Advanced Manufacturing of Scabellone A: Technical Breakthroughs and Commercial Scalability
Advanced Manufacturing of Scabellone A: Technical Breakthroughs and Commercial Scalability
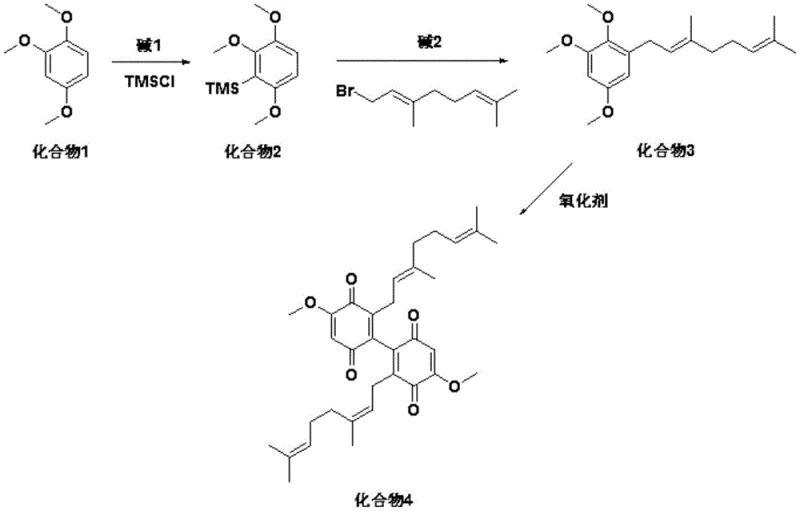
The pharmaceutical industry continuously seeks efficient pathways to access complex natural products with potent biological activities, and Scabellone A stands out as a prime candidate due to its significant anti-inflammatory and antimalarial properties. Derived from marine ascidians of the genus Aplidium, this meroterpenoid has garnered attention for its ability to inhibit the respiratory burst of human neutrophils. However, reliance on natural extraction is unsustainable for commercial drug development, necessitating robust synthetic methodologies. The patent CN107032974B introduces a groundbreaking preparation method for 2-methoxy-6-geranyl-p-benzoquinone derivatives and the subsequent synthesis of Scabellone A. This technical disclosure represents a pivotal shift from laborious total synthesis towards a more streamlined, industrially viable process. By leveraging directed ortho-metalation strategies and controlled oxidative coupling, the inventors have addressed the critical bottlenecks of low yield and operational complexity that have historically plagued the production of this high-value pharmaceutical intermediate.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the synthesis of Scabellone A was characterized by significant inefficiencies that rendered it impractical for large-scale supply. The existing literature, specifically the work by Chan et al., described a route initiating from 2,4-dimethoxybenzaldehyde which involved a Baeyer-Villiger oxidation to generate a phenol, followed by geranylation and subsequent oxidation-reduction sequences to obtain the necessary hydroquinone monomer. This traditional approach was not only chemically tedious, requiring numerous protection and deprotection steps, but it also suffered from abysmal overall yields. Reports indicate that the total yield for Scabellone A using these conventional bio-inspired dimerization methods was less than 2%. Furthermore, these methods often failed to produce related analogues like Scabellone C, limiting the structural diversity available for structure-activity relationship (SAR) studies. The reliance on such low-yielding pathways creates a severe bottleneck for procurement teams, driving up the cost of goods and introducing significant supply chain volatility for any potential API development programs targeting this scaffold.
The Novel Approach
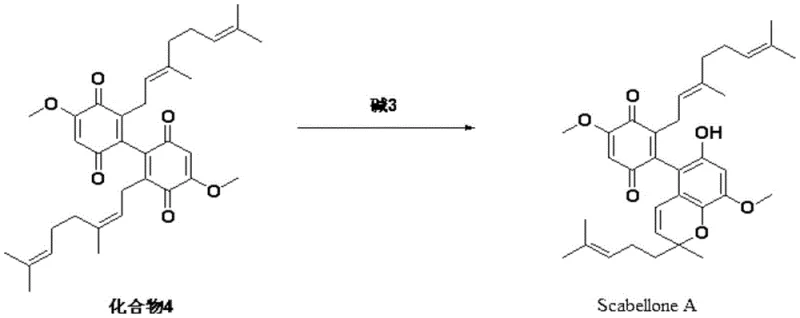
In stark contrast, the methodology disclosed in patent CN107032974B offers a rational redesign of the synthetic tree, focusing on the efficient construction of the dimeric quinone core. The inventors recognized that pre-forming a specific monomeric diquinone intermediate could bypass the stochastic nature of previous dimerization attempts. The new route begins with the readily available 1,2,4-trimethoxybenzene, utilizing a regioselective lithiation-silylation sequence to direct the installation of the geranyl chain precisely at the desired position. This strategic use of a trimethylsilyl blocking group ensures high regioselectivity during the subsequent alkylation step. Following the installation of the terpene tail, the process employs a one-step oxidative coupling using ceric ammonium nitrate (CAN) under mild conditions to directly generate the dimeric quinone derivative. This intermediate is then subjected to a base-mediated cyclization to furnish Scabellone A. This approach drastically reduces the step count and eliminates the need for the inefficient hydroquinone redox cycling found in prior art, thereby laying a solid foundation for the industrial production of this compound.
Mechanistic Insights into CAN-Mediated Oxidative Coupling and Cyclization
The core chemical innovation lies in the oxidative dimerization step, which transforms the geranyl-substituted benzene derivative into the bis-quinone scaffold. This transformation is mediated by ceric ammonium nitrate (CAN), a single-electron oxidant known for its ability to generate phenoxy radicals from electron-rich aromatic systems. In this specific protocol, the reaction is conducted in acetonitrile at controlled low temperatures ranging from 0°C to -40°C. The low temperature is critical to manage the exothermic nature of the oxidation and to prevent the degradation of the sensitive geranyl side chains, which are prone to acid-catalyzed rearrangement or polymerization. The stoichiometry is carefully balanced, with a molar ratio of substrate to oxidant maintained between 1:4 and 1:5, ensuring sufficient oxidative power to drive the dimerization without excessive over-oxidation that could lead to tar formation. The resulting dimeric quinone possesses the necessary electronic configuration to undergo the final ring-closing reaction.
The final cyclization to form Scabellone A is a base-promoted intramolecular oxa-Michael addition or similar nucleophilic attack, depending on the specific tautomeric state of the quinone. The patent highlights the use of pyridine or alpha-picoline as both solvent and base, or as an additive in dichloromethane. The nitrogen lone pair of the pyridine base activates the nucleophilic oxygen or facilitates the deprotonation required for the ring closure. The reaction proceeds at room temperature over a period of 5 to 7 hours, allowing for the thermodynamic equilibration towards the stable tricyclic core of Scabellone A. The choice of base is crucial; while pyridine alone can drive the reaction, the addition of alpha-picoline in dichloromethane offers an alternative pathway that may provide different impurity profiles or solubility advantages during workup. This mechanistic understanding allows process chemists to fine-tune the reaction conditions to maximize purity, a key metric for R&D directors evaluating the feasibility of this intermediate for downstream drug substance manufacturing.
How to Synthesize Scabellone A Efficiently
The synthesis of Scabellone A described in this patent follows a logical progression from simple aromatic starting materials to the complex meroterpenoid target. The process is divided into distinct operational stages: the preparation of the silylated intermediate, the geranylation, the oxidative coupling to form the dimeric quinone, and finally the cyclization. Each step has been optimized for yield and reproducibility, utilizing standard laboratory equipment and commercially available reagents. The detailed standardized synthesis steps see the guide below.
- Perform directed ortho-lithiation of 1,2,4-trimethoxybenzene followed by silylation to protect the reactive position.
- Execute a second lithiation at the remaining ortho position and react with geranyl bromide to install the terpene chain.
- Oxidize the geranyl-substituted benzene derivative using ceric ammonium nitrate (CAN) to form the dimeric quinone intermediate.
- Induce intramolecular cyclization of the quinone dimer using pyridine or alpha-picoline bases to yield Scabellone A.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition from the legacy synthesis routes to this novel methodology presents substantial strategic benefits. The primary advantage is the dramatic improvement in process efficiency, which directly correlates to cost reduction in pharmaceutical intermediate manufacturing. By eliminating multiple low-yielding steps and avoiding the use of expensive or hazardous reagents associated with the older Baeyer-Villiger and redox cycling protocols, the overall cost of production is significantly lowered. The use of 1,2,4-trimethoxybenzene as a starting material is particularly advantageous, as it is a commodity chemical with a stable global supply, reducing the risk of raw material shortages that often plague specialty syntheses relying on custom-synthesized precursors.
- Cost Reduction in Manufacturing: The streamlined synthetic route inherently reduces the consumption of solvents, reagents, and energy. In the prior art, the cumulative loss of material across numerous steps resulted in a total yield of less than 2%, meaning that over 98% of the input mass was wasted. The new process, by consolidating steps and improving the yield of the key oxidative coupling, drastically reduces the mass intensity of the process. Furthermore, the avoidance of transition metal catalysts in the final cyclization step eliminates the need for costly and time-consuming heavy metal scavenging processes, which are often required to meet strict regulatory limits for residual metals in APIs. This simplification of the downstream processing workflow translates directly into lower operational expenditures and a more competitive price point for the final intermediate.
- Enhanced Supply Chain Reliability: Supply chain continuity is heavily dependent on the robustness of the manufacturing process. The new method utilizes reagents such as n-butyllithium, geranyl bromide, and ceric ammonium nitrate, all of which are available in bulk quantities from multiple global suppliers. This diversification of the supply base mitigates the risk of single-source dependency. Additionally, the reaction conditions, while requiring temperature control, do not demand extreme pressures or specialized reactor configurations that are difficult to scale. The ability to perform the critical oxidative coupling in common solvents like acetonitrile ensures that the process can be transferred between different manufacturing sites with minimal friction, ensuring consistent supply for clinical and commercial needs.
- Scalability and Environmental Compliance: From an environmental and safety perspective, the new route offers a cleaner profile. The reduction in step count inherently reduces the volume of waste generated per kilogram of product. The oxidative coupling with CAN produces cerium salts as byproducts, which can be managed through established waste treatment protocols, avoiding the generation of complex organic waste streams associated with multi-step protecting group manipulations. The scalability of the lithiation steps is well-understood in the fine chemical industry, with established engineering controls for handling pyrophoric reagents like n-butyllithium at scale. This makes the commercial scale-up of complex pharmaceutical intermediates like Scabellone A more feasible and compliant with increasingly stringent environmental regulations regarding solvent usage and waste disposal.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Scabellone A based on the patented technology. These insights are derived from the specific experimental data and process descriptions provided in the patent documentation, offering clarity on the feasibility and advantages of this synthetic route for potential partners and licensees.
Q: How does this new synthesis method improve upon previous total synthesis routes for Scabellone A?
A: Previous methods reported in literature suffered from cumbersome multi-step sequences and extremely low overall yields of less than 2%. The patented method streamlines the process by utilizing a direct oxidative coupling strategy with ceric ammonium nitrate, significantly enhancing the overall yield and simplifying the purification workflow.
Q: What are the critical reaction conditions for the oxidative coupling step?
A: The oxidative coupling requires precise temperature control, typically between 0°C and -40°C, using acetonitrile as the solvent. The molar ratio of the substrate to the oxidant, ceric ammonium nitrate, is critical, optimized between 1:4 and 1:5 to ensure complete conversion while minimizing over-oxidation byproducts.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is designed with scalability in mind. It utilizes common organic solvents like tetrahydrofuran and acetonitrile, and avoids exotic catalysts. The use of robust reagents like n-butyllithium and ceric ammonium nitrate allows for adaptation to kilogram-scale manufacturing with standard chemical engineering controls.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Scabellone A Supplier
The development of efficient synthetic routes for bioactive natural products like Scabellone A is only the first step; translating this chemistry into reliable commercial supply requires a partner with deep technical expertise and robust manufacturing capabilities. NINGBO INNO PHARMCHEM stands at the forefront of this translation, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facility is equipped with state-of-the-art reactors capable of handling the cryogenic conditions required for the lithiation steps and the precise temperature control needed for the oxidative coupling, ensuring that the stringent purity specifications demanded by the pharmaceutical industry are consistently met. Our rigorous QC labs employ advanced analytical techniques to monitor every stage of the synthesis, guaranteeing that the impurity profile of our Scabellone A intermediates remains within tight limits suitable for GMP API synthesis.
We invite R&D and procurement leaders to collaborate with us to leverage this advanced technology for your drug development programs. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this optimized route can reduce your overall project costs. We encourage you to contact our technical procurement team to request specific COA data for our pilot batches and to discuss route feasibility assessments for your specific needs. Let us help you secure a stable, high-quality supply of this critical pharmaceutical intermediate, accelerating your path from discovery to market.
