling the exothermic reduction and the subsequent separation processes. In contrast, the catalytic hydrogenation process is highly controllable and reproducible, ensuring consistent product quality. This consistency reduces the risk of batch rejection and supply disruptions. Moreover, the reduced environmental footprint makes the manufacturing process more sustainable and less susceptible to regulatory shutdowns or restrictions, thereby securing long-term supply continuity for downstream customers relying on this critical insecticide intermediate.
- Scalability and Environmental Compliance: The commercial scale-up of complex fluorinated intermediates is often hindered by waste management challenges. This novel process addresses scalability by minimizing the generation of hazardous byproducts. The absence of iron sludge and the reduction in organic solvent usage make the process easier to scale from pilot plant to multi-ton production without encountering prohibitive waste treatment bottlenecks. Compliance with environmental regulations is significantly easier to achieve, as the effluent load is much lower compared to conventional methods. This environmental advantage not only protects the manufacturer from potential fines but also aligns with the sustainability goals of major global agrochemical companies, making the supplier a more attractive partner for long-term contracts.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of 3,5-dichloro-2,4-difluoroaniline. These insights are derived from the technical specifications and experimental data presented in the relevant patent literature. Understanding these details is crucial for R&D teams evaluating process feasibility and procurement teams assessing supplier capabilities. The answers highlight the specific advantages of the catalytic method over legacy technologies, focusing on purity, yield, and environmental impact.
Q: Why is phosphoric acid added during the hydrogenation of 3,5-dichloro-2,4-difluoronitrobenzene?
A: Phosphoric acid acts as a critical dehalogenation inhibitor. Without it, the hydrogenation process tends to strip chlorine and fluorine atoms from the aromatic ring, leading to significant impurities and reduced yield. The acid suppresses this side reaction, ensuring high selectivity for the nitro group reduction.
Q: What are the advantages of using Pt/C over traditional iron powder reduction?
A: Traditional iron powder reduction generates massive amounts of iron sludge and wastewater, creating severe environmental burdens and high disposal costs. The Pt/C catalytic method is a clean technology that eliminates solid waste sludge, simplifies downstream purification, and produces a product with superior color and purity suitable for direct industrial use.
Q: What is the typical purity and yield achievable with this novel process?
A: According to the patent data, optimizing the catalyst and inhibitor ratio allows for product purity exceeding 97% and yields around 96%. This represents a significant improvement over conventional methods which often struggle with dehalogenation byproducts and lower assay values.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,5-Dichloro-2,4-Difluoroaniline Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the efficacy and safety of final agrochemical products. Our technical team has extensively analyzed advanced synthesis routes, including the catalytic hydrogenation methods described in recent patents, to ensure our manufacturing processes meet the highest industry standards. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet both pilot-scale development needs and large-volume commercial demands. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of 3,5-dichloro-2,4-difluoroaniline delivered meets the exacting requirements of global pesticide manufacturers.
We invite potential partners to engage with our technical procurement team to discuss how our optimized manufacturing capabilities can support your supply chain goals. By leveraging our expertise in clean synthesis technologies, we can offer a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us to request specific COA data and route feasibility assessments, allowing you to verify the superior quality and reliability of our products before making any commitment. Let us be your trusted partner in delivering high-performance agrochemical solutions to the global market.
