Revolutionizing TPO Photoinitiator Production with Low-Cost Sodium Chemistry
Revolutionizing TPO Photoinitiator Production with Low-Cost Sodium Chemistry
The global demand for high-performance photoinitiators continues to surge, driven by the rapid expansion of UV-curing technologies in coatings, inks, and advanced composite materials. At the forefront of this technological evolution is Patent CN110734461A, which discloses a groundbreaking synthesis process for 2,4,6-trimethylbenzoyldiphenylphosphine oxide, commonly known as TPO or Lucirin-TPO. This patent introduces a novel 'one-pot' methodology that fundamentally shifts the production paradigm from expensive, hazardous lithium-based chemistry to a robust, cost-effective sodium metal system. By leveraging commercially available diphenylphosphine chloride and metallic sodium, this innovation addresses critical pain points regarding raw material scarcity and environmental compliance that have long plagued the industry. For R&D directors and procurement strategists, understanding the nuances of this sodium-mediated pathway is essential for securing a competitive edge in the supply of high-purity photoinitiators. The following analysis dissects the technical merits and commercial implications of this transformative manufacturing route.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of acylphosphine oxide photoinitiators has relied heavily on organometallic routes involving highly reactive and costly reagents such as n-butyllithium or metallic lithium. These traditional methods, while effective, impose severe economic burdens due to the fluctuating prices and limited availability of lithium resources, which are derived from non-renewable mineral deposits. Furthermore, alternative pathways like the Arbuzov-Michaelis reaction, which utilizes diphenyl phosphites and acid chlorides, generate stoichiometric amounts of volatile chloroalkanes such as methyl chloride or ethyl chloride as by-products. These gaseous emissions are strictly regulated under international environmental protocols due to their ozone-depleting potential, forcing manufacturers to invest heavily in complex scrubbing and recovery systems to meet compliance standards. Additionally, the use of organic tertiary amines as acid-binding agents in these legacy processes introduces further complications regarding purification and waste treatment, often resulting in lower overall atom economy. The cumulative effect of these factors is a production process that is not only capital intensive but also fraught with regulatory risks and operational hazards related to pyrophoric reagents.
The Novel Approach
In stark contrast to these legacy technologies, the process disclosed in Patent CN110734461A utilizes a simple yet highly efficient sodium metal reduction strategy that operates effectively in a 'one-pot' configuration. By substituting expensive lithium with abundant and low-cost metallic sodium, the new method drastically reduces the direct material costs associated with the synthesis of the key phosphine intermediate. The reaction proceeds in ether solvents, preferably dioxane, where the sodium metal reacts with diphenylphosphine chloride to form a reactive sodium diphenylphosphide species in situ without the need for isolation. This intermediate then condenses directly with 2,4,6-trimethylbenzoyl chloride or its corresponding ester derivatives to form the trivalent phosphine precursor, which is subsequently oxidized to the final TPO product. Crucially, the only inorganic by-product generated throughout this entire sequence is sodium chloride, a benign salt that simplifies downstream processing and eliminates the generation of hazardous chloroalkane gases. This streamlined approach not only enhances the economic viability of TPO manufacturing but also aligns perfectly with modern green chemistry principles by minimizing waste and avoiding toxic reagents.
Mechanistic Insights into Sodium-Mediated Phosphine Formation
The success of this novel synthesis hinges on a precise understanding of the reaction kinetics and the formation of specific intermediate species during the reduction of diphenylphosphine chloride by sodium metal. Extensive research revealed that the reaction requires a distinct induction period to initiate efficiently, characterized by the formation of a bright orange-colored species upon the initial addition of a small portion of the phosphine chloride to the sodium suspension. This induction phase is critical; attempting to add the entire charge of diphenylphosphine chloride at once results in poor conversion and the formation of gray, unreactive mixtures, highlighting the sensitivity of the system to addition protocols. The orange intermediate likely represents a solvated electron complex or a specific organosodium cluster that facilitates the subsequent rapid reduction of the remaining phosphine chloride to the desired diphenylphosphide anion. Once this active species is established, the reaction proceeds smoothly under reflux conditions, typically between 80°C and 120°C, ensuring high yields of the trivalent phosphine precursor without the need for cryogenic temperatures often required for lithium analogs.
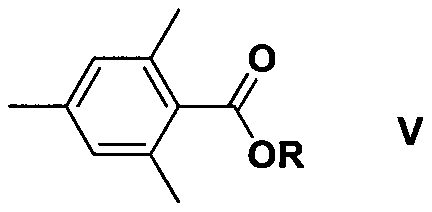
Furthermore, the mechanistic pathway allows for the direct condensation of the in-situ generated phosphide salt with acylating agents without the necessity of proton-donating compounds like tert-butanol, which were previously thought to be essential in sodium-based systems. The patent data indicates that various structural analogues, including structures I through IV shown below, may form as minor isomeric by-products depending on the specific reaction conditions and stoichiometry employed. 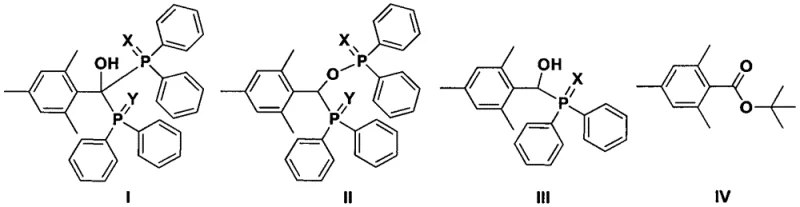 Understanding the formation and suppression of these side products, such as the hydroxy-phosphine oxides or ester derivatives, is vital for optimizing the purity profile of the final TPO product. The ability to control these pathways through temperature modulation and sequential addition strategies demonstrates a sophisticated level of process control that ensures the production of high-purity photoinitiators suitable for demanding applications in electronics and optical coatings. This deep mechanistic understanding provides a robust foundation for scaling the process from laboratory benchtop to multi-ton commercial production.
Understanding the formation and suppression of these side products, such as the hydroxy-phosphine oxides or ester derivatives, is vital for optimizing the purity profile of the final TPO product. The ability to control these pathways through temperature modulation and sequential addition strategies demonstrates a sophisticated level of process control that ensures the production of high-purity photoinitiators suitable for demanding applications in electronics and optical coatings. This deep mechanistic understanding provides a robust foundation for scaling the process from laboratory benchtop to multi-ton commercial production.
How to Synthesize 2,4,6-Trimethylbenzoyldiphenylphosphine Oxide Efficiently
Implementing this synthesis route requires careful attention to the preparation of the sodium suspension and the controlled addition of reagents to maintain the critical induction phase described in the mechanistic analysis. The process begins with the creation of a finely dispersed sodium sand in dry dioxane under reflux, followed by the dropwise addition of a fractional amount of diphenylphosphine chloride to trigger the formation of the active orange intermediate. Once the induction is complete, the remainder of the phosphine chloride is added, followed by the acylating agent, allowing the reaction to proceed to completion in a single vessel before the final oxidation step. This consolidated workflow minimizes unit operations and solvent usage, making it an ideal candidate for continuous manufacturing or large-batch processing. The detailed standardized synthetic steps, including specific molar ratios and workup procedures, are outlined in the guide below.
- Preparation of Sodium Suspension: Reflux metallic sodium in dry dioxane with high-shear stirring to create a fine suspension.
- Induction and Addition: Add a small portion of diphenylphosphine chloride to induce the formation of an orange intermediate species before adding the remainder.
- Condensation and Oxidation: React the intermediate with 2,4,6-trimethylbenzoyl chloride or ester, followed by oxidation with hydrogen peroxide to yield TPO.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this sodium-based synthesis route offers profound strategic advantages that extend far beyond simple raw material substitution. The elimination of metallic lithium from the supply chain removes exposure to the volatility of the lithium market, which is increasingly constrained by the demands of the electric vehicle battery sector, thereby stabilizing long-term input costs. Moreover, the removal of ozone-depleting chloroalkane by-products simplifies regulatory compliance and reduces the capital expenditure required for废气 treatment systems, directly contributing to a lower cost of goods sold. The simplified 'one-pot' nature of the reaction also reduces cycle times and labor requirements, enhancing overall plant throughput and asset utilization rates without compromising on product quality or safety standards.
- Cost Reduction in Manufacturing: The substitution of scarce metallic lithium with abundant metallic sodium fundamentally alters the economic landscape of TPO production by significantly lowering the cost of the primary reducing agent. By avoiding the use of expensive organic tertiary amines and complex phosphite esters required in alternative routes, the process achieves substantial savings in auxiliary material costs. The generation of benign sodium chloride as the sole inorganic by-product eliminates the need for costly waste disposal services associated with hazardous halogenated organic wastes. Furthermore, the high atom economy of the direct condensation step ensures that a greater proportion of raw materials are converted into valuable product, maximizing yield and minimizing waste-related expenses.
- Enhanced Supply Chain Reliability: Sourcing metallic sodium is inherently more stable and geographically diverse compared to the concentrated supply chains for battery-grade lithium, reducing the risk of raw material shortages. The use of common ether solvents like dioxane and standard acyl chlorides ensures that the majority of inputs are commodity chemicals available from multiple global suppliers, preventing single-source bottlenecks. This diversification of the supply base enhances resilience against geopolitical disruptions and logistics delays, ensuring consistent production schedules for downstream customers. Additionally, the robustness of the reaction conditions allows for flexible sourcing of raw material grades, further insulating the manufacturing process from minor supply chain fluctuations.
- Scalability and Environmental Compliance: The absence of pyrophoric organolithium reagents and gaseous chloroalkane emissions makes this process significantly safer and easier to scale to multi-ton capacities without extensive retrofitting of existing facilities. The simplified waste stream, consisting primarily of aqueous salt solutions and organic solvents, aligns with stringent environmental regulations and facilitates easier permitting for capacity expansions. This environmental friendliness serves as a strong value proposition for end-users in the coatings and electronics industries who are increasingly demanding sustainable and eco-friendly supply chains. The ability to produce high-purity TPO with a minimal environmental footprint positions manufacturers as preferred partners for green chemistry initiatives and corporate sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel sodium-based synthesis technology for TPO photoinitiators. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for evaluating the feasibility of this route. Understanding these details is crucial for technical teams assessing the transition from legacy lithium processes to this more economical alternative.
Q: Why is the sodium-based method superior to traditional lithium routes for TPO production?
A: The sodium-based method eliminates the reliance on expensive and scarce metallic lithium, significantly reducing raw material costs while avoiding the safety hazards associated with organolithium reagents.
Q: How does this process address environmental compliance issues regarding chloroalkanes?
A: Unlike the Arbuzov-Michaelis process which generates ozone-depleting chloromethane or chloroethane by-products, this novel route produces only sodium chloride as a by-product, ensuring full environmental compliance.
Q: What are the critical reaction conditions for achieving high yields in this one-pot synthesis?
A: Successful execution requires a specific induction period where a characteristic orange intermediate is formed, typically achieved by adding a fraction of the phosphine chloride first under reflux in dioxane.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,4,6-Trimethylbenzoyldiphenylphosphine Oxide Supplier
As the global market for UV-curing materials expands, the ability to deliver high-purity photoinitiators with consistent quality and competitive pricing becomes a decisive factor for success. NINGBO INNO PHARMCHEM leverages its extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to bring this advanced sodium-based technology to the marketplace. Our state-of-the-art facilities are equipped with rigorous QC labs and stringent purity specifications to ensure that every batch of TPO meets the exacting standards required for high-performance applications in TFT-LCD displays and optical fibers. We are committed to providing a reliable supply of this critical fine chemical intermediate, supporting our partners' growth with uninterrupted delivery and technical excellence.
We invite procurement leaders and R&D directors to collaborate with us to explore how this innovative synthesis route can optimize your supply chain and reduce manufacturing costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate the tangible benefits of partnering with a forward-thinking chemical manufacturer dedicated to innovation and sustainability.
