Advanced Refining Technology for Nicardipine Hydrochloride: Ensuring Ultra-High Purity and Commercial Scalability
Advanced Refining Technology for Nicardipine Hydrochloride: Ensuring Ultra-High Purity and Commercial Scalability
The pharmaceutical industry constantly demands higher purity standards for cardiovascular medications, particularly for calcium channel blockers like Nicardipine Hydrochloride. A significant breakthrough in this domain is documented in patent CN113072483A, which introduces a sophisticated refining method capable of drastically reducing persistent impurities without compromising yield. This technology addresses a critical bottleneck in the manufacturing of high-purity API intermediates, where traditional recrystallization often fails to remove complex, polymer-like byproducts formed during synthesis. By leveraging a selective alkaline hydrolysis strategy followed by precise solvent engineering, this process achieves product purity levels ranging from 99.7% to 99.9%, setting a new benchmark for quality control in cardiovascular drug manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional purification routes for Nicardipine Hydrochloride often struggle with a specific class of stubborn contaminants known as polymer-like impurities. These byproducts, structurally depicted in Formula I, are generated inadvertently during the esterification stages of the synthetic pathway. Because their chemical structure closely mimics the target molecule, standard physical separation techniques such as simple recrystallization are frequently ineffective. Consequently, manufacturers are forced to perform multiple refining cycles to meet regulatory purity specifications. This repetitive processing not only drastically reduces the overall production yield but also escalates operational costs due to increased solvent consumption and extended processing times. Furthermore, the accumulation of these impurities can negatively impact the stability and bioavailability of the final drug product, posing significant risks for downstream formulation.
The Novel Approach
The innovative method described in the patent circumvents these limitations by chemically modifying the impurities rather than just physically separating them. Instead of relying solely on solubility differences, the process employs a controlled alkaline hydrolysis step that selectively degrades the ester linkages within the polymer-like impurities. This chemical transformation converts the stubborn, lipophilic impurities into more polar, water-soluble derivatives that can be easily washed away during the workup phase. Following this chemical purification, a final crystallization using a high-polarity organic solvent ensures the precipitation of ultra-pure Nicardipine Hydrochloride. This dual-mechanism approach—chemical degradation followed by physical crystallization—allows for the removal of impurities in a single pass, eliminating the need for repetitive refining loops and significantly streamlining the production workflow.
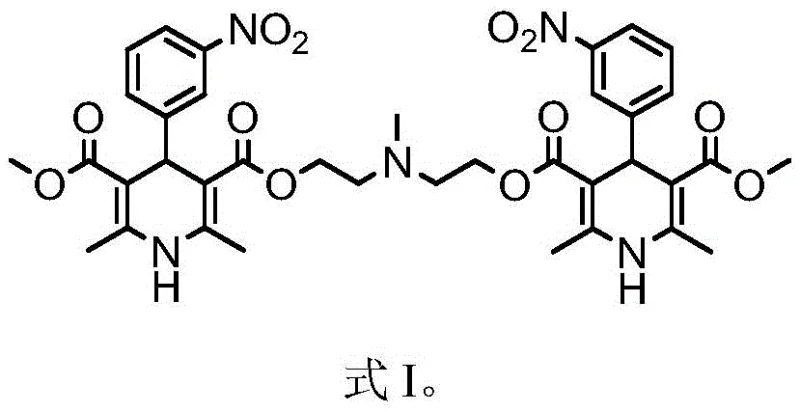
Mechanistic Insights into Alkaline Hydrolysis Purification
The core of this refining technology lies in the precise manipulation of reaction conditions to exploit the chemical vulnerability of the impurities. As illustrated in the reaction scheme, the impurity molecule contains multiple ester functionalities that are susceptible to nucleophilic attack by hydroxide ions. When the crude product mixture is treated with an aqueous alkaline solution, such as sodium carbonate or sodium hydroxide, these ester bonds undergo hydrolysis. This reaction cleaves the molecular framework of the impurity, breaking it down into smaller, more polar fragments like carboxylic acids and alcohols. Crucially, the reaction conditions—specifically the temperature range of 10-60°C and the concentration of the base—are optimized to ensure that while the impurities are degraded, the dihydropyridine core of the Nicardipine Hydrochloride remains intact and stable.
Following the hydrolysis, the system utilizes a biphasic extraction strategy to separate the degraded impurities from the target API. The addition of dichloromethane creates an organic phase that preferentially dissolves the Nicardipine Hydrochloride, while the newly formed polar hydrolysis products partition into the aqueous phase or remain insoluble in the crystallization solvent. This selectivity is further enhanced by the final crystallization step using acetonitrile or ethanol. These polar solvents have a high affinity for the hydrolyzed impurity fragments, keeping them in the mother liquor while allowing the pure Nicardipine Hydrochloride to precipitate out as a high-quality solid. This mechanistic understanding allows for robust process control, ensuring that batch-to-batch variability is minimized and purity specifications are consistently met.
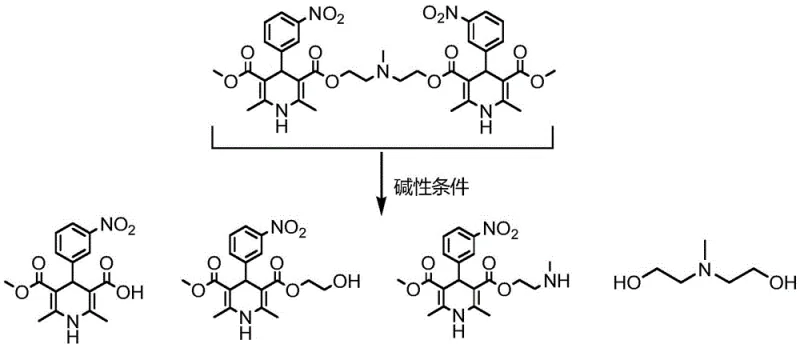
How to Synthesize Nicardipine Hydrochloride Efficiently
Implementing this refining protocol requires careful attention to solvent ratios and reaction kinetics to maximize efficiency. The process begins with the complete dissolution of the crude material in methanol, followed by solvent exchange into dichloromethane to facilitate the subsequent aqueous workup. The critical hydrolysis step is then performed under mild heating, typically around 40°C, to activate the degradation of impurities without risking thermal decomposition of the API. After phase separation and washing, the final isolation is achieved through controlled cooling crystallization. For a detailed breakdown of the specific mass ratios, temperatures, and timing required for each stage of this optimized workflow, please refer to the standardized synthesis guide below.
- Dissolve crude Nicardipine Hydrochloride in methanol, then remove the solvent under reduced pressure to obtain Oily Substance I.
- Dissolve Oily Substance I in dichloromethane and react with an aqueous alkaline solution (e.g., sodium carbonate) at 10-60°C to hydrolyze impurities, then separate the organic layer.
- Concentrate the organic phase to obtain Oily Substance II, then add a polar organic solvent like acetonitrile to crystallize and dry the final high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this refining technology translates directly into tangible operational efficiencies and cost optimizations. By eliminating the need for multiple recrystallization cycles, the process significantly reduces solvent consumption and waste generation, leading to a leaner and more environmentally compliant manufacturing footprint. The ability to achieve target purity in a single refining pass also shortens the overall production cycle time, allowing for faster turnaround on orders and improved responsiveness to market demand fluctuations. Furthermore, the use of common, industrially available solvents like methanol, dichloromethane, and acetonitrile ensures that the supply chain remains resilient and unaffected by shortages of exotic reagents.
- Cost Reduction in Manufacturing: The primary economic driver of this technology is the substantial reduction in processing steps. Traditional methods often require repeated dissolution and crystallization to chip away at impurity levels, which consumes vast amounts of energy and labor. By chemically destroying the impurities early in the refinement stage, this method consolidates the purification into a streamlined sequence. This consolidation leads to significant cost savings in utility usage, labor hours, and raw material consumption, ultimately lowering the cost of goods sold (COGS) for the final API without sacrificing quality standards.
- Enhanced Supply Chain Reliability: Consistency is paramount in pharmaceutical supply chains, and this method offers superior batch-to-batch reproducibility. The chemical specificity of the hydrolysis step ensures that variations in the crude input are normalized, resulting in a final product that consistently meets the stringent 99.7%-99.9% purity specification. This reliability reduces the risk of batch rejection and minimizes the need for safety stock, allowing procurement teams to operate with leaner inventory levels while maintaining confidence in supply continuity for critical cardiovascular medications.
- Scalability and Environmental Compliance: From a scale-up perspective, the process is highly robust, utilizing standard unit operations such as liquid-liquid extraction and crystallization that are easily transferable from pilot plant to commercial scale. Additionally, the reduction in solvent volume and the elimination of heavy metal catalysts or exotic reagents simplify waste treatment protocols. This aligns with modern green chemistry principles, reducing the environmental burden of the manufacturing process and ensuring compliance with increasingly strict global environmental regulations regarding pharmaceutical effluent.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this refining process. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing clarity on critical process parameters such as base selection, temperature control, and solvent choices. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this method into existing production lines.
Q: What specific impurities does the alkaline hydrolysis method target in Nicardipine Hydrochloride?
A: The method specifically targets polymer-like impurities (Formula I) generated during the esterification synthesis route. These impurities structurally resemble dimers or oligomers linked by ester bridges, which are difficult to remove via standard recrystallization.
Q: Why is dichloromethane used in conjunction with methanol in the refining process?
A: Methanol is initially used for its superior solubility to dissolve the crude product completely. However, dichloromethane is subsequently added because it facilitates effective phase separation with the aqueous alkaline solution, allowing the hydrolyzed impurities to be washed away while protecting the active pharmaceutical ingredient.
Q: What represents the optimal reaction temperature for the hydrolysis step?
A: While the process operates effectively between 10°C and 60°C, experimental data indicates that maintaining the reaction at approximately 40°C for about 2 hours provides the optimal balance between impurity degradation and product stability, ensuring maximum yield and purity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Nicardipine Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize that the complexity of modern API synthesis requires a partner with deep technical expertise and robust manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the alkaline hydrolysis refining method can be seamlessly translated from the laboratory to large-scale manufacturing. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify every batch against the highest international standards.
We invite pharmaceutical partners to collaborate with us to leverage these advanced purification technologies for your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized refining processes can enhance the quality and economics of your Nicardipine Hydrochloride supply.
