Scalable Synthesis of 1-Phenyl-1234-Tetrahydroisoquinoline for Commercial Pharmaceutical Production
The pharmaceutical industry continuously seeks robust synthetic routes for critical intermediates like 1-phenyl-1,2,3,4-tetrahydroisoquinoline, a key building block for Solifenacin. Patent CN103159677A introduces a transformative preparation method that addresses longstanding inefficiencies in traditional manufacturing protocols. This innovation focuses on optimizing the initial amide formation step by utilizing an aqueous phase reaction system, which fundamentally alters the downstream processing requirements. By eliminating the need for organic solvents in the first stage, the process significantly reduces operational complexity and environmental burden. The technical breakthrough lies in the strategic selection of reagents that allow the product to precipitate directly from the aqueous solution, thereby simplifying isolation. This approach not only enhances the overall yield but also aligns with modern green chemistry principles demanded by regulatory bodies. For R&D directors and procurement managers, understanding this patent is crucial for evaluating supply chain resilience and cost structures in the production of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for 1-phenyl-1,2,3,4-tetrahydroisoquinoline often rely heavily on organic solvents during the initial amide coupling reaction between phenethylamine and benzoyl chloride. These conventional methods typically necessitate extensive post-reaction workup procedures including separatory funnel extraction, acid washing, base washing, and drying steps to isolate the intermediate amide. Such multi-step purification processes are not only labor-intensive but also introduce significant opportunities for product loss, thereby reducing the overall reaction yield. Furthermore, the extensive use of volatile organic compounds poses serious environmental hazards and increases the cost of waste disposal and solvent recovery. The reliance on organic media also complicates the scale-up process due to safety concerns regarding flammability and toxicity. Consequently, manufacturers face higher operational costs and longer lead times when adhering to these legacy synthetic protocols.
The Novel Approach
The novel approach described in the patent data revolutionizes the synthesis by conducting the initial amide formation in an aqueous environment using alkali metal hydroxide as a base. This strategic shift allows the resulting N-(2-phenethyl) benzamide to precipitate directly from the water due to its insolubility, enabling simple filtration instead of complex extraction. By removing organic solvents from this critical first step, the process drastically simplifies the after-treatment operation and reduces the consumption of hazardous chemicals. This modification leads to substantial cost savings associated with solvent procurement and waste management while improving the safety profile of the manufacturing facility. Additionally, the subsequent cyclization step utilizes phosphorus pentoxide and phosphorus oxychloride instead of polyphosphoric acid, avoiding the generation of toxic phosphorus oxide exhaust gas. This comprehensive optimization ensures a more sustainable and economically viable pathway for producing reliable pharmaceutical intermediates supplier products.
Mechanistic Insights into P2O5-Mediated Cyclization
The core of this synthetic strategy involves a sophisticated cyclization mechanism driven by the synergistic action of phosphorus pentoxide and phosphorus oxychloride in a benzene-type solvent. This reagent combination facilitates the dehydration and ring-closure of the amide intermediate to form 1-phenyl-3,4-dihydro-isoquinoline with high efficiency. The reaction proceeds under reflux conditions at temperatures between 107°C and 110°C, ensuring complete conversion of the starting material while minimizing side reactions. The use of phosphorus oxychloride acts as a chlorinating agent that activates the amide carbonyl, making it susceptible to intramolecular electrophilic aromatic substitution. This mechanism is superior to traditional polyphosphoric acid methods because it prevents the thermal decomposition that leads to hazardous emissions. The careful control of stoichiometry, with a molar ratio of amide to phosphorus pentoxide to phosphorus chloride around 1:1.1:2.5, ensures optimal reaction kinetics and product quality.
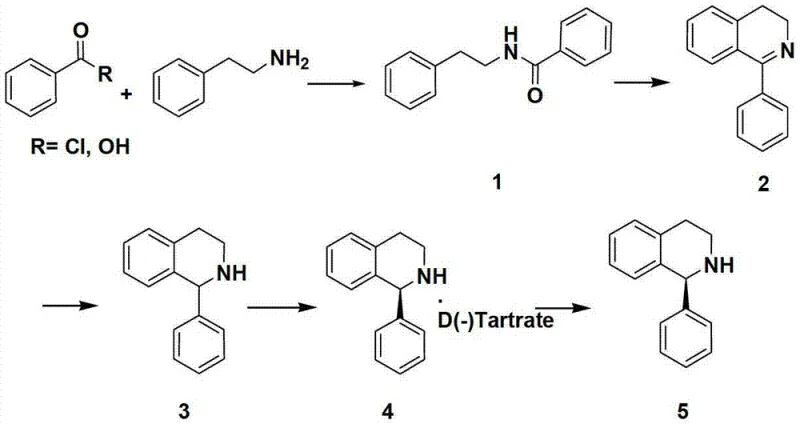
Impurity control is meticulously managed throughout the synthesis to ensure the final product meets stringent purity specifications required for pharmaceutical applications. The aqueous workup following the cyclization step involves careful pH adjustment using alkali metal hydroxide solutions to neutralize acidic byproducts and facilitate phase separation. Extraction with ethyl acetate allows for the selective isolation of the dihydro-isoquinoline intermediate while leaving inorganic salts in the aqueous phase. The subsequent reduction step using borohydride reagents is conducted at controlled temperatures to prevent over-reduction or degradation of the sensitive heterocyclic ring system. Crystallization from alcohol solvents further purifies the product by excluding structurally similar impurities that might co-elute during chromatography. This multi-layered purification strategy guarantees that the final 1-phenyl-1,2,3,4-tetrahydroisoquinoline achieves purity levels greater than 98%, satisfying the rigorous demands of high-purity pharmaceutical intermediates manufacturing.
How to Synthesize 1-Phenyl-1234-Tetrahydroisoquinoline Efficiently
Implementing this synthesis route requires precise adherence to the patented conditions to maximize yield and safety during commercial scale-up of complex pharmaceutical intermediates. The process begins with the careful mixing of phenethylamine and alkali hydroxide in water, followed by the controlled addition of benzoyl chloride under ice bath cooling to manage exothermic heat. Once the amide intermediate is isolated via filtration, it is dissolved in toluene and subjected to cyclization conditions with phosphorus reagents under reflux. The final reduction step utilizes sodium borohydride in methanol to convert the dihydro-intermediate into the target tetrahydroisoquinoline structure. Detailed standardized synthesis steps see the guide below for specific operational parameters and safety precautions.
- Perform aqueous amide formation using phenethylamine and benzoyl chloride with alkali hydroxide to precipitate N-(2-phenethyl) benzamide without organic solvents.
- Execute cyclization using phosphorus pentoxide and phosphorus oxychloride in benzene solvent under reflux to form 1-phenyl-34-dihydro-isoquinoline.
- Conclude with borohydride reduction in alcohol solvent to yield the final 1-phenyl-1234-tetrahydroisoquinoline product with high purity.
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthesis pathway offers profound commercial benefits for procurement managers and supply chain heads focused on cost reduction in pharmaceutical intermediates manufacturing. By eliminating organic solvents in the first step, the process removes the need for expensive solvent recovery systems and reduces the volume of hazardous waste requiring disposal. The simplified workup procedure translates directly into reduced labor hours and faster batch turnover times, enhancing overall production efficiency. Furthermore, the avoidance of toxic exhaust gases during cyclization lowers the regulatory burden and insurance costs associated with environmental compliance. These factors combine to create a more resilient supply chain capable of meeting demanding delivery schedules without compromising on quality or safety standards.
- Cost Reduction in Manufacturing: The elimination of organic solvents in the initial amide formation step removes significant expenses related to solvent procurement, storage, and recovery infrastructure. Without the need for complex extraction and washing procedures, the consumption of auxiliary chemicals is drastically reduced, leading to lower variable costs per kilogram of product. The simplified isolation method via filtration rather than separation reduces energy consumption associated with distillation and drying processes. Additionally, the higher overall yield minimizes the cost of raw materials wasted due to side reactions or processing losses. These cumulative effects result in substantial cost savings that can be passed on to customers or reinvested into further process optimization.
- Enhanced Supply Chain Reliability: The use of readily available starting materials such as phenethylamine and benzoyl chloride ensures a stable supply of raw inputs without reliance on exotic or scarce reagents. The robust nature of the aqueous reaction conditions makes the process less sensitive to minor fluctuations in temperature or mixing efficiency, ensuring consistent batch-to-batch quality. Simplified processing steps reduce the risk of operational delays caused by equipment failures or complex purification bottlenecks. This reliability allows for more accurate forecasting of production timelines and reduces the lead time for high-purity pharmaceutical intermediates delivery to global clients. Consequently, partners can maintain leaner inventory levels while ensuring continuity of supply for critical drug manufacturing programs.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing standard reactor equipment and common solvents that are easily sourced for large-scale production. The avoidance of toxic phosphorus oxide exhaust gas simplifies the engineering controls required for ventilation and scrubbing systems, facilitating easier permitting and regulatory approval. Reduced waste generation aligns with increasingly strict environmental regulations, minimizing the risk of fines or shutdowns due to non-compliance. The ability to scale from laboratory to commercial production without significant process re-engineering ensures a smooth transition for new product introductions. This environmental and operational flexibility supports long-term sustainability goals while maintaining competitive manufacturing capabilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and supply of this critical pharmaceutical intermediate based on the patented technology. Understanding these details helps stakeholders make informed decisions about integrating this route into their supply chain strategies. The answers are derived directly from the technical specifications and beneficial effects outlined in the patent documentation to ensure accuracy and relevance.
Q: How does this method reduce environmental impact compared to conventional synthesis?
A: The initial amide formation occurs in water without organic solvents, eliminating complex extraction steps and reducing hazardous waste generation significantly.
Q: What safety advantages does the cyclization step offer over polyphosphoric acid methods?
A: Using phosphorus pentoxide and phosphorus oxychloride avoids the decomposition of polyphosphoric acid which generates toxic phosphorus oxide exhaust gas.
Q: Is this process suitable for large-scale commercial manufacturing of Solifenacin intermediates?
A: Yes, the simplified workup and high yields across steps make it highly scalable for commercial production of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-Phenyl-1234-Tetrahydroisoquinoline Supplier
NINGBO INNO PHARMCHEM stands ready to support your pharmaceutical development needs with extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our technical team possesses deep expertise in optimizing complex synthetic routes to meet stringent purity specifications and rigorous QC labs standards. We understand the critical importance of supply continuity and cost efficiency in the global pharmaceutical market. By leveraging advanced manufacturing technologies and robust quality management systems, we ensure that every batch of 1-phenyl-1,2,3,4-tetrahydroisoquinoline meets the highest industry benchmarks. Our commitment to innovation allows us to adapt quickly to changing market demands while maintaining the highest levels of safety and environmental stewardship.
We invite you to contact our technical procurement team to request specific COA data and route feasibility assessments for your projects. Our experts are available to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. By partnering with us, you gain access to a reliable supply chain partner dedicated to your success. Let us help you optimize your manufacturing process and secure a competitive advantage in the marketplace through superior chemical solutions and dedicated support services.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
