Advanced Vacuum Reflux Technology for High-Purity Eprosartan Manufacturing and Commercial Scale-Up
Advanced Vacuum Reflux Technology for High-Purity Eprosartan Manufacturing and Commercial Scale-Up
The pharmaceutical industry constantly seeks robust synthetic routes that balance high purity with economic viability, particularly for critical antihypertensive agents like Eprosartan. Patent CN101215284B introduces a transformative preparation method that addresses the longstanding thermal instability issues associated with the Knoevenagel condensation step in Eprosartan synthesis. By shifting from traditional atmospheric reflux to a controlled vacuum reflux system, this technology effectively suppresses the spontaneous decarboxylation of key intermediates, a defect that has historically plagued production yields. This technical breakthrough not only elevates the crude product purity to approximately 98% but also streamlines the purification workflow by eliminating the need for complex column chromatography. For global supply chain stakeholders, this represents a pivotal shift towards more reliable pharmaceutical intermediates supplier capabilities, ensuring consistent quality for downstream API manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methodologies, specifically those outlined in US Patent 5185351, rely on atmospheric reflux in solvents like toluene, which necessitates temperatures around 108°C to drive the dehydration condensation. While thermodynamically sufficient for water removal, this elevated thermal energy creates a significant kinetic penalty for the thiophene-based diester reactant. Under these harsh conditions, the excess diester undergoes rapid spontaneous decarboxylation, converting into 2-thiophenepropionate before it can react with the imidazole aldehyde. This parasitic side reaction consumes valuable raw materials without generating the desired carbon-carbon double bond, resulting in abysmal isolated yields often hovering around 40%. Furthermore, the resulting crude mixture is chemically complex, typically requiring expensive and time-consuming column chromatography to separate the target molecule from decarboxylated byproducts, rendering the process economically unviable for large-scale industrial application.
The Novel Approach
The innovative strategy disclosed in CN101215284B fundamentally re-engineers the reaction thermodynamics by utilizing reduced pressure to lower the boiling point of the azeotropic solvent system. By maintaining the reflux temperature within a precise window of 50–100°C, and optimally between 65–75°C, the process kinetically favors the desired condensation over the undesired decarboxylation pathway. This thermal moderation allows the thiophene diester to remain stable long enough to react efficiently with the imidazole aldehyde in the presence of a piperidine-benzoic acid catalyst. The result is a dramatic enhancement in mass efficiency, with yields surging to the 70–85% range. Additionally, the cleaner reaction profile facilitates a simplified work-up procedure involving direct alkaline hydrolysis and acid precipitation, completely bypassing the need for silica gel purification and thereby drastically reducing both operational time and solvent consumption costs.
Mechanistic Insights into Vacuum-Assisted Knoevenagel Condensation
The core of this synthesis lies in the formation of the acrylic acid double bond linking the imidazole and thiophene moieties via a Knoevenagel condensation mechanism. In this specific embodiment, the reaction proceeds through the nucleophilic attack of the active methylene group of the thiophene diester on the carbonyl carbon of the imidazole aldehyde. The catalyst system, formed in situ from piperidine and benzoic acid, acts as a mild base to generate the enolate species while simultaneously facilitating the elimination of water. Crucially, the vacuum environment serves a dual purpose: it continuously removes the generated water to drive the equilibrium forward according to Le Chatelier's principle, and it strictly limits the thermal energy available to the system. This precise thermal control is vital because the activation energy for the decarboxylation of the thiophene diester is lower than previously assumed at high temperatures; by keeping the bulk temperature below 80°C, the rate constant for decarboxylation is minimized, preserving the integrity of the diester functionality until it is incorporated into the final scaffold.
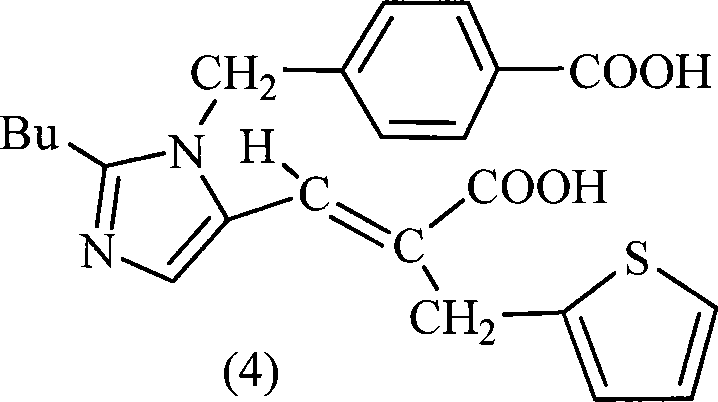
Following the condensation, the intermediate diester undergoes saponification using inorganic bases such as sodium hydroxide. This step cleaves the ester groups to reveal the free carboxylic acids necessary for biological activity. The patent highlights a critical purification nuance during the final isolation: adjusting the pH of the aqueous layer to a weakly acidic range of 3.0–6.5, preferably 5.0–5.5, induces the precipitation of Eprosartan. The addition of co-solvents like ethanol during this acidification step further enhances the crystallinity and purity of the precipitate by modifying the solubility parameters of the solution. This meticulous control over pH and solvent composition ensures that impurities, including any residual decarboxylated byproducts or unreacted starting materials, remain in the mother liquor, yielding a solid product with HPLC purity exceeding 95% without further recrystallization.
How to Synthesize Eprosartan Efficiently
The implementation of this vacuum reflux protocol requires precise engineering controls but follows a straightforward chemical logic suitable for standard reactor setups. The process begins with the dissolution of the imidazole aldehyde and thiophene diester in an azeotropic solvent like toluene, followed by the addition of the catalytic amine and acid components. The reactor is then sealed and subjected to vacuum to establish the target reflux temperature, typically maintained for 15 to 25 hours to ensure complete conversion. This operational simplicity makes the transition from laboratory benchtop to pilot plant highly feasible, minimizing the risk of scale-up failures often associated with complex multi-step syntheses.
- Condense the imidazole aldehyde and thiophene diester in toluene under reduced pressure (50-100°C) using a piperidine/benzoic acid catalyst.
- Hydrolyze the resulting diester intermediate using aqueous sodium hydroxide under reflux conditions.
- Acidify the reaction mixture to pH 3.0-6.5 to precipitate the final Eprosartan product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this vacuum-mediated synthesis route offers profound strategic advantages beyond mere yield improvements. The elimination of column chromatography is perhaps the most significant cost driver, as chromatographic purification is notoriously difficult to scale, consumes vast quantities of silica and solvents, and creates substantial hazardous waste streams. By replacing this unit operation with a simple crystallization induced by pH adjustment, the process achieves a substantial cost savings in both raw materials and waste disposal. Furthermore, the higher yield directly translates to a lower cost of goods sold (COGS), as less starting material is required to produce the same mass of active pharmaceutical ingredient, enhancing the overall margin structure for the final drug product.
- Cost Reduction in Manufacturing: The shift to a vacuum reflux system eliminates the thermal degradation of expensive thiophene raw materials, ensuring that a higher percentage of input mass is converted into saleable product. By avoiding the use of large volumes of chromatography solvents and stationary phases, the process significantly reduces variable production costs. The simplified downstream processing also lowers labor hours and utility consumption, contributing to a more competitive pricing structure for bulk pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: The robustness of this method against thermal side reactions means that batch-to-batch variability is minimized, a critical factor for maintaining consistent supply to API manufacturers. The use of common, commodity chemicals like toluene, piperidine, and sodium hydroxide ensures that raw material sourcing remains stable and unaffected by niche supply shocks. This reliability allows for longer production campaigns and better inventory planning, reducing the risk of stockouts for critical hypertension medications.
- Scalability and Environmental Compliance: From an environmental perspective, the reduction in solvent usage and the avoidance of silica waste align perfectly with modern green chemistry initiatives and regulatory expectations. The process is inherently scalable because vacuum distillation and reflux are standard unit operations in any GMP facility, requiring no exotic equipment. This ease of scale-up facilitates the rapid expansion of production capacity from 100 kgs to multi-tonne levels, ensuring that the supply chain can flexibly respond to market demand fluctuations without compromising quality or compliance standards.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this improved Eprosartan synthesis route. These insights are derived directly from the experimental data and comparative analysis provided in the patent literature, offering clarity on process parameters and quality outcomes. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this methodology for commercial production.
Q: How does the vacuum reflux method improve yield compared to conventional synthesis?
A: Conventional methods reflux at atmospheric pressure (~108°C in toluene), causing the excess thiophene diester reactant to spontaneously decarboxylate, lowering yield to ~40%. The vacuum method lowers the temperature to 65-75°C, suppressing this side reaction and boosting yield to 70-85%.
Q: What catalyst system is recommended for this condensation reaction?
A: The patent specifies a catalytic system comprising piperidine and benzoic acid (forming piperidine benzoate in situ), which effectively drives the dehydration condensation while maintaining mild reaction conditions suitable for sensitive functional groups.
Q: Is column chromatography required for purification in this new process?
A: No. Unlike prior art methods that require column chromatography, this improved process utilizes controlled crystallization via pH adjustment and solvent selection (adding ethanol/methanol), significantly simplifying downstream processing and reducing production costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Eprosartan Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires deep process engineering expertise and rigorous quality oversight. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of vacuum reflux technology are fully realized in our manufacturing suites. We maintain stringent purity specifications through our rigorous QC labs, utilizing advanced HPLC and NMR techniques to verify that every batch of Eprosartan meets the highest international pharmacopoeia standards. Our commitment to process excellence ensures that we deliver not just a chemical compound, but a validated, reliable supply solution for your antihypertensive drug portfolio.
We invite global partners to engage with our technical procurement team to discuss how this optimized synthesis route can enhance your supply chain resilience. By requesting a Customized Cost-Saving Analysis, you can gain specific insights into how our manufacturing efficiencies translate into tangible value for your organization. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your volume requirements, securing a partnership built on transparency, quality, and shared success in the competitive pharmaceutical marketplace.
