Revolutionizing 5-Hydroxy-1-indanone Production: A Strategic Analysis of Patent CN101585758B for Global Supply Chains
The pharmaceutical and fine chemical industries are constantly seeking robust synthetic pathways that balance high purity with economic viability, a challenge prominently addressed in patent CN101585758B. This intellectual property discloses a sophisticated methodology for the preparation of 5-hydroxy-1-indanone, a critical building block utilized extensively in medicinal chemistry and complex organic synthesis. The innovation lies in its departure from traditional, cumbersome protocols, introducing a streamlined approach that leverages trifluoromethanesulfonic acid to facilitate efficient ring closure. For R&D Directors and Procurement Managers alike, understanding the nuances of this patent is essential, as it represents a significant leap forward in process reliability and operational safety. By shifting the paradigm from harsh Lewis acid melts to a more controlled acidic mediation, the technology offers a compelling value proposition for manufacturers aiming to optimize their supply chains for high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
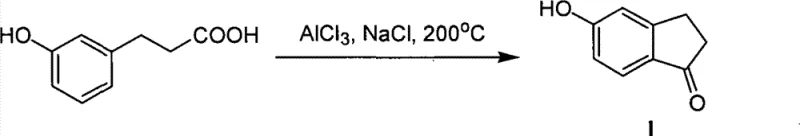
Historically, the synthesis of 5-hydroxy-1-indanone has been plagued by severe operational bottlenecks that hinder scalable manufacturing and inflate production costs. One prevalent legacy method involves the intramolecular cyclization of hydroxyphenylacetic acid derivatives using a molten mixture of aluminum chloride and sodium chloride at temperatures reaching 200°C. This approach, while chemically feasible, presents catastrophic engineering challenges, including the necessity to physically break reaction vessels to retrieve the solidified product mass, which drastically increases labor costs and safety risks. Furthermore, the reliance on stoichiometric amounts of corrosive Lewis acids generates substantial quantities of hazardous waste, complicating environmental compliance and disposal logistics. Alternative routes involving the demethylation of methoxy-precursors or the transformation of amino-derivatives often suffer from multi-step inefficiencies and the requirement for scarce or expensive starting materials. These legacy constraints create significant friction in the supply chain, leading to inconsistent batch quality and extended lead times that are unacceptable for modern just-in-time manufacturing environments.
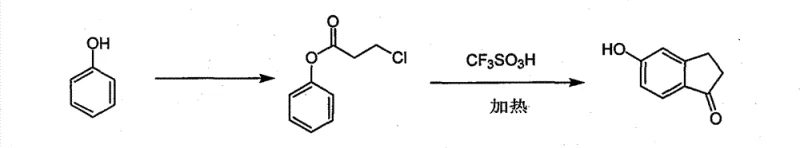
The Novel Approach
In stark contrast to these archaic techniques, the methodology outlined in CN101585758B introduces a elegant two-step sequence that prioritizes operational simplicity and raw material accessibility. The process initiates with the esterification of phenol, a widely available and cost-effective commodity chemical, with 3-chloropropionyl chloride to form a stable ester intermediate. This initial step proceeds under relatively mild thermal conditions, avoiding the extreme temperatures that degrade equipment and compromise safety. The subsequent cyclization step utilizes trifluoromethanesulfonic acid not merely as a catalyst but as a reaction medium, enabling the intramolecular Friedel-Crafts acylation to proceed with high efficiency. This strategic shift eliminates the need for solid-state melting processes and allows for homogeneous reaction conditions that are far easier to monitor and control. For a reliable pharmaceutical intermediate supplier, adopting such a route translates directly into enhanced process robustness and the ability to consistently deliver high-purity materials without the logistical nightmares associated with traditional methods.
Mechanistic Insights into Trifluoromethanesulfonic Acid Mediated Cyclization
The core chemical innovation of this patent resides in the utilization of trifluoromethanesulfonic acid (TfOH) to drive the intramolecular acylation of the phenolic ester intermediate. Mechanistically, the superacidic nature of TfOH serves to protonate the carbonyl oxygen of the ester side chain, generating a highly reactive acylium ion species in situ. This electrophilic center is then poised for attack by the electron-rich aromatic ring of the phenol moiety, facilitating the formation of the new carbon-carbon bond required to close the five-membered indanone ring. Unlike traditional Lewis acids which often form stable complexes with the product requiring harsh hydrolysis for release, the TfOH mediated system allows for a cleaner reaction profile. The acidity strength ensures that the cyclization occurs rapidly even at moderate temperatures compared to molten salt methods, reducing the thermal stress on the molecular framework and minimizing the formation of thermal degradation by-products. This mechanistic elegance is crucial for R&D teams focused on impurity profiling, as it inherently limits the generation of complex polymeric tars often seen in high-temperature Friedel-Crafts reactions.
Furthermore, the control over regioselectivity and impurity formation is significantly enhanced through the precise modulation of reaction temperature and acid equivalents. The patent data indicates that maintaining the reaction temperature within the range of 120°C to 200°C is critical; lower temperatures may result in incomplete conversion or the isolation of transition state intermediates, while excessive heat could promote polymerization. The use of 5 to 20 equivalents of trifluoromethanesulfonic acid provides a tunable parameter to optimize the reaction kinetics without necessitating exotic catalysts. From a quality control perspective, this homogeneity ensures that the final crude product contains fewer inorganic salts and metal residues, simplifying the downstream purification process. For manufacturers targeting high-purity OLED material or pharmaceutical grades, this reduction in metallic contamination is a decisive advantage, as it reduces the burden on recrystallization steps and improves overall yield recovery. The ability to quench the reaction simply by pouring into ice water further underscores the practical superiority of this chemical design over methods requiring complex workups.
How to Synthesize 5-Hydroxy-1-indanone Efficiently
Implementing this synthesis route requires strict adherence to the thermal profiles and stoichiometric ratios defined in the patent to ensure optimal yield and safety. The process begins with the preparation of the 3-chloropropionic acid phenyl ester, where phenol is reacted with 3-chloropropionyl chloride at approximately 100°C under nitrogen protection to prevent oxidation. Once the esterification is complete, as confirmed by TLC analysis, the intermediate is isolated and subsequently dissolved in trifluoromethanesulfonic acid for the cyclization step. The detailed standardized synthesis steps see the guide below, which outlines the precise addition rates and temperature ramps necessary to maximize the formation of the target indanone structure while minimizing side reactions. Operators must ensure that the cooling phase is managed carefully to prevent thermal shock during the aqueous quench, which is a critical safety consideration when handling strong acids.
- React phenol with 3-chloropropionyl chloride at 100°C to form 3-chloropropionic acid phenyl ester.
- Dissolve the ester intermediate in trifluoromethanesulfonic acid acting as both solvent and catalyst.
- Heat the mixture to 120-200°C to induce intramolecular Friedel-Crafts acylation and ring closure.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of the synthesis method described in CN101585758B offers tangible strategic benefits that extend beyond mere chemical curiosity. The primary advantage lies in the drastic simplification of the manufacturing workflow, which directly correlates to reduced operational expenditures and enhanced throughput. By eliminating the need for specialized high-temperature melting equipment and the labor-intensive process of breaking reaction vessels, facilities can repurpose existing standard glass-lined reactors for production. This flexibility allows for cost reduction in fine chemical manufacturing by lowering capital expenditure requirements and minimizing downtime associated with equipment maintenance and cleaning. Moreover, the use of phenol as a starting material ensures a stable supply base, as it is a globally traded commodity with multiple sourcing options, thereby mitigating the risk of raw material shortages that often plague niche synthetic routes.
- Cost Reduction in Manufacturing: The elimination of stoichiometric aluminum chloride and sodium chloride salts removes a significant cost center related to both reagent purchase and hazardous waste disposal. Traditional methods generate massive amounts of aluminum sludge that require expensive treatment, whereas the TfOH method allows for potential acid recovery or simpler neutralization workflows. Additionally, the higher selectivity of the new route means less raw material is wasted on by-products, improving the effective atom economy of the process. This efficiency translates into substantial cost savings per kilogram of finished product, allowing suppliers to offer more competitive pricing structures without compromising margin integrity. The simplified workup also reduces solvent consumption and energy usage during the drying and purification phases, further contributing to a leaner cost profile.
- Enhanced Supply Chain Reliability: Relying on readily available industrial chemicals like phenol and 3-chloropropionyl chloride decouples production from the volatility of specialized precursor markets. This stability is crucial for reducing lead time for high-purity pharmaceutical intermediates, as manufacturers do not need to wait for custom synthesis of starting materials. The robustness of the reaction conditions also implies a lower failure rate for production batches, ensuring consistent delivery schedules for downstream clients. In a global market where continuity of supply is paramount, a process that minimizes the risk of batch rejection due to impurity spikes provides a significant competitive edge. Suppliers utilizing this technology can confidently commit to long-term contracts, knowing that their production capacity is not constrained by fragile or unpredictable chemical steps.
- Scalability and Environmental Compliance: The transition from a solid-state melt process to a liquid-phase reaction significantly eases the challenges of commercial scale-up of complex pharmaceutical intermediates. Liquid handling is inherently easier to automate and control at the multi-ton scale compared to managing viscous molten salts. From an environmental standpoint, the reduction in heavy metal waste aligns with increasingly stringent global regulations regarding industrial effluent. This compliance reduces the regulatory burden on manufacturing sites and lowers the risk of production halts due to environmental audits. The ability to run the reaction in standard reactors also means that capacity can be scaled up rapidly by utilizing existing infrastructure, facilitating a quicker response to surges in market demand without the need for lengthy construction projects.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this novel synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on how this method outperforms historical precedents. Understanding these details is vital for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios. The answers highlight the specific operational improvements and quality enhancements that stakeholders can expect when transitioning to this advanced protocol.
Q: What are the primary advantages of the TfOH mediated route over traditional AlCl3 methods?
A: The trifluoromethanesulfonic acid method eliminates the need for harsh molten salt conditions and difficult post-reaction processing associated with aluminum chloride, significantly simplifying purification and reducing equipment corrosion risks.
Q: Is the raw material phenol readily available for large-scale production?
A: Yes, phenol is a ubiquitous industrial commodity chemical, ensuring a stable and cost-effective supply chain compared to specialized precursors required by older synthetic methodologies.
Q: How does this synthesis method impact environmental compliance?
A: By avoiding heavy metal catalysts and simplifying the workup to standard aqueous extraction, the process generates less hazardous waste and aligns better with modern green chemistry standards.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Hydroxy-1-indanone Supplier
At NINGBO INNO PHARMCHEM, we recognize that the theoretical advantages of a patent must be translated into tangible production capabilities to truly serve our global partners. As a premier CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the efficiencies promised by CN101585758B are fully realized in practice. Our facilities are equipped with the corrosion-resistant reactor systems necessary to handle trifluoromethanesulfonic acid safely and efficiently, guaranteeing a seamless transition from lab scale to industrial volume. We maintain stringent purity specifications and operate rigorous QC labs to verify that every batch of 5-hydroxy-1-indanone meets the exacting standards required for pharmaceutical and electronic applications. Our commitment to quality ensures that the impurity profiles remain tightly controlled, providing our clients with the consistency needed for their own regulatory filings.
We invite you to collaborate with us to leverage this advanced synthesis route for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this methodology for your supply chain. We encourage you to contact us to request specific COA data and route feasibility assessments tailored to your volume requirements. By partnering with us, you gain access to a supply chain that is not only cost-effective but also resilient and compliant with the highest international standards. Let us help you optimize your sourcing strategy and secure a reliable flow of high-quality intermediates for your future production cycles.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
