Advanced Green Synthesis of Diethyl Azodicarboxylate for High-Purity Pharmaceutical Intermediates
Advanced Green Synthesis of Diethyl Azodicarboxylate for High-Purity Pharmaceutical Intermediates
The global demand for high-performance reagents in organic synthesis continues to drive innovation in process chemistry, particularly for critical compounds like Diethyl Azodicarboxylate (DEAD). As detailed in patent CN102898328A, a novel synthetic methodology has been developed that fundamentally shifts the production paradigm from hazardous halogenated precursors to cleaner carbonate-based chemistry. This technological breakthrough addresses long-standing safety and environmental concerns associated with traditional manufacturing while delivering superior yield and purity profiles essential for modern API production. For R&D leaders and procurement strategists, understanding this shift is vital for securing a reliable pharmaceutical intermediate supplier capable of meeting stringent regulatory and quality standards. The following analysis dissects the technical merits and commercial implications of this advanced route.
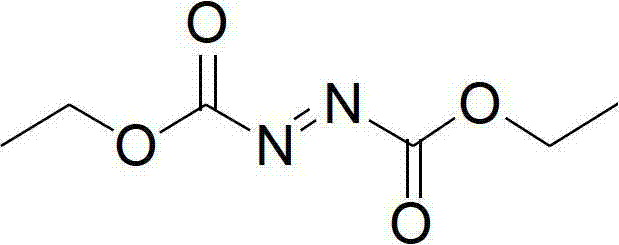
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of azodicarboxylates relied heavily on the Rabjohn method or variations utilizing ethyl chloroformate and hydrazine hydrate, followed by oxidation with chlorine gas or concentrated nitric acid. These legacy processes present severe operational bottlenecks; ethyl chloroformate is a volatile, flammable liquid with potent lachrymatory effects, posing significant occupational health risks and requiring expensive containment infrastructure. Furthermore, the oxidation step using chlorine gas generates substantial quantities of sodium hypochlorite waste, creating a heavy burden on wastewater treatment facilities and increasing the environmental footprint of the facility. Alternative methods using fuming nitric acid operate at cryogenic temperatures (0-5°C), demanding high energy input for cooling while releasing corrosive yellow nitrogen oxide fumes that degrade equipment integrity over time. These factors collectively inflate the cost of production and introduce supply chain volatility due to strict regulatory oversight on hazardous material transport and disposal.
The Novel Approach
In stark contrast, the methodology disclosed in the referenced patent utilizes diethyl carbonate and ethyl carbazate as primary feedstocks, catalyzed by sodium ethoxide to form the hydrazo-intermediate. This transesterification-based approach eliminates the need for toxic chloroformates entirely, replacing them with a greener, more stable carbonate source that aligns with modern green chemistry principles. The subsequent oxidation step employs hydrogen peroxide activated by a bromide catalyst system (such as hydrobromic acid or sodium bromide) within a mild temperature window of -15°C to 45°C. This shift not only mitigates the risks associated with handling elemental chlorine but also simplifies the workup procedure, as the byproducts are primarily water and benign salts. The result is a robust, scalable process that offers cost reduction in API manufacturing by streamlining waste management and reducing the need for specialized corrosion-resistant reactors, thereby enhancing overall process economics.
Mechanistic Insights into Bromide-Catalyzed Oxidative Coupling
The core of this synthetic innovation lies in the efficient conversion of diethyl hydroazodicarboxylate to the final azo compound via a bromide-catalyzed oxidation mechanism. In the first stage, sodium ethoxide facilitates the nucleophilic attack of ethyl carbazate on diethyl carbonate, driving the equilibrium towards the formation of the hydrazo-intermediate through the elimination of ethanol. This step is carefully controlled by adjusting the pH to between 3 and 8, ensuring the precipitation of high-purity white crystals of the intermediate, which can be further purified via recrystallization using acetone and petroleum ether. The structural integrity of this intermediate is crucial, as it serves as the direct precursor for the final oxidative coupling. By isolating this intermediate, manufacturers can implement rigorous quality control checkpoints before proceeding to the oxidation stage, ensuring that impurities do not carry over into the final active reagent.
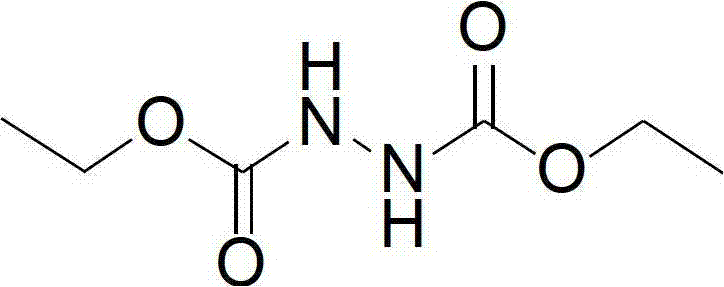
The second stage involves the oxidation of the hydrazo-linkage (-NH-NH-) to the azo-linkage (-N=N-) using hydrogen peroxide in an acidic medium. The presence of bromide ions acts as a redox mediator, generating reactive bromine species in situ that facilitate the electron transfer required for dehydrogenation. This catalytic cycle allows the reaction to proceed rapidly at ambient or slightly sub-ambient temperatures (5°C to 25°C is preferred), minimizing thermal stress on the sensitive ester functionalities. Unlike nitric acid oxidation, which can lead to over-oxidation or nitration side products, this system is highly selective, yielding the characteristic orange-yellow liquid of high-purity pharmaceutical intermediates with minimal byproduct formation. The ability to tune the reaction temperature between -15°C and 45°C provides process engineers with flexibility to optimize kinetics versus safety, ensuring consistent batch-to-batch reproducibility essential for commercial scale-up of complex pharmaceutical intermediates.
How to Synthesize Diethyl Azodicarboxylate Efficiently
Implementing this synthesis route requires precise control over stoichiometry and reaction conditions to maximize yield and safety. The process begins with the preparation of ethyl carbazate from hydrazine hydrate and excess diethyl carbonate, followed by the base-catalyzed condensation to form the hydrazo-intermediate. Critical parameters include maintaining the molar ratio of diethyl carbonate to ethyl carbazate between 1:1 and 1.3:1 and utilizing sodium ethoxide at 10% to 110% molar equivalence relative to the carbazate. Following isolation of the intermediate, the oxidation is performed by dissolving the solid in dilute sulfuric acid, adding the bromide catalyst, and slowly dosing hydrogen peroxide (10%-40% concentration). Detailed standard operating procedures regarding mixing rates, addition times, and quenching protocols are essential for safe operation. For the complete standardized synthesis steps and specific parameter optimization data, please refer to the guide below.
- Condense diethyl carbonate and ethyl carbazate using sodium ethoxide at 70-140°C, then adjust pH to precipitate diethyl hydroazodicarboxylate.
- Oxidize the intermediate using hydrogen peroxide and a bromide catalyst in acidic solution at temperatures between -15°C and 45°C.
- Extract the final product with organic solvents like dichloromethane or ethyl acetate and purify via vacuum distillation.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, adopting this patented synthesis route offers tangible benefits that extend beyond mere chemical yield. The substitution of hazardous raw materials with commercially abundant and less regulated chemicals like diethyl carbonate and hydrogen peroxide significantly de-risks the supply chain. Procurement managers can secure raw materials with greater ease and at lower price points compared to specialized chloroformates, which are often subject to volatile market pricing and strict transportation regulations. Furthermore, the recyclability of solvents and excess reagents mentioned in the patent documentation implies a circular economy approach within the manufacturing plant, directly contributing to substantial cost savings in raw material consumption. This efficiency translates into more competitive pricing structures for downstream customers without compromising on the quality or purity specifications required for sensitive pharmaceutical applications.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous chlorine gas handling systems reduces capital expenditure (CAPEX) on specialized containment and scrubbing infrastructure. Additionally, the ability to recycle excess diethyl carbonate and solvents like acetone and petroleum ether drastically lowers the variable cost per kilogram of production. By avoiding the generation of heavy metal waste or complex halogenated byproducts, the facility also sees a reduction in waste disposal fees and environmental compliance costs. These cumulative efficiencies allow for a leaner manufacturing model that can withstand market fluctuations better than traditional chloroformate-based processes, ensuring long-term price stability for buyers.
- Enhanced Supply Chain Reliability: The operational simplicity of this method, characterized by a wide reaction temperature range and stable reaction kinetics, minimizes the risk of batch failures due to thermal runaway or equipment malfunction. Traditional methods requiring cryogenic conditions (-78°C or lower) or highly corrosive acids are prone to unplanned downtime for maintenance and repair. In contrast, this process operates comfortably between -15°C and 45°C, utilizing standard glass-lined or stainless steel reactors available in most multipurpose chemical plants. This compatibility with existing infrastructure ensures reducing lead time for high-purity pharmaceutical intermediates, as production slots can be secured more easily without needing dedicated, specialized lines that are often bottlenecked in the industry.
- Scalability and Environmental Compliance: Scaling this process from laboratory to industrial tonnage is facilitated by the exothermic nature of the oxidation being manageable through standard cooling jackets, removing the need for complex cryogenic engineering. The use of hydrogen peroxide as the terminal oxidant results in water as the primary byproduct, aligning with increasingly stringent global environmental regulations regarding effluent discharge. Facilities adopting this technology can market their production as 'green' or 'sustainable,' a valuable attribute for multinational corporations aiming to reduce their Scope 3 emissions. This environmental compliance future-proofs the supply chain against tightening regulations, ensuring uninterrupted supply continuity even as regulatory landscapes evolve globally.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of Diethyl Azodicarboxylate using this advanced methodology. These insights are derived directly from the technical specifications and experimental data provided in the patent literature, ensuring accuracy and relevance for industry professionals. Understanding these nuances helps stakeholders make informed decisions regarding vendor selection and process integration. For further technical clarification or custom synthesis requests, our team is available to provide detailed discussions.
Q: What are the safety advantages of this DEAD synthesis method compared to traditional routes?
A: This method replaces hazardous ethyl chloroformate and chlorine gas with diethyl carbonate and hydrogen peroxide, significantly reducing toxicity and corrosion risks while minimizing hazardous waste generation.
Q: How does the new oxidation system improve product purity?
A: The use of a bromide-catalyzed hydrogen peroxide oxidation system operates under milder conditions (-15°C to 45°C) compared to nitric acid oxidation, reducing side reactions and thermal decomposition, thereby yielding higher purity orange-yellow liquid product.
Q: Can the raw materials in this process be recycled to lower costs?
A: Yes, the process allows for the recycling of excess diethyl carbonate, recrystallization solvents like acetone and petroleum ether, and extraction agents, which substantially lowers the overall cost of goods sold (COGS) for large-scale production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Diethyl Azodicarboxylate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to greener, more efficient synthetic routes is critical for the future of the fine chemical industry. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in practical, large-scale manufacturing. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch of Diethyl Azodicarboxylate meets the exacting standards required for Mitsunobu reactions and other sensitive organic transformations. Our commitment to quality assurance ensures that your R&D and production timelines are never compromised by reagent variability.
We invite procurement leaders and technical directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements. By leveraging our optimized production capabilities, we can help you achieve significant reductions in total cost of ownership while securing a stable supply of critical reagents. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments for your upcoming projects. Let us partner with you to drive innovation and efficiency in your supply chain, ensuring you stay ahead in a competitive global market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
