Advanced Synthesis of 4-(1-H Indole) Phenol Derivatives for Next-Generation Antibacterial Drug Development
The escalating crisis of bacterial resistance represents one of the most formidable challenges facing the modern pharmaceutical industry, necessitating the urgent discovery of novel antimicrobial scaffolds with distinct mechanisms of action. Patent CN110054579B introduces a groundbreaking class of 4-(1-H indole) phenol derivatives that demonstrate potent inhibitory activity against multidrug-resistant pathogens, including Methicillin-Resistant Staphylococcus aureus (MRSA). This technological breakthrough offers a vital new avenue for R&D teams seeking to diversify their antibiotic pipelines with high-purity pharmaceutical intermediates that possess proven biological efficacy. The structural novelty of these compounds, characterized by a specific linkage between an indole moiety and a phenol ring, provides a unique chemical space for further medicinal chemistry optimization. As a reliable pharmaceutical intermediate supplier, understanding the synthesis and potential of these molecules is crucial for securing a competitive edge in the development of next-generation anti-infective therapies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the construction of aryl-substituted indole scaffolds has relied heavily on transition-metal catalyzed cross-coupling reactions, such as Suzuki-Miyaura or Buchwald-Hartwig couplings, which require pre-functionalized starting materials. These conventional pathways often necessitate the use of halogenated indoles or aryl boronic acids, which can be significantly more expensive and less readily available than their non-functionalized counterparts. Furthermore, these methods frequently generate stoichiometric amounts of salt waste and require rigorous purification steps to remove residual halides and metal contaminants that could compromise the safety profile of the final drug substance. The reliance on sensitive organometallic reagents also imposes strict handling requirements, such as moisture-free environments, which can complicate process engineering and increase operational expenditures in a manufacturing setting. Consequently, the search for more direct and atom-economical routes has become a priority for cost reduction in antibacterial drug manufacturing.
The Novel Approach
The methodology disclosed in the patent data presents a paradigm shift by employing a direct oxidative coupling strategy between substituted indoles and 1,4-benzoquinone mediated by a palladium catalyst. This innovative approach bypasses the need for pre-halogenation or borylation, utilizing simple, commodity-grade indoles and benzoquinone as the primary building blocks. The reaction proceeds efficiently in dimethyl sulfoxide (DMSO) at moderate temperatures ranging from 40°C to 120°C, offering a robust and operationally simple protocol that is highly amenable to industrial scale-up. By streamlining the synthetic sequence and reducing the number of unit operations, this novel approach significantly enhances the overall process efficiency and reduces the environmental footprint associated with waste disposal. This strategic advancement aligns perfectly with the goals of commercial scale-up of complex pharmaceutical intermediates, providing a sustainable pathway for producing high-value antibacterial agents.
Mechanistic Insights into Pd-Catalyzed Oxidative Coupling
The core of this synthetic transformation lies in the palladium-catalyzed C-H activation and subsequent oxidative coupling, a sophisticated mechanism that allows for the direct functionalization of the indole ring at the C3 position. The catalytic cycle likely initiates with the coordination of the palladium species to the indole nitrogen or the pi-system, facilitating the cleavage of the C-H bond and the formation of a reactive organopalladium intermediate. This intermediate then undergoes transmetallation or insertion with the benzoquinone, followed by a beta-hydride elimination or reductive elimination step to forge the new carbon-carbon bond. The use of oxygen or the quinone itself as a terminal oxidant regenerates the active palladium species, ensuring the catalytic turnover continues efficiently throughout the reaction duration. Understanding these mechanistic nuances is essential for R&D directors aiming to optimize reaction parameters and ensure consistent batch-to-batch quality.
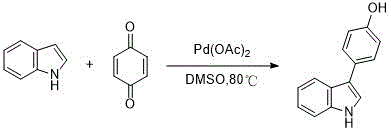
Impurity control is a critical aspect of this process, particularly given the potential for over-oxidation or polymerization of the reactive quinone species under thermal conditions. The patent specifies the use of column chromatography with silica gel and specific eluent systems, such as ethyl acetate and petroleum ether, to achieve high purity levels required for pharmaceutical applications. The choice of solvent, DMSO, plays a dual role in stabilizing the polar transition states and solubilizing the reactants, which helps in minimizing side reactions that could lead to difficult-to-remove impurities. Furthermore, the ability to tune the reaction temperature between 40°C and 120°C allows for precise control over the reaction kinetics, enabling the suppression of thermal degradation pathways. This level of control ensures that the final 4-(1-H indole) phenol derivatives meet the stringent purity specifications demanded by regulatory bodies for clinical use.
How to Synthesize 4-(1-H Indole) Phenol Derivatives Efficiently
The synthesis of these valuable antibacterial intermediates follows a straightforward yet highly effective protocol that balances yield and purity. The process begins with the dissolution of the selected substituted indole and 1,4-benzoquinone in DMSO, maintaining a molar ratio that favors the formation of the desired coupled product while minimizing excess reagent waste. A palladium catalyst, such as palladium acetate, is introduced to initiate the coupling, and the mixture is heated under an inert atmosphere to prevent unwanted oxidation of the sensitive indole core. Detailed standardized synthesis steps are provided in the guide below to ensure reproducibility and safety during laboratory and pilot-scale operations.
- Dissolve substituted indole and 1,4-benzoquinone in dimethyl sulfoxide (DMSO) with a molar ratio ranging from 1: 1 to 1:6.
- Add a palladium catalyst such as palladium acetate or palladium chloride and heat the mixture to 40-120°C under inert gas protection.
- After 12-24 hours, quench the reaction, extract with ethyl acetate, and purify the crude product via column chromatography using silica gel.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, this synthetic route offers substantial strategic benefits by leveraging widely available and cost-effective raw materials. The elimination of specialized halogenated precursors means that purchasing managers can source starting materials from a broader base of suppliers, reducing dependency on single-source vendors and mitigating supply chain risks. The simplified workflow also translates to reduced processing time and lower energy consumption, as the reaction does not require extreme cryogenic conditions or prolonged heating cycles typical of traditional cross-coupling methods. These factors collectively contribute to a more resilient and cost-efficient supply chain for high-purity antibacterial agents, allowing companies to maintain competitive pricing without compromising on quality standards.
- Cost Reduction in Manufacturing: The direct use of unsubstituted or simply substituted indoles avoids the premium costs associated with pre-functionalized building blocks like boronic acids or halides. By removing the need for separate halogenation or borylation steps, the overall material cost is significantly lowered, and the consumption of auxiliary reagents is drastically reduced. This streamlined approach minimizes waste generation and lowers the burden on waste treatment facilities, resulting in comprehensive operational savings that enhance the overall profitability of the manufacturing process.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as benzoquinone and basic indole derivatives ensures a stable and continuous supply of raw materials, even during market fluctuations. Since these materials are produced on a large scale for various industrial applications, the risk of shortages is minimal, guaranteeing consistent production schedules. This reliability is crucial for reducing lead time for high-purity antibacterial agents, enabling faster response to market demands and ensuring uninterrupted availability of critical drug intermediates for downstream formulation.
- Scalability and Environmental Compliance: The reaction conditions are mild and utilize common solvents, making the transition from laboratory bench to commercial production seamless and predictable. The process generates fewer hazardous byproducts compared to traditional methods, aligning with increasingly strict environmental regulations and sustainability goals. This ease of scale-up ensures that production volumes can be rapidly increased to meet global demand without the need for extensive re-engineering of the manufacturing infrastructure, supporting long-term business growth.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these 4-(1-H indole) phenol derivatives. The answers are derived directly from the patented technology and practical manufacturing experience, providing clarity on process feasibility and performance characteristics. Understanding these details helps stakeholders make informed decisions about integrating this technology into their existing development pipelines.
Q: What is the primary advantage of this Pd-catalyzed method over traditional cross-coupling?
A: This method utilizes direct oxidative coupling with benzoquinone, eliminating the need for pre-functionalized halogenated aromatics or boronic acids, thereby simplifying the raw material supply chain and reducing precursor costs.
Q: What is the antibacterial spectrum of these 4-(1-H indole) phenol derivatives?
A: The derivatives exhibit significant inhibitory activity against Methicillin-Resistant Staphylococcus aureus (MRSA), Staphylococcus aureus, and Bacillus subtilis, addressing critical gaps in current antibiotic efficacy.
Q: Is this synthesis route scalable for commercial production?
A: Yes, the reaction operates at moderate temperatures (40-120°C) using common solvents like DMSO and commercially available catalysts, making it highly suitable for scale-up from kilogram to multi-ton production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-(1-H Indole) Phenol Derivative Supplier
NINGBO INNO PHARMCHEM stands at the forefront of custom synthesis, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our technical team is adept at optimizing Pd-catalyzed processes to ensure stringent purity specifications and rigorous QC labs verify every batch against the highest international standards. We understand the critical nature of antibacterial drug development and are committed to providing a secure, high-quality supply of these advanced intermediates to support your clinical and commercial needs.
We invite you to engage with our technical procurement team to discuss a Customized Cost-Saving Analysis tailored to your specific project requirements. By collaborating with us, you can access specific COA data and route feasibility assessments that will accelerate your timeline to market. Let us help you overcome synthesis challenges and secure a sustainable supply chain for your next-generation antibiotic programs.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
