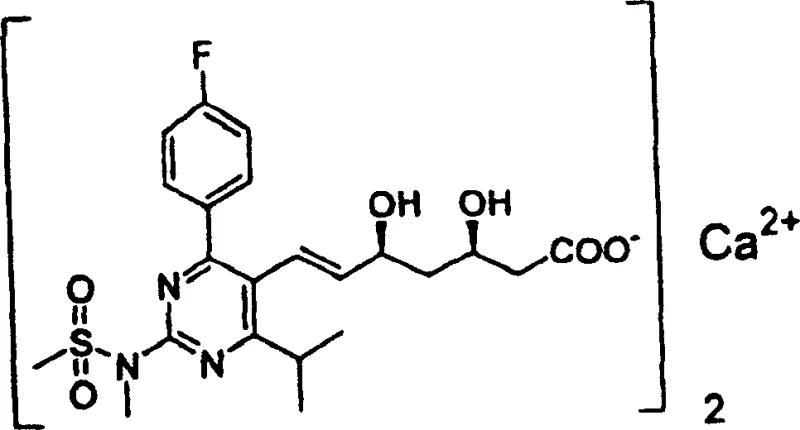
Advanced Synthesis of Aminopyrimidine Compounds for Commercial Pharmaceutical Manufacturing
The pharmaceutical industry continuously seeks robust synthetic routes for critical intermediates that ensure both high purity and economic viability. Patent CN100349877C introduces a groundbreaking preparation method for aminopyrimidine compounds, specifically targeting the synthesis of 2-(N-methyl-N-methanesulfonylamino)pyrimidine derivatives. These structures are not merely academic curiosities but serve as the foundational backbone for potent HMG-CoA reductase inhibitors, widely recognized for their efficacy in managing cholesterol levels. The technical breakthrough lies in a multi-step sequence that begins with the condensation of isobutyrylacetate esters and culminates in a highly selective amination process. By leveraging specific metal salt catalysts and controlled oxidation conditions, this methodology overcomes the yield limitations and safety hazards associated with earlier generations of synthetic protocols. For R&D directors and procurement specialists, understanding this pathway is essential for securing a reliable supply chain of high-purity pharmaceutical intermediates. The process demonstrates exceptional adaptability, allowing for the modification of R groups to tailor the final compound for specific therapeutic applications without compromising the integrity of the core pyrimidine ring. This flexibility is paramount in the fast-paced environment of drug development where structural optimization is frequent.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to the innovations detailed in this patent, the industrial preparation of similar aminopyrimidine compounds relied heavily on methodologies that were fraught with significant inefficiencies and safety concerns. For instance, earlier disclosures such as WO01/04100 described routes that necessitated the use of toxic reagents like mauguinite, which posed severe environmental and occupational health risks during large-scale manufacturing. Furthermore, the overall recovery rates associated with these conventional methods were disappointingly low, often hovering around 45.5% based on the starting isobutyrylacetate ester. Such low yields translate directly into increased waste generation, higher raw material consumption, and escalated production costs, creating a substantial bottleneck for procurement managers aiming to optimize budgets. The reliance on hazardous chemicals also complicates the waste treatment process, requiring specialized infrastructure to handle toxic byproducts safely. Additionally, the multi-step nature of older routes often involved unstable intermediates that degraded easily, leading to inconsistent product quality and purity profiles. These factors combined to make the conventional synthesis of complex pyrimidine derivatives a high-risk endeavor, unsuitable for the rigorous demands of modern Good Manufacturing Practice (GMP) environments where consistency and safety are non-negotiable standards.
The Novel Approach
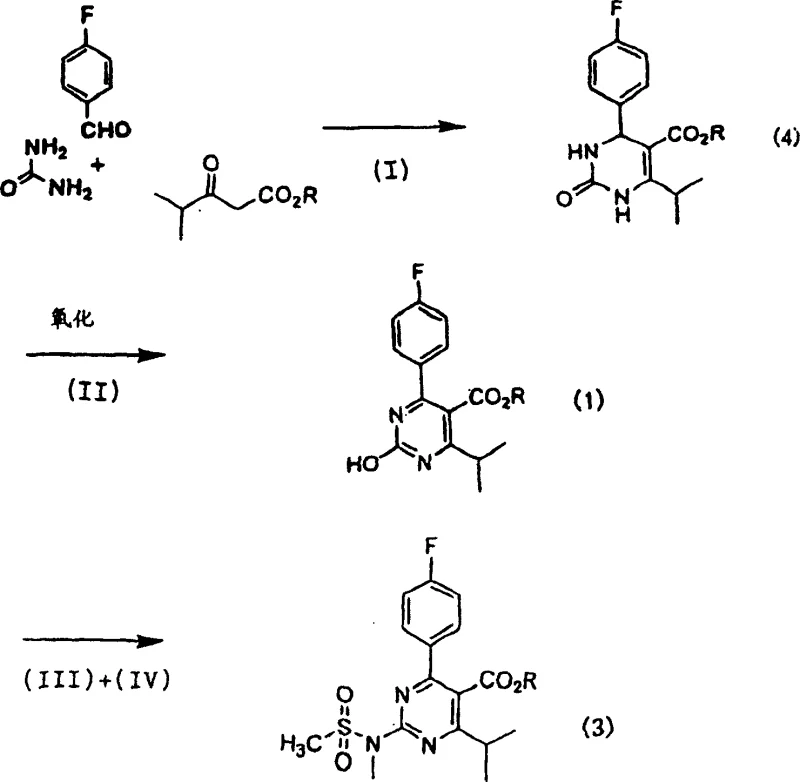
In stark contrast, the novel approach outlined in CN100349877C presents a streamlined and chemically elegant solution that addresses the core deficiencies of the prior art. By utilizing a combination of protonic compounds and specific metal salts such as cuprous chloride or ferric chloride, the new method facilitates a highly efficient condensation reaction that forms the dihydropyrimidinone core with exceptional precision. This catalytic system not only accelerates the reaction kinetics but also minimizes the formation of unwanted side products, thereby simplifying the downstream purification process. The subsequent oxidation step employs nitric acid under controlled conditions, a reagent that is both cost-effective and readily available on a global scale, ensuring supply chain stability. Crucially, this route eliminates the need for toxic starting materials, replacing them with safer alternatives that align with modern environmental regulations and corporate sustainability goals. The result is a synthesis pathway that delivers significantly higher yields, often exceeding 85% in the initial condensation step and maintaining high efficiency through subsequent transformations. This dramatic improvement in process efficiency means that manufacturers can produce more active pharmaceutical ingredient (API) intermediate per batch, effectively reducing the cost of goods sold while enhancing the reliability of supply for downstream drug manufacturers.
Mechanistic Insights into Metal-Catalyzed Condensation and Oxidation
The heart of this synthetic innovation lies in the intricate interplay between the metal salt catalyst and the protonic acid during the initial Biginelli-like condensation reaction. The mechanism begins with the activation of the carbonyl group in the isobutyrylacetate ester by the metal salt, which increases its electrophilicity and makes it more susceptible to nucleophilic attack by the urea nitrogen. Simultaneously, the protonic compound, such as sulfuric acid or p-toluenesulfonic acid, facilitates the dehydration steps necessary to close the pyrimidine ring, driving the equilibrium towards the desired dihydropyrimidinone product. This dual-activation strategy ensures that the reaction proceeds smoothly even at moderate temperatures, typically between 30°C and 100°C, which helps to preserve the integrity of sensitive functional groups like the fluoro-phenyl moiety. The choice of solvent, ranging from alcohols like methanol to aprotic solvents like acetonitrile, further fine-tunes the reaction environment to maximize solubility and reaction rates. Following the ring formation, the oxidation step utilizes nitric acid to introduce the necessary unsaturation or hydroxyl functionality at the 2-position of the pyrimidine ring. This oxidation is carefully managed to prevent over-oxidation or degradation of the ester side chain, a common pitfall in less optimized protocols. The precise control of stoichiometry and temperature during this phase is critical for maintaining the high purity required for pharmaceutical applications, ensuring that the final intermediate meets stringent regulatory specifications for impurity profiles.
Impurity control is another cornerstone of this mechanistic design, particularly given the complex nature of heterocyclic synthesis where side reactions can easily occur. The protocol incorporates specific purification steps, such as crystallization and recrystallization from solvents like toluene or isopropyl alcohol, which are strategically selected to exclude structurally similar byproducts. The use of metal salts like cuprous chloride not only catalyzes the reaction but also helps in suppressing the formation of polymeric impurities that often plague condensation reactions involving urea and beta-keto esters. Furthermore, the oxidation step is followed by a neutralization and extraction process that effectively removes inorganic salts and acidic residues, preventing them from carrying over into the final sulfonylation stages. This rigorous attention to detail in the workup procedure ensures that the hydroxy pyrimidine intermediate entering the final steps is of exceptional quality. By minimizing the impurity load early in the synthesis, the burden on the final purification stages is significantly reduced, leading to higher overall recovery and a cleaner final product. For quality control teams, this means more consistent analytical data and a reduced risk of batch failure due to out-of-specification impurity levels, which is a critical factor in maintaining a reliable supply of high-purity pharmaceutical intermediates.
How to Synthesize 2-(N-methyl-N-methanesulfonylamino)pyrimidine Efficiently
Executing this synthesis requires a disciplined approach to reaction conditions and reagent addition to fully realize the yield and purity benefits described in the patent. The process is designed to be robust, allowing for operation in standard glass-lined reactors commonly found in fine chemical manufacturing facilities. Operators must pay close attention to the temperature profiles during the exothermic condensation and oxidation phases to ensure safety and reproducibility. The detailed standardized synthesis steps involve precise molar ratios of the metal catalyst and proton source, which are critical for initiating the reaction correctly. Following the initial condensation, the oxidation must be quenched carefully to avoid thermal runaways, and the subsequent sulfonylation requires anhydrous conditions to prevent hydrolysis of the sulfonyl halide. These operational nuances are key to translating the laboratory-scale success of the patent into a commercially viable manufacturing process. For technical teams looking to implement this route, adherence to the specified parameters is essential for achieving the reported high yields and maintaining the structural integrity of the final aminopyrimidine compound.
- Condense isobutyrylacetate ester with 4-fluorobenzaldehyde and urea using a metal salt catalyst to form dihydropyrimidinone.
- Oxidize the dihydropyrimidinone intermediate using nitric acid to generate the hydroxy pyrimidine compound.
- React the hydroxy pyrimidine with organic sulfonyl halide followed by N-methyl-N-methanesulfonamide to finalize the aminopyrimidine structure.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthesis route offers profound advantages for procurement managers and supply chain heads tasked with securing cost-effective and reliable sources of complex intermediates. The elimination of toxic reagents like mauguinite not only reduces the environmental footprint of the manufacturing process but also significantly lowers the costs associated with hazardous waste disposal and regulatory compliance. This shift towards safer chemistry translates into a more sustainable supply chain that is less vulnerable to regulatory crackdowns or environmental shutdowns, ensuring greater continuity of supply for downstream pharmaceutical customers. Furthermore, the use of readily available starting materials such as 4-fluorobenzaldehyde and urea mitigates the risk of raw material shortages, as these commodities are produced by a wide network of global suppliers. The high yield of the process means that less raw material is required to produce the same amount of product, directly impacting the cost of goods sold and allowing for more competitive pricing strategies in the market. For supply chain planners, the scalability demonstrated in the patent, with successful runs in 200L reactors, provides confidence that the process can be ramped up to meet large-volume demands without significant re-engineering. This scalability is crucial for supporting the commercial launch of new drugs where demand can surge rapidly, requiring a supplier capable of scaling production from kilograms to metric tons seamlessly.
- Cost Reduction in Manufacturing: The streamlined nature of this synthesis route drives substantial cost savings by reducing the number of purification steps and minimizing raw material waste. By achieving higher yields in the initial condensation and oxidation steps, the process maximizes the conversion of expensive starting materials into valuable intermediates, thereby lowering the effective cost per kilogram of the final product. The avoidance of specialized toxic reagents also eliminates the need for costly safety infrastructure and specialized waste treatment protocols, further reducing the overhead associated with production. Additionally, the use of common solvents and catalysts allows for procurement teams to leverage existing supplier relationships and bulk purchasing power, driving down input costs even further. These cumulative efficiencies result in a manufacturing process that is not only chemically superior but also economically optimized for high-volume commercial production.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals and standard reaction conditions enhances the resilience of the supply chain against disruptions. Since the reagents required for this synthesis are widely available from multiple global sources, the risk of supply bottlenecks due to single-source dependency is significantly minimized. The robustness of the reaction conditions also means that the process can be transferred between different manufacturing sites with minimal loss of efficiency, providing flexibility in production planning. This geographical flexibility allows supply chain managers to diversify production locations, mitigating risks associated with regional instability or logistics challenges. Furthermore, the high purity of the intermediate reduces the likelihood of batch rejections due to quality issues, ensuring a steady flow of material to the API synthesis stage. This reliability is essential for maintaining the production schedules of finished dosage forms and meeting the critical delivery timelines expected by pharmaceutical partners.
- Scalability and Environmental Compliance: The process is inherently designed for scale, with patent examples demonstrating successful execution in large-scale glass-lined reactors, proving its readiness for industrial adoption. The simplified workup procedures and the use of recyclable solvents contribute to a greener manufacturing profile that aligns with increasingly stringent environmental regulations. By reducing the generation of hazardous waste and lowering energy consumption through optimized reaction temperatures, this method supports corporate sustainability initiatives and reduces the carbon footprint of the supply chain. The ability to scale up without compromising safety or quality makes this route an ideal choice for long-term commercial partnerships where volume requirements are expected to grow over time. This scalability ensures that the supplier can grow alongside the customer, supporting the commercial success of the final drug product from clinical trials through to full-scale market launch.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of these aminopyrimidine compounds, based on the detailed data provided in the patent documentation. Understanding these aspects is crucial for stakeholders evaluating the feasibility of integrating this intermediate into their supply chain. The answers reflect the specific advantages of the disclosed method over traditional approaches, highlighting its suitability for modern pharmaceutical manufacturing standards. These insights are intended to clarify the technical capabilities and commercial benefits associated with this advanced synthetic route.
Q: What are the advantages of this synthesis method over prior art?
A: This method avoids toxic reagents like mauguinite used in previous processes and achieves significantly higher yields, making it safer and more economically viable for industrial scale-up.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the patent explicitly describes successful execution in 200L glass-lined reactors, demonstrating robust scalability and reproducibility for commercial manufacturing.
Q: What is the primary application of these aminopyrimidine compounds?
A: These compounds serve as critical intermediates in the synthesis of HMG-CoA reductase inhibitors, specifically for producing cholesterol-lowering agents like rosuvastatin calcium.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aminopyrimidine Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development and commercialization of life-saving medications. Our team of expert chemists and engineers possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and reliability. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs that employ state-of-the-art analytical techniques to verify every batch. Our facility is equipped to handle complex synthetic routes like the one described in CN100349877C, leveraging our expertise in heterocyclic chemistry to optimize yields and minimize impurities. By partnering with us, you gain access to a supply chain that is not only robust and scalable but also deeply committed to quality and regulatory compliance. We understand the pressures of drug development timelines and are dedicated to providing a seamless supply experience that allows you to focus on your core research and commercialization goals.
We invite you to engage with our technical procurement team to discuss how we can support your specific requirements for aminopyrimidine compounds and related intermediates. Request a Customized Cost-Saving Analysis to understand how our optimized manufacturing processes can reduce your overall production costs without compromising quality. Our team is ready to provide specific COA data and route feasibility assessments to help you make informed decisions about your supply chain strategy. Whether you are in the early stages of process development or looking to secure a long-term commercial supplier, NINGBO INNO PHARMCHEM is positioned to be your trusted partner in bringing innovative therapies to market. Contact us today to initiate a conversation about your project and discover the value of our technical expertise.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
