Scalable Synthesis of Aryl Substituted Olefinic Amines for CNS Therapeutics
Scalable Synthesis of Aryl Substituted Olefinic Amines for CNS Therapeutics
The pharmaceutical industry continuously seeks robust manufacturing pathways for complex intermediates, particularly those targeting central nervous system (CNS) disorders. Patent CN103180277A discloses a groundbreaking改进 process for the preparation of aryl substituted olefinic amines, specifically compounds like (2S)-(4E)-N-methyl-5-[3-(5-isopropoxypyridinyl)]-4-penten-2-amine. These molecules serve as potent neuronal nicotinic receptor agonists, holding significant therapeutic potential for treating Alzheimer's disease, schizophrenia, and cognitive impairments. The disclosed technology represents a paradigm shift from traditional stoichiometric methods to highly efficient catalytic systems, addressing critical pain points in yield, purity, and environmental impact. By optimizing the Grignard reaction conditions and refining the palladium-catalyzed cross-coupling steps, this methodology offers a reliable pharmaceutical intermediate supplier with a distinct competitive advantage in producing high-purity CNS drug precursors.
The core innovation lies in the strategic modification of reaction parameters that govern selectivity and conversion rates. Traditional syntheses often suffer from excessive reagent consumption and difficult downstream processing, which can bottleneck commercial scale-up of complex pharmaceutical intermediates. This new approach introduces a catalytic copper system assisted by lithium chloride, dramatically reducing metal loadings while enhancing reaction kinetics. Furthermore, the implementation of a biphasic solvent system in the final coupling stage facilitates unprecedented ease of purification. For R&D directors and process chemists, understanding these mechanistic nuances is vital for translating laboratory success into industrial reality, ensuring that the final active pharmaceutical ingredients meet stringent regulatory specifications for residual metals and impurities.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral pentenyl amine intermediates relied heavily on stoichiometric amounts of copper salts to mediate Grignard additions to epoxides. In previous methodologies, such as those referenced in WO2006/053082, the process required approximately 0.15 molar equivalents of copper iodide relative to the epoxide substrate. This high loading not only inflated raw material costs but also generated substantial quantities of heavy metal waste, complicating disposal and increasing the environmental footprint of the manufacturing process. Additionally, the workup procedures were cumbersome, necessitating filtration steps to remove insoluble copper salts, which often led to product entrapment and yield losses. The use of polar aprotic solvents like N,N-dimethylformamide (DMF) and dichloromethane in subsequent steps further exacerbated these issues, as these solvents are difficult to recover and pose significant health and safety risks in large-scale operations.
Another critical bottleneck in conventional routes was the extended reaction times and thermal instability of intermediates. For instance, the amination step typically required refluxing for up to 48 hours to achieve complete conversion, exposing the sensitive olefinic backbone to potential degradation and isomerization. The purification of the resulting amine often involved distillation of oily residues, a process prone to thermal decomposition and polymerization. Moreover, the final palladium-catalyzed coupling steps frequently resulted in the precipitation of Pd(0) black at the end of the reaction, leading to equipment fouling and inconsistent catalyst performance in batch-to-batch operations. These cumulative inefficiencies made cost reduction in API manufacturing challenging, as the overall yield from starting materials to the final protected amine hovered around merely 44%, leaving significant room for process intensification.
The Novel Approach
The improved process described in the patent fundamentally re-engineers the synthetic pathway to overcome these historical limitations through precise catalytic tuning and solvent engineering. A standout improvement is the replacement of stoichiometric copper with a catalytic system comprising only 0.01 molar equivalents of cuprous chloride and 0.02 molar equivalents of lithium chloride. This synergistic combination not only maintains high conversion rates but also eliminates the need for filtration, allowing the reaction mixture to be telescoped directly into the subsequent tosylation step. The switch to ether-based solvents like tetrahydrofuran (THF) and methyl tert-butyl ether (MTBE) enhances safety profiles and simplifies solvent recovery. Consequently, the reaction time for the amination step is drastically reduced from 48 hours to just 8 hours, significantly boosting throughput capacity without compromising the stereochemical integrity of the chiral center.
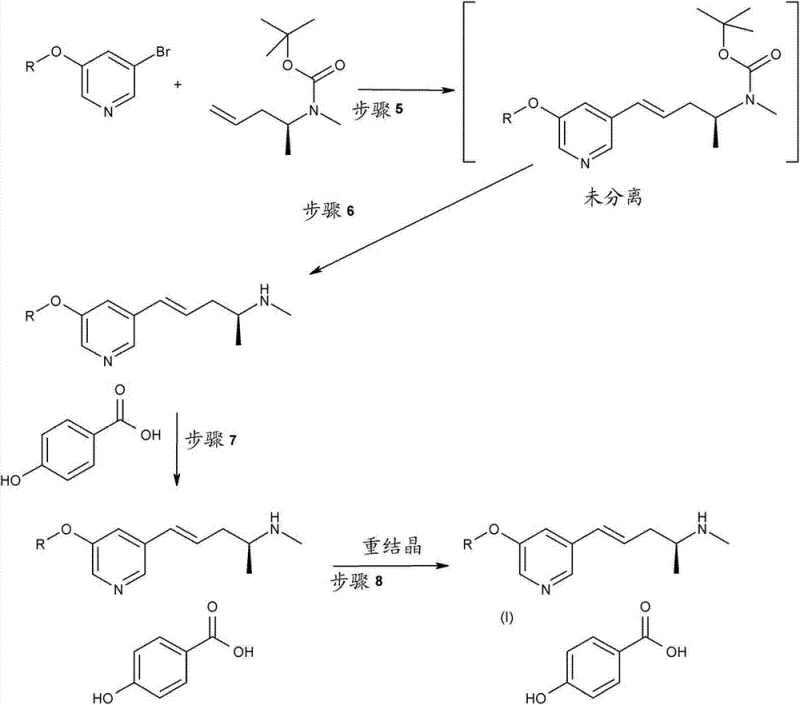
In the critical cross-coupling stage, the novel approach utilizes a toluene-water biphasic system instead of homogeneous DMF solutions. This strategic solvent choice leverages the immiscibility of toluene and water to create an efficient partitioning mechanism. Upon completion of the Heck coupling, the addition of acid causes the basic amine product to migrate into the aqueous phase, while over 99% of the expensive palladium catalyst remains dissolved in the toluene layer. This physical separation allows for the direct recycling of the catalyst-containing organic phase for up to nine consecutive batches, a feature that drastically reduces the cost of goods sold. Furthermore, the use of excess 5-bromo-3-alkoxypyridine prevents the precipitation of Pd(0), maintaining the catalyst in a stable Pd(II) state and ensuring consistent reaction kinetics throughout the campaign. This holistic optimization elevates the overall yield of the initial steps from 44% to approximately 50-55%, demonstrating a clear path toward commercial viability.
Mechanistic Insights into Cu/LiCl-Assisted Grignard and Pd-Catalyzed Coupling
The mechanistic superiority of this process begins with the copper-catalyzed Grignard addition. The inclusion of lithium chloride is not merely incidental; it plays a crucial role in modifying the aggregation state of the organocuprate species formed in situ. The LiCl likely coordinates with the magnesium and copper centers, increasing the solubility of the active catalytic species and enhancing the nucleophilicity of the vinyl group towards the epoxide. This results in higher regioselectivity for the desired 4-penten-2-ol isomer, minimizing the formation of branched byproducts that are difficult to separate. The reduction of copper loading to catalytic levels (0.01 eq) implies a highly efficient turnover number, where the copper species is rapidly regenerated in the catalytic cycle rather than being consumed as a stoichiometric reagent. This efficiency is paramount for impurity control, as lower metal concentrations reduce the risk of copper-induced oxidative degradation of the sensitive alkene moiety during subsequent processing steps.
Regarding the palladium-catalyzed Heck coupling, the mechanism benefits significantly from the presence of water as an additive. Water facilitates the activation of the palladium precursor, likely aiding in the generation of the active Pd(0) species required for the oxidative addition into the aryl bromide bond. The choice of tris-o-tolylphosphine as a ligand provides the necessary steric bulk to stabilize the palladium center while allowing for efficient migratory insertion and beta-hydride elimination. Crucially, the biphasic nature of the reaction medium serves as an in-situ purification mechanism. As the reaction proceeds, the basic amine product is protonated by the added hydrochloric acid, forming a water-soluble ammonium salt. This ionization drives the equilibrium towards product formation and physically separates the organic product from the lipophilic palladium-phosphine complexes retained in the toluene phase. This elegant design minimizes the need for aggressive metal scavengers like silica-bound thiols, although they remain an option for polishing, thereby streamlining the downstream processing workflow.
How to Synthesize (2S)-(4E)-N-methyl-5-[3-(5-isopropoxypyridinyl)]-4-penten-2-amine Efficiently
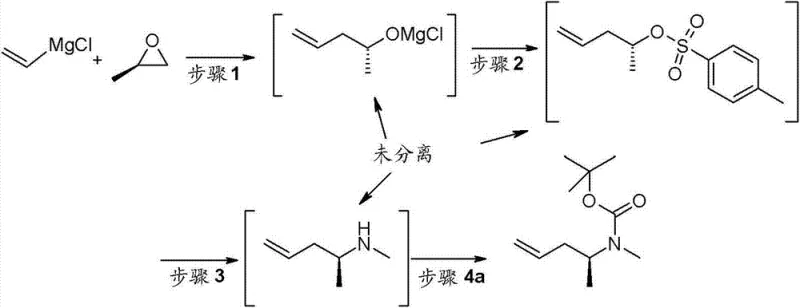
The synthesis of this high-value intermediate requires strict adherence to the optimized parameters regarding temperature, stoichiometry, and phase separation to ensure maximum yield and purity. The process begins with the low-temperature addition of vinylmagnesium chloride to the Cu/LiCl catalyst mixture, followed by the controlled addition of propylene oxide to manage exotherms. Following the formation of the tosylate and subsequent amination, the crude amine is protected or converted to a salt to facilitate handling. The final coupling step demands precise control of the water content and pH during the workup to ensure effective phase separation. For detailed operational protocols, including specific addition rates and quenching procedures, refer to the standardized guide below.
- Prepare 4-penten-2-ol via vinylmagnesium chloride addition to propylene oxide using catalytic CuCl/LiCl.
- Convert the alcohol to tosylate and react with methylamine to form the amine intermediate.
- Perform Pd-catalyzed coupling with 5-bromo-3-alkoxypyridine in toluene/water followed by salt formation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel manufacturing process offers tangible strategic benefits beyond mere technical elegance. The most significant advantage is the drastic simplification of the supply chain for raw materials and the reduction of dependency on scarce resources. By lowering the copper requirement from stoichiometric to catalytic levels, the process reduces the procurement burden for heavy metal salts and minimizes the logistical challenges associated with hazardous waste disposal. The ability to recycle the palladium catalyst and toluene solvent for multiple batches creates a closed-loop system that insulates the manufacturing cost from fluctuations in precious metal markets. This stability is crucial for long-term contract negotiations and ensures a predictable cost structure for the final API, making cost reduction in pharmaceutical intermediate manufacturing a reality rather than just a goal.
- Cost Reduction in Manufacturing: The elimination of stoichiometric copper reagents and the reduction of reaction times from days to hours directly translate to lower utility and labor costs per kilogram of product. The switch to cheaper, more recyclable solvents like toluene and MTBE, replacing expensive and difficult-to-remove solvents like DMF, further decreases the operational expenditure. Additionally, the high conversion rates and improved yields mean less raw material is wasted, maximizing the atom economy of the entire synthetic sequence. These cumulative efficiencies allow for a substantial reduction in the cost of goods without compromising quality standards.
- Enhanced Supply Chain Reliability: The robustness of the catalytic system ensures consistent batch-to-batch quality, reducing the risk of production delays caused by failed runs or out-of-specification results. The simplified workup procedures, which avoid complex filtrations and distillations of thermally sensitive oils, decrease the turnaround time between batches. This increased throughput capacity enables manufacturers to respond more agilely to market demand spikes, ensuring a continuous supply of critical intermediates for downstream drug formulation. The use of commercially available and stable reagents further mitigates the risk of supply disruptions.
- Scalability and Environmental Compliance: The process has been validated in reactors with volumes up to 4 cubic meters, demonstrating its readiness for commercial scale-up of complex pharmaceutical intermediates. The significant reduction in heavy metal waste and the avoidance of chlorinated solvents align with increasingly stringent global environmental regulations. This green chemistry approach not only reduces the environmental footprint but also simplifies the regulatory filing process by presenting a cleaner impurity profile. The ability to effectively remove palladium residues through phase separation ensures that the final product meets strict limits for residual metals, a critical requirement for regulatory approval.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced synthesis route. These insights are derived directly from the experimental data and process descriptions within the patent documentation, providing clarity on the practical aspects of adoption. Understanding these details is essential for technical teams evaluating the feasibility of integrating this methodology into existing production lines.
Q: How does the new process improve upon conventional copper-mediated Grignard reactions?
A: The novel method reduces copper usage from stoichiometric 0.15 molar equivalents to catalytic 0.01 molar equivalents by utilizing a CuCl/LiCl mixture. This significantly lowers heavy metal waste, eliminates complex filtration steps, and improves overall environmental compliance.
Q: What strategy is employed for palladium removal in the final coupling step?
A: The process utilizes a biphasic toluene-water system where over 99% of the palladium catalyst remains in the organic toluene phase while the product partitions into the acidic aqueous phase. This simple phase separation drastically reduces residual metal levels without needing extensive scavenging.
Q: Is this synthesis route suitable for large-scale commercial production?
A: Yes, the patent explicitly demonstrates scalability in reactors ranging from 2.5 cubic meters to 4 cubic meters. Furthermore, the palladium catalyst and toluene solvent can be recycled for up to nine consecutive batches without loss of yield, ensuring robust supply chain continuity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aryl Substituted Olefinic Amines Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthesis routes for CNS drug development. Our team of expert process chemists has extensively analyzed the innovations presented in CN103180277A and possesses the technical capability to implement these optimized pathways immediately. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from pilot plant to full-scale manufacturing. Our facilities are equipped with rigorous QC labs and stringent purity specifications to guarantee that every batch of aryl substituted olefinic amines meets the highest international standards for pharmaceutical intermediates.
We invite you to collaborate with us to leverage these process improvements for your specific drug development programs. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your supply chain is built on a foundation of scientific excellence and commercial reliability. Let us help you accelerate your timeline to market with a supply partner dedicated to innovation and quality.
