Advanced Catalytic Synthesis of 5-Octanoyl Salicylic Acid for Scalable Commercial Production
Introduction to Next-Generation Synthesis Technology
The global demand for high-performance cosmetic active ingredients continues to surge, driven by consumer preference for effective yet gentle skincare solutions. At the forefront of this trend is 5-octanoyl salicylic acid, also known as Beta-LHA, a lipophilic derivative of salicylic acid renowned for its superior exfoliating properties and enhanced skin permeability. A pivotal advancement in the manufacturing of this critical intermediate is detailed in patent CN113200843A, which discloses a novel preparation method that fundamentally reimagines the Friedel-Crafts acylation process. Unlike traditional methodologies that rely on harsh conditions and toxic solvents, this innovation employs a sophisticated composite catalyst system comprising a Lewis acid and tris(pentafluorophenyl)borane. This technical breakthrough not only addresses long-standing purity challenges but also aligns with modern green chemistry principles, offering a robust pathway for reliable cosmetic intermediate suppliers to meet escalating market requirements with greater efficiency and environmental responsibility.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of 5-octanoyl salicylic acid has been plagued by significant technical and environmental hurdles that compromise both product quality and operational safety. The conventional route, as illustrated in prior art such as US 1988/4767750A, typically utilizes methyl salicylate and octanoyl chloride in the presence of aluminum trichloride, with carbon disulfide serving as the reaction solvent. This legacy approach suffers from severe drawbacks, primarily due to the excessive activity of aluminum trichloride which promotes uncontrolled side reactions. These side reactions lead to the formation of substantial amounts of tar-like octenone polymers, resulting in a crude product that is dark in color and heavily contaminated. Consequently, manufacturers are forced to engage in labor-intensive post-treatment procedures, often requiring multiple rounds of recrystallization to achieve acceptable purity levels. Furthermore, the reliance on carbon disulfide presents a major liability; as a highly toxic and volatile solvent, its use necessitates stringent safety controls and expensive waste management protocols, making it increasingly untenable for modern industrial production.
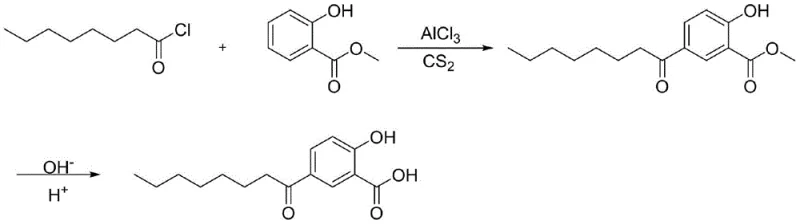
The Novel Approach
In stark contrast to these outdated practices, the methodology disclosed in patent CN113200843A introduces a paradigm shift through the implementation of a dual-catalyst system. By combining a standard Lewis acid with tris(pentafluorophenyl)borane, the reaction achieves a level of regioselectivity and control that was previously unattainable. This synergistic catalytic effect allows for the substitution of hazardous carbon disulfide with safer, more manageable organic solvents such as dichloromethane or dichloroethane. The result is a reaction profile that drastically minimizes the generation of polymeric tars, yielding a product with a significantly lighter color profile right from the crude stage. Moreover, the process incorporates an innovative purification strategy involving salt formation with an organic base, which effectively isolates the target molecule from impurities in a single step. This streamlined workflow eliminates the need for repetitive recrystallization, thereby reducing processing time and solvent consumption while ensuring a consistent, high-quality output suitable for sensitive cosmetic applications.
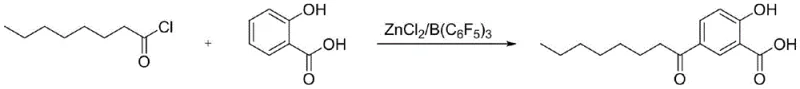
Mechanistic Insights into Composite Catalyst Friedel-Crafts Acylation
The core of this technological advancement lies in the unique mechanistic interaction between the Lewis acid and the boron-based co-catalyst. In a standard Friedel-Crafts acylation, the Lewis acid activates the acyl chloride to form an acylium ion, which then attacks the aromatic ring. However, in the case of salicylic acid derivatives, the presence of hydroxyl and carboxyl groups can lead to complexation issues and poor regioselectivity. The addition of tris(pentafluorophenyl)borane acts as a powerful Lewis acid enhancer, likely coordinating with the carbonyl oxygen or the phenolic hydroxyl group to further activate the substrate or stabilize the transition state. This cooperative catalysis ensures that the acylation occurs preferentially at the 5-position of the salicylic acid ring, minimizing the formation of ortho- or other meta-substituted isomers. The precise tuning of electronic effects by the pentafluorophenyl groups on the boron atom creates a highly specific reactive environment that suppresses competing polymerization pathways, which are the root cause of the tar formation seen in conventional methods.
Furthermore, the purification mechanism employed in this process is equally critical for maintaining high purity standards essential for a reliable cosmetic intermediate supplier. Following the acylation, the crude reaction mixture is treated with an organic base such as DABCO, DBU, or diisopropylethylamine. This step converts the acidic 5-octanoyl salicylic acid into a soluble salt, while neutral impurities and unreacted starting materials remain in the organic phase or precipitate out. Subsequent acidification with dilute sulfuric or hydrochloric acid regenerates the free acid in a highly pure crystalline form. This acid-base extraction technique is far superior to simple recrystallization because it exploits the chemical properties of the target molecule rather than just solubility differences, effectively scrubbing away structurally similar byproducts. The result is a final product with an HPLC purity of up to 99.7% achieved in a single pass, demonstrating the robustness of this chemical design for producing high-purity personal care active ingredients.
How to Synthesize 5-Octanoyl Salicylic Acid Efficiently
Implementing this advanced synthesis route requires careful attention to reaction parameters and stoichiometry to maximize the benefits of the composite catalyst system. The process begins with the preparation of the catalyst solution under inert atmosphere, followed by the controlled addition of reactants to manage exothermicity. Detailed operational guidelines regarding temperature profiles, molar ratios, and workup procedures are critical for reproducibility. For R&D teams looking to adopt this technology, the following standardized synthesis steps outline the optimal protocol derived from the patent examples to ensure successful scale-up and consistent quality.
- Prepare the catalyst system by dissolving a Lewis acid (such as ZnCl2, FeCl3, AlCl3, or BCl3) and tris(pentafluorophenyl)borane in an organic solvent like dichloromethane.
- Perform Friedel-Crafts acylation by dropwise adding octanoyl chloride and salicylic acid solution at controlled low temperatures (0-30°C).
- Purify the crude product through organic base salt formation, followed by acid dissociation to isolate the final high-purity active ingredient.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this novel synthesis route offers compelling economic and logistical benefits that extend beyond mere technical superiority. The elimination of carbon disulfide from the process removes a significant regulatory and safety burden, simplifying facility compliance and reducing the costs associated with hazardous material handling and disposal. Additionally, the reduction in tar formation means that reactor cleaning cycles can be extended, and filtration equipment experiences less fouling, leading to improved asset utilization and lower maintenance overheads. The ability to achieve high purity without multiple recrystallization steps translates directly into reduced solvent usage and shorter batch cycle times, which are key drivers for cost reduction in fine chemical manufacturing. These efficiencies collectively enhance the overall profitability of the production line while ensuring a more stable and predictable supply of this valuable active ingredient.
- Cost Reduction in Manufacturing: The adoption of the composite catalyst system fundamentally alters the cost structure of production by minimizing raw material waste and energy consumption. By avoiding the formation of heavy tars, the process significantly reduces the loss of valuable starting materials into byproduct streams, thereby improving the effective yield of the reaction. Furthermore, the streamlined purification process eliminates the need for energy-intensive repeated recrystallizations, which traditionally consume large volumes of solvents and require extensive heating and cooling cycles. This reduction in downstream processing complexity leads to substantial cost savings in utilities and waste treatment, allowing manufacturers to offer competitive pricing without compromising on margin. The use of readily available and relatively inexpensive Lewis acids like zinc chloride or iron chloride, supplemented by a catalytic amount of the borane species, ensures that catalyst costs remain manageable even at large scales.
- Enhanced Supply Chain Reliability: From a supply chain perspective, the robustness of this synthetic route mitigates many of the risks associated with specialty chemical production. The raw materials required, specifically salicylic acid and octanoyl chloride, are commodity chemicals with well-established global supply networks, reducing the risk of raw material shortages. The process tolerance to slight variations in reaction conditions, afforded by the stabilizing effect of the boron co-catalyst, ensures consistent batch-to-batch quality, which is crucial for maintaining long-term contracts with major cosmetic brands. Moreover, the shift away from controlled solvents like carbon disulfide removes potential bottlenecks related to strict transportation and storage regulations, facilitating smoother logistics and faster delivery times. This reliability makes the manufacturer a preferred partner for clients seeking a dependable source of high-purity cosmetic actives.
- Scalability and Environmental Compliance: As the industry moves towards more sustainable manufacturing practices, this process stands out for its alignment with green chemistry principles. The replacement of toxic solvents with dichloromethane or dichloroethane, while still requiring care, represents a significant improvement in terms of worker safety and environmental impact compared to carbon disulfide. The drastic reduction in solid waste (tar) simplifies effluent treatment and lowers the environmental footprint of the facility. This cleaner production profile not only aids in meeting increasingly stringent environmental regulations but also enhances the brand image of the end-product as being sustainably sourced. The process is inherently scalable, having been designed with industrial constraints in mind, allowing for seamless expansion from pilot plant quantities to multi-ton commercial production runs without the need for complex re-engineering.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis method is vital for stakeholders evaluating its potential for their supply chains. The following questions address common inquiries regarding the practical implementation, quality control, and regulatory aspects of the new process. These insights are derived directly from the experimental data and technical disclosures within the patent, providing a clear picture of what partners can expect when integrating this technology into their operations.
Q: What are the primary advantages of the composite catalyst system over traditional aluminum trichloride methods?
A: The composite catalyst system significantly reduces tar formation and eliminates the need for toxic carbon disulfide solvents, resulting in a lighter product color and simplified downstream processing.
Q: How does the new purification method impact the overall yield and purity?
A: By utilizing organic base salt formation followed by acidification, the process achieves a single-step purity of up to 99.7% without the need for repeated recrystallization, thereby improving overall recovery rates.
Q: Is this synthesis route suitable for large-scale industrial manufacturing?
A: Yes, the use of common solvents like dichloromethane and readily available Lewis acids, combined with reduced waste generation, makes this route highly scalable and economically viable for commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Octanoyl Salicylic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced manufacturing processes requires a partner with deep technical expertise and a commitment to excellence. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of this novel catalytic route are fully realized at an industrial level. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of 5-octanoyl salicylic acid meets the highest standards required for premium cosmetic formulations. We understand the critical nature of active ingredients in personal care and are dedicated to delivering products that empower our clients to innovate with confidence.
We invite you to collaborate with us to explore how this optimized synthesis route can enhance your product portfolio and operational efficiency. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating the tangible economic benefits of switching to this superior method. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a sustainable and cost-effective supply of this high-value cosmetic intermediate.
