Advanced Solvent-Free Synthesis of N-(2,6-Xylyl) Methyl Aminopropionate for High-Efficiency Fungicide Production
The global demand for high-efficiency, low-toxicity systemic bactericides continues to drive innovation in the agrochemical intermediate sector, specifically for phenylamide compounds used in combating downy mildew and related plant diseases. Patent CN111747859A introduces a groundbreaking method for synthesizing N-(2,6-xylyl) methyl aminopropionate, a pivotal chiral intermediate that forms the active skeleton of these advanced fungicides. Unlike traditional routes that struggle with solvent recovery and thermal instability, this novel approach leverages a solvent-free coupling reaction followed by precision molecular distillation. For R&D directors and procurement specialists seeking a reliable agrochemical intermediate supplier, this technology represents a significant leap forward in process intensification. By operating without added solvents during the critical coupling phase and utilizing high-vacuum purification, the process achieves yields ranging from 95.0% to 97.0%, drastically outperforming conventional methods that often plateau around 82%. This technical breakthrough not only ensures a consistent supply of high-purity materials but also aligns with increasingly stringent environmental regulations regarding volatile organic compound (VOC) emissions in chemical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of N-(2,6-xylyl) methyl aminopropionate has been plagued by significant operational inefficiencies and environmental burdens that hinder scalable production. Conventional pathways typically rely on alpha-chloropropionic acid as a starting material, necessitating a multi-step reaction sequence that inherently accumulates impurities and lowers overall throughput. Alternatively, methods utilizing 2,6-dimethylaniline and 2-methyl chloropropionate in amide (AM) solvents suffer from severe drawbacks, including substantial solvent pollution and difficult recovery processes. These traditional routes often require high reaction temperatures that promote side reactions, leading to a product yield of only approximately 82% and purity levels that barely meet the 95% threshold. Furthermore, the reliance on large volumes of organic solvents increases the fire hazard potential and necessitates expensive wastewater treatment infrastructure to handle solvent-laden effluents. For supply chain managers, these factors translate into unpredictable lead times and inflated logistics costs associated with hazardous material handling, making the conventional supply chain fragile and economically unsustainable in the long term.
The Novel Approach
In stark contrast, the methodology disclosed in CN111747859A revolutionizes the production landscape by eliminating the solvent requirement during the primary synthetic reaction between 2,6-dimethylaniline and methyl p-toluenesulfonate propionate. This solvent-free strategy not only mitigates the risk of solvent-related contamination but also simplifies the downstream processing workflow by removing the need for complex solvent recovery units. The reaction is conducted at a moderate temperature range of 80-85°C, which is significantly lower than many traditional high-energy processes, thereby preserving the integrity of the chiral center and minimizing thermal degradation. Following the reaction, the process employs a sophisticated alkaline hydrolysis and extraction protocol using toluene and sodium bicarbonate to efficiently separate the desired product from p-toluenesulfonic acid byproducts. 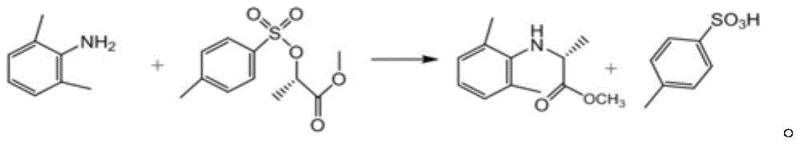 This streamlined approach ensures that the crude product enters the purification stage with a much higher initial quality, setting the stage for the final high-yield refinement via molecular distillation.
This streamlined approach ensures that the crude product enters the purification stage with a much higher initial quality, setting the stage for the final high-yield refinement via molecular distillation.
Mechanistic Insights into Solvent-Free Nucleophilic Substitution
The core of this innovative synthesis lies in the direct nucleophilic substitution reaction between the amine group of 2,6-dimethylaniline and the activated ester group of methyl p-toluenesulfonate propionate. In the absence of a solvent, the reactant molecules are in closer proximity, which can enhance the collision frequency and reaction rate, provided that mixing efficiency is maintained through rigorous stirring at speeds between 550 rpm and 850 rpm. The methyl p-toluenesulfonate group acts as an excellent leaving group, facilitating the formation of the new carbon-nitrogen bond while generating p-toluenesulfonic acid as a stoichiometric byproduct. This mechanism is particularly advantageous for preserving the stereochemistry of the chiral carbon atom, which is critical because the R-(-)-isomer possesses superior bactericidal activity compared to its S-(+)-counterpart. By avoiding protic or highly polar solvents that might interfere with the transition state or promote racemization, the solvent-free condition helps maintain the optical purity of the intermediate, a key quality metric for downstream fungicide efficacy.
Impurity control is further enhanced during the workup phase, where the reaction mixture is subjected to alkaline hydrolysis using a 30% sodium bicarbonate aqueous solution. This step serves a dual purpose: it neutralizes the acidic p-toluenesulfonic acid byproduct, converting it into water-soluble sodium p-toluenesulfonate, and simultaneously facilitates the evolution of carbon dioxide gas which aids in mixing. The subsequent liquid-liquid extraction with toluene selectively partitions the organic product into the organic phase while leaving inorganic salts and water-soluble impurities in the aqueous layer. This robust separation mechanism ensures that the crude material fed into the distillation unit is free from corrosive acids and ionic contaminants, protecting the sensitive molecular distillation equipment and ensuring the final product meets stringent purity specifications required by top-tier agrochemical manufacturers.
How to Synthesize N-(2,6-Xylyl) Methyl Aminopropionate Efficiently
To implement this high-yield synthesis route effectively, manufacturers must adhere to precise operational parameters regarding temperature control, vacuum levels, and feed ratios as detailed in the patent examples. The process begins with the careful metering of high-purity 2,6-dimethylaniline (98.9-99.2%) and methyl p-toluenesulfonate propionate (98.0-98.5%) into a heated reactor, followed by a sustained reaction period of 20 to 36 hours. Adhering to these standardized steps ensures reproducibility and maximizes the economic potential of the solvent-free methodology, allowing production teams to consistently achieve yields exceeding 95% while minimizing waste generation.
- Conduct a solvent-free synthetic reaction between 2,6-dimethylaniline and methyl p-toluenesulfonate propionate at 80-85°C for 20-36 hours.
- Perform separation and crude extraction using alkaline hydrolysis with sodium bicarbonate and toluene extraction to remove byproducts.
- Execute distillation and refining using a two-stage molecular short-path rectifying still under high vacuum (80-100 Pa) to isolate the pure product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this solvent-free technology offers transformative benefits that extend far beyond simple yield improvements. The elimination of solvents in the main reaction step fundamentally alters the cost structure of manufacturing by removing the capital and operational expenditures associated with solvent storage, recycling, and disposal. This reduction in process complexity directly translates to a more resilient supply chain, as the production timeline is no longer bottlenecked by solvent recovery rates or wastewater treatment capacity. Furthermore, the use of molecular distillation allows for the processing of heat-sensitive materials with minimal degradation, ensuring that the final product retains its high value and efficacy, which is crucial for maintaining long-term contracts with pharmaceutical and agrochemical clients who demand consistent quality.
- Cost Reduction in Manufacturing: The most significant economic driver of this process is the complete removal of solvents during the coupling reaction, which eradicates the costs associated with purchasing, storing, and recovering large volumes of organic liquids. By operating under solvent-free conditions, the facility reduces its energy footprint significantly, as there is no need to heat or distill off massive quantities of solvent mass, leading to substantial utility savings. Additionally, the high conversion efficiency minimizes the loss of expensive raw materials like 2,6-dimethylaniline, ensuring that every kilogram of input contributes maximally to the final output. This lean manufacturing approach results in a drastically simplified cost model, making the final intermediate more price-competitive in the global market without compromising on quality standards.
- Enhanced Supply Chain Reliability: The robustness of the solvent-free reaction coupled with the efficiency of molecular distillation creates a production workflow that is less prone to interruptions and delays. Traditional methods often face bottlenecks during solvent recovery or wastewater treatment, which can halt production lines; this new method bypasses those vulnerabilities entirely. The ability to run the reaction at moderate temperatures (80-85°C) also reduces the wear and tear on reactor vessels and heating systems, leading to lower maintenance downtime and higher asset utilization rates. For supply chain planners, this means more predictable lead times and the ability to scale production volumes rapidly to meet fluctuating market demands for agrochemical intermediates without the risk of process failure.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this process is inherently safer and more sustainable, aligning perfectly with modern green chemistry principles. The absence of solvent vapors reduces the risk of workplace exposure and fire hazards, creating a safer environment for operators and lowering insurance premiums. Moreover, the generation of wastewater is significantly reduced because the aqueous workup is highly efficient and the volume of organic waste is minimized. This makes the process easier to permit and operate in regions with strict environmental regulations, ensuring long-term operational continuity. The scalability is further supported by the use of continuous or semi-continuous molecular distillation units, which can be easily expanded to meet commercial tonnage requirements.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced synthesis technology, providing clarity for stakeholders evaluating its adoption. These insights are derived directly from the experimental data and process descriptions found in the patent literature, ensuring accuracy and relevance for technical decision-makers.
Q: What are the primary advantages of the solvent-free method described in CN111747859A?
A: The solvent-free method eliminates the need for organic solvents during the coupling reaction, significantly reducing solvent pollution, energy consumption for recovery, and overall production costs while maintaining high reaction yields between 95.0% and 97.0%.
Q: How does molecular distillation improve the purity of the final intermediate?
A: Molecular short-path rectification allows for separation at lower temperatures (110-118°C) under high vacuum (80-100 Pa), preventing thermal degradation of the chiral amino acid ester and ensuring the removal of residual 2,6-dimethylaniline for superior purity.
Q: Why is this intermediate critical for phenylamide fungicide production?
A: N-(2,6-xylyl) methyl aminopropionate serves as the key chiral skeleton for phenylamide fungicides, where the R-(-)-isomer exhibits significantly higher bactericidal activity against downy mildew and other plant diseases compared to the S-(+)-isomer.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-(2,6-Xylyl) Methyl Aminopropionate Supplier
At NINGBO INNO PHARMCHEM, we understand that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with unwavering consistency. Our facilities are equipped with state-of-the-art molecular distillation units and rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of N-(2,6-xylyl) methyl aminopropionate meets the highest industry standards. We are committed to delivering high-purity agrochemical intermediates that empower your R&D teams to develop next-generation fungicides with confidence.
We invite you to collaborate with us to leverage this cutting-edge solvent-free technology for your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized manufacturing processes can drive value and efficiency for your organization.
