Advanced Cyclopropane Skeleton Ligands for Efficient Organoboron Intermediate Production
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to access complex organoboron intermediates, which serve as critical building blocks for Suzuki-Miyaura cross-coupling reactions. A significant technological breakthrough in this domain is detailed in Chinese Patent CN115850328A, which discloses a novel class of diphosphine ligands featuring a rigid cyclopropane skeleton and their corresponding cobalt complexes. This innovation addresses long-standing challenges in the hydroboration of internal alkynes, a transformation that has historically suffered from poor regioselectivity and a reliance on scarce precious metals. By leveraging the unique steric and electronic properties of gem-diaryl substituted cyclopropane backbones, this technology enables the use of earth-abundant cobalt to achieve high activity and unprecedented selectivity profiles. For R&D teams and procurement strategists alike, this represents a pivotal shift towards more sustainable and cost-effective catalytic solutions for generating high-value alkenyl boron compounds.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
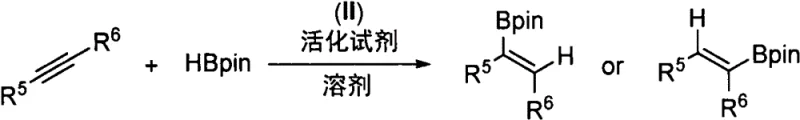
Traditional methods for synthesizing organoboron compounds often rely on the Miyaura borylation reaction, which necessitates the pre-functionalization of substrates with halides or triflates, thereby increasing step count and waste generation. Alternatively, direct hydroboration using precious metal catalysts like iridium or rhodium has been explored, but these systems frequently struggle with internal alkynes. Specifically, controlling the regioselectivity between alpha-addition and beta-addition remains a formidable hurdle, particularly when the steric difference between the alkyne substituents is minimal. Furthermore, the high cost and supply chain volatility associated with noble metals like iridium pose significant economic risks for large-scale manufacturing processes. Existing base metal alternatives often lack the necessary activity or selectivity, leading to mixtures of isomers that require difficult and yield-eroding purification steps, ultimately compromising the efficiency of the overall synthetic route.
The Novel Approach
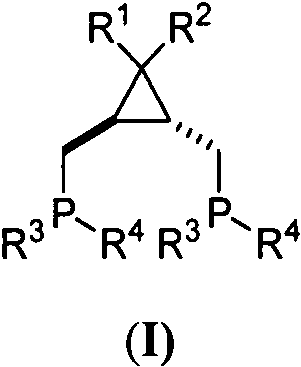
The technology described in Patent CN115850328A introduces a paradigm shift by utilizing a specifically designed cyclopropane-based diphosphine ligand coordinated to cobalt dichloride. This novel catalyst system effectively overcomes the regioselectivity barriers inherent in internal alkyne hydroboration. Unlike conventional systems that default to addition at the less hindered position, this cobalt complex can be tuned to favor beta-addition in aryl-alkyl internal alkynes and even achieve the previously elusive alpha-addition selectivity in dialkyl internal alkynes. The rigid cyclopropane scaffold imposes a defined geometric constraint on the cobalt center, creating a steric environment that directs the pinacolborane (HBpin) addition with high precision. This capability not only simplifies the synthesis of diverse alkenyl boronates but also eliminates the need for expensive noble metals, offering a robust alternative for the cost reduction in organoboron intermediate manufacturing.
Mechanistic Insights into Cobalt-Catalyzed Hydroboration
The exceptional performance of this catalyst stems from the intricate interplay between the ligand architecture and the cobalt metal center. The ligand features a trans-substituted cyclopropane ring with gem-diaryl groups at the apex, which serves as a bulky, rigid anchor. This structural motif prevents unfavorable conformational rotations that typically lead to loss of selectivity in flexible ligand systems. When coordinated with cobalt, the diphosphine ligand creates a distinct coordination sphere that activates the B-H bond of pinacolborane while simultaneously orienting the internal alkyne substrate. The mechanistic pathway likely involves the formation of a cobalt-hydride species that inserts into the alkyne triple bond. The steric bulk of the gem-diaryl groups on the cyclopropane ring effectively shields one face of the metal center, forcing the alkyne to approach in a specific orientation that dictates whether the boron adds to the alpha or beta carbon. This precise control allows for the synthesis of specific alkene isomers that are difficult to access via other methods.
Furthermore, the catalyst exhibits remarkable functional group tolerance, a critical factor for its application in complex molecule synthesis. The mild reaction conditions, often proceeding at room temperature in common solvents like toluene, ensure that sensitive moieties such as esters, ethers, halides, and amines remain intact throughout the transformation. This compatibility reduces the need for extensive protecting group strategies, thereby streamlining the synthetic sequence and improving overall atom economy. The ability to handle a broad substrate scope, ranging from aryl-alkyl alkynes to dialkyl alkynes, underscores the versatility of this catalytic system. For process chemists, this means a single catalyst platform can potentially service multiple stages of a synthesis campaign, reducing the inventory of specialized reagents required and simplifying the supply chain logistics for high-purity organoboron intermediates.
How to Synthesize Cyclopropane Diphosphine Ligand Cobalt Complex Efficiently
The preparation of these advanced catalysts follows a logical and scalable four-step sequence that begins with readily available starting materials. The process initiates with the cyclopropanation of dimethyl fumarate with diaryl diazo compounds to establish the core skeleton, followed by reduction to the diol and conversion to the bis-mesylate. The final ligand is generated via nucleophilic substitution with lithium diphenylphosphide, which is then complexed with cobalt chloride. This route avoids extreme cryogenic conditions beyond standard laboratory capabilities and utilizes standard unit operations, making it highly amenable to technology transfer. The detailed standardized synthesis steps for producing the active catalyst are outlined in the guide below.
- Perform cyclopropanation of dimethyl fumarate with diaryl diazo compounds at 50-120°C to form trans-substituted cyclopropane diesters.
- Reduce the diester intermediate using LiAlH4 at -30 to 30°C to obtain the corresponding trans-hydroxymethylene cyclopropane derivative.
- Convert the hydroxyl groups to mesylates using MsCl and Et3N, followed by substitution with lithium diphenylphosphide to yield the diphosphine ligand.
- Complex the purified ligand with CoCl2 in an organic solvent at 0-120°C to generate the final active cobalt catalyst.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this cobalt-based catalytic technology offers substantial strategic benefits beyond mere technical performance. The most immediate impact is seen in the raw material cost structure, where replacing scarce precious metals with abundant cobalt drastically reduces the bill of materials. Additionally, the simplified downstream processing resulting from high selectivity minimizes solvent usage and waste disposal costs, contributing to a greener and more economical manufacturing footprint. These factors collectively enhance the commercial viability of producing complex organoboron intermediates at scale.
- Cost Reduction in Manufacturing: The substitution of expensive iridium or rhodium catalysts with earth-abundant cobalt represents a significant opportunity for cost optimization. Precious metals are subject to volatile market pricing and geopolitical supply risks, whereas cobalt is more readily available and cost-stable. Furthermore, the high regioselectivity of this system reduces the formation of unwanted isomers, which eliminates the need for costly and yield-loss-inducing chromatographic separations. This streamlined purification process directly translates to lower operational expenditures and higher overall process efficiency, ensuring a more competitive cost structure for the final organoboron products.
- Enhanced Supply Chain Reliability: Relying on noble metal catalysts often introduces bottlenecks due to limited global supply and long lead times for high-purity grades. By transitioning to a cobalt-based system, manufacturers can diversify their supplier base and mitigate the risk of raw material shortages. The ligand synthesis itself utilizes commodity chemicals such as fumarates and diazo compounds, which are produced on a large industrial scale. This reliance on widely available feedstocks ensures a consistent and reliable supply of the catalyst, preventing production delays and enabling better long-term planning for continuous manufacturing campaigns.
- Scalability and Environmental Compliance: The reaction conditions described in the patent are mild, typically operating at room temperature or slightly elevated temperatures in common organic solvents like toluene or THF. This eliminates the need for energy-intensive heating or cooling infrastructure, reducing the carbon footprint of the manufacturing process. Moreover, the high atom economy of the hydroboration reaction, combined with the reduced need for purification solvents, aligns with increasingly stringent environmental regulations. The robustness of the catalyst allows for potential recycling or recovery strategies, further minimizing waste generation and supporting sustainability goals within the chemical supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this cyclopropane ligand technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear understanding of the system's capabilities and limitations for potential adopters.
Q: What is the primary advantage of this cobalt catalyst over traditional iridium systems?
A: The primary advantage is the use of earth-abundant cobalt instead of expensive precious metals like iridium or rhodium, significantly lowering raw material costs while achieving unique regioselectivity (beta-addition for aryl-alkyl alkynes) that is difficult to control with conventional systems.
Q: Can this catalyst system tolerate sensitive functional groups?
A: Yes, the patent data demonstrates excellent functional group tolerance, successfully catalyzing reactions with substrates containing esters, ethers, halides, and amines without requiring protective group strategies, which streamlines the synthetic route.
Q: Is the ligand synthesis scalable for industrial production?
A: The synthesis utilizes robust, scalable reactions such as cyclopropanation and nucleophilic substitution under mild conditions (-78°C to room temperature), avoiding cryogenic extremes and hazardous reagents, making it highly suitable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Organoboron Intermediates Supplier
The development of cyclopropane skeleton diphosphine ligands and their cobalt complexes marks a significant advancement in the field of transition metal catalysis, offering a powerful tool for the synthesis of valuable alkenyl boron compounds. At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this technology for the pharmaceutical and agrochemical sectors. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, ensuring that every batch of catalyst or intermediate delivered meets the highest international standards for quality and consistency.
We invite you to explore how this innovative catalytic system can optimize your current synthetic routes and reduce your overall manufacturing costs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific project needs. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments. By collaborating with us, you gain access to cutting-edge chemical technologies and a reliable supply chain partner committed to driving your projects from bench-scale discovery to full-scale commercial success.
