Scalable Synthesis of 2-Alkoxy-2-cyclopenten-1-one via Iodine-Copper Catalysis for Commercial API Production
The landscape of pharmaceutical intermediate manufacturing is constantly evolving, driven by the need for more efficient, cost-effective, and environmentally sustainable synthetic routes. A significant advancement in this domain is detailed in Chinese Patent CN112811970B, which discloses a novel preparation method for 2-alkoxy-2-cyclopenten-1-one derivatives. These cyclic enone structures serve as critical scaffolds in the synthesis of various bioactive molecules and drug candidates. The patented methodology represents a paradigm shift from conventional approaches by utilizing a tandem reaction sequence involving iodination, substitution, oxidation, and addition-elimination. By employing cyclopentanone and aliphatic alcohols as primary substrates in the presence of equivalent iodine and specific copper salts, this technology offers a streamlined pathway that addresses long-standing challenges in yield optimization and raw material accessibility. For R&D directors and procurement specialists seeking reliable sources for complex heterocyclic building blocks, understanding the mechanistic nuances and commercial implications of this patent is essential for strategic supply chain planning.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-alkoxy-2-cyclopenten-1-one has relied heavily on the condensation of 1,2-cyclopentanedione with alkoxy reagents under acidic catalysis. While chemically feasible, this traditional route suffers from several intrinsic drawbacks that hinder its utility in modern industrial settings. Firstly, the starting material, 1,2-cyclopentanedione, is not as commercially ubiquitous or cost-effective as simple cyclopentanone, leading to higher raw material costs and potential supply chain bottlenecks. Secondly, the reaction conditions often require stringent control to prevent over-reaction or decomposition, resulting in generally low yields that are economically unsustainable for large-scale production. Furthermore, the narrow substrate scope of the acid-catalyzed method limits the structural diversity accessible to medicinal chemists, restricting the exploration of structure-activity relationships (SAR) during drug discovery phases. The operational complexity associated with handling strong acids and sensitive diketones also poses safety and environmental concerns, necessitating specialized equipment and waste treatment protocols that increase the overall manufacturing footprint.
The Novel Approach
In stark contrast, the innovative strategy outlined in the patent data leverages a direct oxidative functionalization of cyclopentanone, a commodity chemical available in bulk quantities globally. This approach utilizes a synergistic catalytic system comprising molecular iodine and copper salts, such as copper(II) oxide, to facilitate the transformation under relatively mild thermal conditions. The reaction proceeds efficiently at 100°C over a 12-hour period, eliminating the need for cryogenic temperatures or ultra-high pressure equipment. As illustrated in the reaction scheme below, the process directly couples cyclopentanone with a wide variety of aliphatic alcohols to generate the target enone structure with high atom economy.
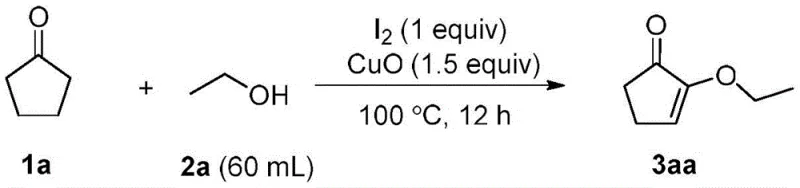
This novel route not only expands the substrate scope to include methyl, ethyl, propyl, butyl, and isopropyl alcohols but also simplifies the downstream processing. The byproducts generated are predominantly inorganic salts and water, which are far easier to manage and dispose of compared to the organic waste streams typical of traditional acid-catalyzed processes. This fundamental shift in synthetic design translates directly into enhanced process robustness and operational simplicity, making it an attractive candidate for adoption by forward-thinking chemical manufacturers.
Mechanistic Insights into Iodine-Copper Catalyzed Oxidative Functionalization
The success of this transformation lies in the intricate interplay between the iodine oxidant and the copper catalyst, which orchestrates a multi-step tandem sequence. Mechanistically, the reaction initiates with the alpha-iodination of cyclopentanone, activated by the iodine species. This is followed by a nucleophilic substitution where the aliphatic alcohol attacks the activated position. Subsequently, the copper salt, acting as both a Lewis acid and an oxidant, facilitates the dehydrogenation and elimination steps required to establish the conjugated enone system. The use of copper(II) oxide is particularly advantageous as it serves as a heterogeneous oxidant that can be easily removed via filtration post-reaction, thereby minimizing metal contamination in the final product. This is a critical consideration for pharmaceutical applications where residual heavy metal levels are strictly regulated by guidelines such as ICH Q3D.
From an impurity control perspective, this mechanism offers distinct advantages over acid-catalyzed alternatives. The absence of strong protic acids reduces the likelihood of polymerization side reactions or etherification of the alcohol solvent, which are common impurities in traditional methods. The reaction environment is sufficiently controlled to favor the formation of the desired 2-alkoxy-2-cyclopenten-1-one isomer, minimizing the generation of regioisomers or over-oxidized byproducts. The patent data indicates that optimization of the copper salt loading (ranging from 0.5 to 1.5 equivalents) allows for fine-tuning the reaction kinetics to maximize conversion while maintaining high selectivity. This level of control ensures that the resulting crude product requires less intensive purification, further enhancing the overall process efficiency and reducing solvent consumption during chromatography.
How to Synthesize 2-Alkoxy-2-cyclopenten-1-one Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific operational parameters to ensure reproducibility and safety. The general protocol involves charging a reaction vessel with cyclopentanone, molecular iodine, and the selected copper salt, followed by the addition of the alcohol which serves as both reactant and solvent. The mixture is then heated to reflux conditions, typically around 100°C, and maintained for a duration of 12 hours to ensure complete conversion. Post-reaction, the solid copper residues are removed by simple filtration, and the filtrate is concentrated under reduced pressure. Final purification is achieved through standard column chromatography techniques to isolate the pure enone derivative. For detailed standard operating procedures and specific stoichiometric ratios tailored to different alcohol substrates, please refer to the step-by-step guide below.
- Charge a reaction vessel with cyclopentanone, molecular iodine, and copper oxide in a molar ratio of approximately 1: 1:1.5.
- Add the corresponding aliphatic alcohol (20-60 mL) as both reactant and solvent to the mixture.
- Heat the reaction mixture to 100°C and maintain stirring for 12 hours to complete the oxidative coupling.
- Upon completion, filter the mixture, concentrate via vacuum distillation, and purify the crude product using column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic route offers compelling economic and logistical benefits that extend beyond mere chemical yield. The primary advantage stems from the utilization of cyclopentanone and common aliphatic alcohols as feedstocks. These are high-volume commodity chemicals with stable global supply chains, insulating manufacturers from the volatility often associated with specialized fine chemical precursors. By shifting away from scarce 1,2-cyclopentanedione, companies can secure long-term raw material contracts at significantly lower price points, directly impacting the cost of goods sold (COGS). Furthermore, the operational simplicity of the process—specifically the lack of requirement for anhydrous or oxygen-free environments—reduces the capital expenditure needed for specialized reactor infrastructure. Facilities can utilize standard glass-lined or stainless steel reactors without the need for extensive drying systems or inert gas blanketing, thereby lowering overhead costs.
- Cost Reduction in Manufacturing: The elimination of expensive and sensitive starting materials combined with the use of earth-abundant copper catalysts drastically lowers the direct material costs. Additionally, the simplified workup procedure, which relies on filtration and distillation rather than complex extraction or quenching steps, reduces labor hours and solvent usage. This streamlined workflow translates into substantial cost savings per kilogram of produced intermediate, allowing for more competitive pricing strategies in the global market.
- Enhanced Supply Chain Reliability: Dependence on a single source for exotic reagents is a major risk factor in pharmaceutical supply chains. This method mitigates that risk by relying on widely available reagents that can be sourced from multiple vendors globally. The robustness of the reaction conditions also means that production schedules are less likely to be disrupted by minor fluctuations in environmental conditions or reagent quality, ensuring consistent delivery timelines to downstream API manufacturers.
- Scalability and Environmental Compliance: The generation of benign byproducts such as water and inorganic salts aligns perfectly with increasingly stringent environmental regulations. This reduces the burden on wastewater treatment facilities and lowers the costs associated with hazardous waste disposal. The process is inherently scalable, having been demonstrated to work effectively with varying volumes of alcohol solvent, which suggests a smooth path from gram-scale R&D to multi-ton commercial production without significant re-engineering of the process parameters.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of this technology for their specific projects, we have compiled answers to common inquiries regarding the reaction scope, catalyst selection, and scalability. These insights are derived directly from the experimental data provided in the patent documentation, ensuring accuracy and relevance for process development discussions. Understanding these technical details is crucial for making informed decisions about adopting this methodology for your pipeline.
Q: What are the advantages of this iodine-copper method over traditional acid-catalyzed routes?
A: Unlike traditional methods requiring scarce 1,2-cyclopentanedione and harsh acidic conditions, this patent-disclosed route utilizes readily available cyclopentanone and common alcohols. It operates under neutral to mild conditions without strict anhydrous requirements, significantly simplifying operational complexity and improving overall yield.
Q: Is this process suitable for large-scale manufacturing of pharmaceutical intermediates?
A: Yes, the process is highly amenable to scale-up. It avoids sensitive reagents and complex inert atmosphere operations. The workup involves simple filtration and distillation, and the byproducts are primarily inorganic salts and water, aligning well with green chemistry principles required for industrial production.
Q: What is the substrate scope for the alcohol component in this reaction?
A: The method demonstrates broad substrate tolerance. It effectively converts various primary and secondary aliphatic alcohols, including methanol, ethanol, n-propanol, n-butanol, isobutanol, and isopropanol, into the corresponding 2-alkoxy-2-cyclopenten-1-one derivatives with varying efficiencies.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Alkoxy-2-cyclopenten-1-one Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced synthetic methodologies like the iodine-copper catalyzed route described in CN112811970B. As a premier CDMO partner, we possess the technical expertise and infrastructure to translate such innovative patent technologies into robust commercial processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab bench to market is seamless. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of 2-alkoxy-2-cyclopenten-1-one meets the highest industry standards for pharmaceutical intermediates.
We invite you to collaborate with us to leverage this cost-effective and scalable synthesis for your drug development programs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to innovation can drive value and efficiency in your supply chain.
