Optimizing Selinexor Production: A Novel Green Route for Pharmaceutical Intermediates
Optimizing Selinexor Production: A Novel Green Route for Pharmaceutical Intermediates
The pharmaceutical industry is constantly seeking more efficient and environmentally sustainable pathways for synthesizing complex oncology drugs. Patent CN112679477B introduces a significant breakthrough in the preparation of Selinexor, a potent exportin 1 (XPO1) inhibitor, and its critical intermediate, (Z)-3-(3-(3,5-bis(trifluoromethyl)phenyl)-1H-1,2,4-triazol-1-yl)acrylic acid. This patent addresses long-standing challenges in the existing synthetic routes, such as the generation of toxic gases, the use of hazardous solvents, and difficult purification processes. By leveraging a novel sequence of addition, substitution, and coupling reactions, this technology offers a robust framework for producing high-purity intermediates essential for the commercial manufacturing of Selinexor.
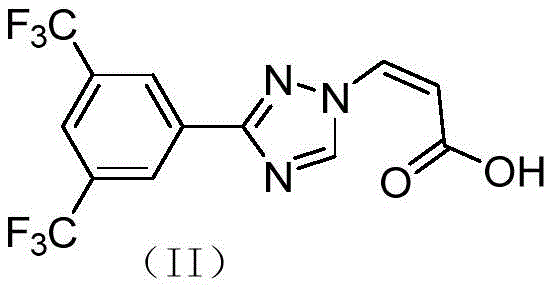
For R&D directors and process chemists, the structural integrity and purity of this intermediate are paramount. The patent details a method that not only ensures high chemical purity but also simplifies the isolation steps, making it an attractive candidate for technology transfer. As a reliable pharmaceutical intermediate supplier, understanding these mechanistic nuances allows us to offer superior quality control and consistency to our global partners.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Selinexor intermediates has been plagued by significant operational and environmental hurdles. Early synthetic routes, such as those disclosed in U.S. Pat. No. 5,907,9865, relied heavily on the use of sodium hydrosulfide to generate key triazole precursors. This step inevitably produces hydrogen sulfide gas, a highly toxic and malodorous byproduct that requires specialized scrubbing equipment and poses severe safety risks to plant personnel. Furthermore, these traditional methods often necessitate the use of N,N-dimethylformamide (DMF) as a primary solvent.
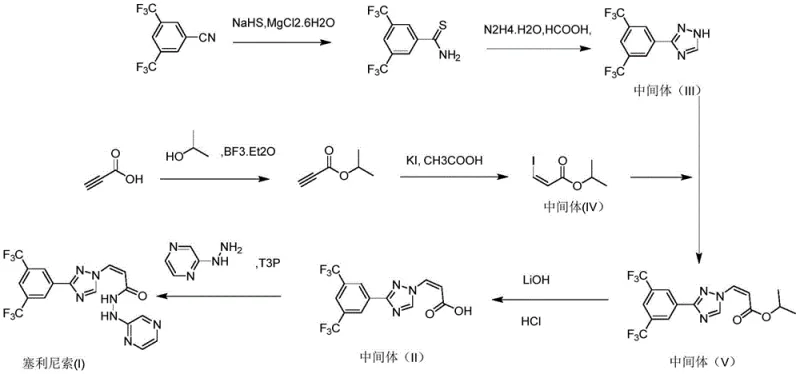
The reliance on DMF creates substantial downstream processing challenges, as it is difficult to remove completely and generates large volumes of wastewater that are costly to treat. Additionally, older routes frequently produce trans-isomer byproducts that require labor-intensive column chromatography for separation. This reliance on silica gel and large volumes of elution solvents not only increases production costs but also generates significant solid waste, making the process economically and environmentally unsustainable for large-scale commercial production.
The Novel Approach
The methodology outlined in patent CN112679477B represents a paradigm shift towards greener and more efficient chemistry. Instead of starting with unstable or hazardous reagents, this novel approach utilizes readily available propiolic acid esters and simple halide sources to construct the acrylic acid backbone. The core innovation lies in the strategic order of bond formation: first establishing the vinyl halide motif, then performing a nucleophilic substitution with the triazole ring, and finally installing the trifluoromethyl phenyl group via a palladium-catalyzed coupling reaction.
This rearrangement of synthetic steps effectively bypasses the generation of hydrogen sulfide gas entirely. Moreover, the process demonstrates a wide tolerance for safer solvents such as ethanol, isopropanol, and tetrahydrofuran, which are easier to recover and recycle compared to polar aprotic solvents like DMF. By eliminating the need for column chromatography and relying on crystallization for purification, this route significantly reduces the environmental footprint while enhancing the overall yield and purity of the final intermediate, making it particularly suitable for industrial production.
Mechanistic Insights into the Substitution and Coupling Strategy
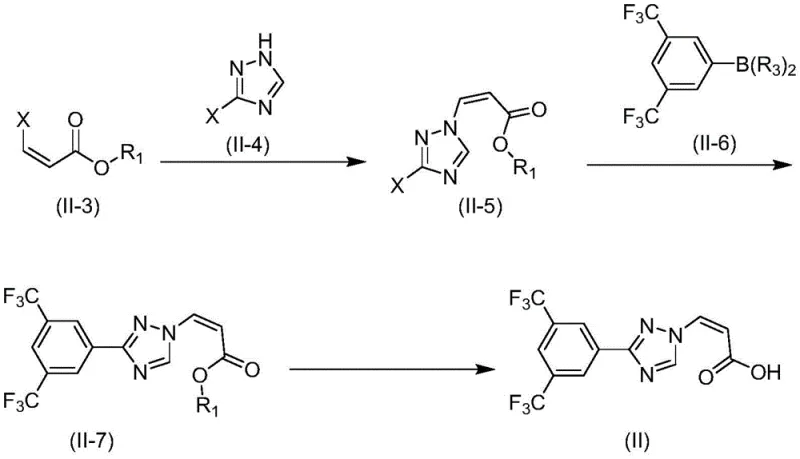
The success of this synthetic route hinges on two critical transformations: the nucleophilic substitution of the vinyl halide and the subsequent Suzuki-Miyaura cross-coupling. In the substitution step, a compound of formula (II-3), typically a (Z)-3-haloacrylate, reacts with a 3-halo-1H-1,2,4-triazole (formula II-4). The reaction is facilitated by a base, such as potassium carbonate or triethylamine, which deprotonates the triazole nitrogen, enabling it to attack the electron-deficient vinyl carbon. The stereochemistry is preserved during this SN2'-like displacement, ensuring the formation of the desired (Z)-isomer without significant isomerization to the thermodynamically more stable (E)-form.
Following the substitution, the resulting intermediate (II-5) undergoes a palladium-catalyzed cross-coupling with a boronic acid derivative (formula II-6). This step is crucial for introducing the 3,5-bis(trifluoromethyl)phenyl moiety, which is essential for the biological activity of the final drug. The use of catalysts like tetrakis(triphenylphosphine)palladium in a biphasic solvent system (e.g., toluene/water or THF/water) ensures efficient turnover. The presence of a base like sodium carbonate activates the boronic acid, forming a boronate species that transmetallates with the palladium complex. This mechanistic precision allows for high conversion rates and minimizes the formation of homocoupling byproducts, thereby simplifying the purification profile.
How to Synthesize (Z)-3-(3-(3,5-bis(trifluoromethyl)phenyl)-1H-1,2,4-triazol-1-yl)acrylic acid Efficiently
The synthesis of this high-value intermediate involves a logical progression of four distinct chemical transformations that can be executed in standard reactor setups. The process begins with the preparation of the vinyl halide precursor, followed by the assembly of the triazole core, the installation of the aryl group, and finally, the hydrolysis of the ester. Each step has been optimized to maximize yield and minimize impurity carryover. For a detailed breakdown of the specific reaction conditions, stoichiometry, and workup procedures required to replicate this process, please refer to the standardized guide below.
- Prepare compound (II-3) via addition reaction of propiolic acid ester (II-1) and halide source (II-2).
- React compound (II-3) with triazole derivative (II-4) under basic conditions to form intermediate (II-5).
- Perform Suzuki coupling between intermediate (II-5) and boronic acid (II-6) to install the trifluoromethyl phenyl group.
- Hydrolyze the ester group in compound (II-7) using alkali followed by acidification to yield the final acid intermediate (II).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic route offers tangible benefits beyond mere chemical elegance. The primary advantage lies in the drastic simplification of the supply chain for raw materials. By avoiding reagents like sodium hydrosulfide and hydrogen peroxide, which are classified as dangerous goods with strict transportation and storage regulations, the logistical burden is significantly reduced. This shift allows for more flexible sourcing strategies and reduces the risk of supply disruptions caused by regulatory compliance issues associated with hazardous chemicals.
- Cost Reduction in Manufacturing: The elimination of column chromatography is a major driver for cost efficiency. In traditional routes, the consumption of silica gel and organic eluents represents a substantial variable cost that scales linearly with production volume. By replacing this with crystallization-based purification, the new method drastically lowers material costs and waste disposal fees. Furthermore, the ability to use cheaper, recyclable solvents like ethanol instead of expensive, hard-to-remove solvents like DMF contributes to a leaner cost structure in API manufacturing.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions translates directly to supply reliability. The process operates under mild temperatures and pressures, reducing the likelihood of batch failures due to thermal runaways or equipment limitations. The high purity achieved at each step means that intermediates can be carried forward with minimal rework, ensuring consistent lead times for high-purity pharmaceutical intermediates. This predictability is crucial for maintaining uninterrupted production schedules for downstream API synthesis.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this route is inherently more scalable. The absence of malodorous gas generation removes the need for complex off-gas treatment systems, lowering capital expenditure for new production lines. Additionally, the reduction in wastewater volume and solid waste (silica gel) aligns with increasingly stringent global environmental regulations, ensuring long-term operational continuity without the risk of regulatory shutdowns.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the comparative data and experimental examples provided in the patent documentation, highlighting the practical benefits for potential licensees and manufacturing partners.
Q: How does this new synthesis route improve safety compared to traditional methods?
A: Unlike previous methods that generate toxic hydrogen sulfide gas or use dangerous oxidants like hydrogen peroxide, this route utilizes mild addition and substitution reactions that do not produce malodorous or hazardous byproducts, significantly enhancing operational safety.
Q: What are the advantages regarding solvent usage and waste treatment?
A: The process avoids the extensive use of difficult-to-remove solvents like N,N-dimethylformamide (DMF) found in older routes. Instead, it employs safer, easily recoverable solvents like ethanol and tetrahydrofuran, drastically reducing wastewater volume and simplifying post-treatment.
Q: Is this method suitable for large-scale industrial production?
A: Yes, the method is specifically designed for industrial scalability. It eliminates the need for column chromatography purification, which is a bottleneck in large-scale manufacturing, and relies on robust crystallization techniques to achieve high purity efficiently.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Selinexor Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of life-saving oncology therapies. Our technical team has thoroughly analyzed the pathway described in CN112679477B and is well-positioned to execute this chemistry at scale. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale optimization to full-scale manufacturing is seamless. Our facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of Selinexor intermediate meets the exacting standards required by global regulatory bodies.
We invite pharmaceutical companies and contract research organizations to collaborate with us to leverage this advanced synthetic route. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our manufacturing capabilities can enhance your supply chain efficiency and reduce your overall cost of goods sold.
