Scalable Manufacturing of High-Purity Diosmetin via Novel Oxidative Dehydrogenation Technology
Scalable Manufacturing of High-Purity Diosmetin via Novel Oxidative Dehydrogenation Technology
The global demand for high-purity flavonoids, particularly Diosmetin (CAS 520-34-3), has surged due to its potent antioxidant, anti-inflammatory, and venotonic properties, finding critical applications in pharmaceutical formulations, functional foods, and cosmeceuticals. However, securing a reliable supply of this valuable intermediate has historically been challenged by the limitations of natural extraction, which suffers from low yields and seasonal variability. A groundbreaking technical solution is presented in patent CN111100104A, which details a robust, cost-effective synthetic pathway converting Hesperetin into Diosmetin. This method represents a significant paradigm shift in fine chemical manufacturing, moving away from resource-intensive extraction toward a controlled, catalytic oxidative dehydrogenation process. By leveraging a unique Potassium Iodide (KI) and Dimethyl Sulfoxide (DMSO) catalytic system, this technology addresses the core pain points of purity, cost, and scalability that have long plagued the supply chain for this high-value flavonoid.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the procurement of Diosmetin relied heavily on isolation from natural sources such as citrus fruits or specific medicinal herbs. This approach is fundamentally flawed for industrial applications because the natural content of Diosmetin in plant matrices is exceptionally low, leading to exorbitant production costs and inconsistent batch-to-batch quality. Furthermore, alternative synthetic routes reported in prior art often depend on harsh reaction conditions, utilizing strong inorganic acids as hydrolysis media or expensive stoichiometric oxidants like DDQ (2,3-Dichloro-5,6-dicyano-1,4-benzoquinone). These conventional chemical methods frequently generate substantial amounts of acidic wastewater and require complex purification steps to remove toxic byproducts or residual heavy metals. The reliance on such aggressive reagents not only escalates the environmental compliance burden but also introduces significant risks of side reactions that degrade the sensitive flavonoid structure, ultimately compromising the final product's purity and biological activity.
The Novel Approach
The innovative methodology disclosed in the patent data circumvents these historical bottlenecks by employing a mild yet highly efficient oxidative system. Instead of harsh inorganic acids, the process utilizes a mixed solution of an organic acid compound (such as trifluoroacetic anhydride or p-toluenesulfonic acid) and DMSO, activated by trace amounts of Potassium Iodide. This catalytic system facilitates the selective dehydrogenation of the C2-C3 bond in the Hesperetin flavanone skeleton under moderate heating conditions (80°C to 120°C). The result is a streamlined workflow that eliminates the need for toxic heavy metal catalysts and minimizes the generation of hazardous waste. By optimizing the solvent system with Dimethyl Formamide (DMF) and implementing a straightforward workup procedure involving alkaline neutralization and acid reflux, this novel approach ensures high conversion rates while maintaining the integrity of the phenolic hydroxyl groups essential for the molecule's bioactivity.
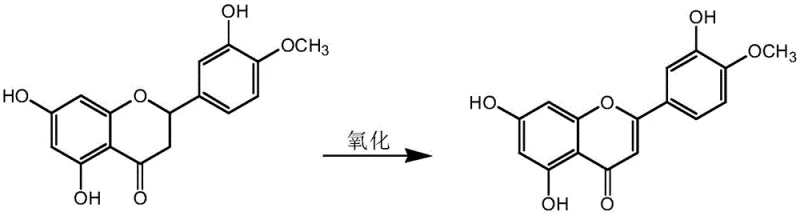
Mechanistic Insights into KI-Catalyzed Oxidative Dehydrogenation
At the heart of this synthesis lies a sophisticated yet elegant mechanistic pathway driven by the synergistic interaction between the iodide catalyst and the sulfoxide oxidant. The reaction initiates with the activation of DMSO by the acidic component, generating a highly reactive sulfonium species. The trace Potassium Iodide serves as a nucleophilic catalyst, facilitating the transfer of oxygen to the substrate. Specifically, the mechanism targets the saturated C2-C3 bond of the flavanone ring in Hesperetin. Through a sequence of proton abstraction and hydride transfer steps, the system effectively removes two hydrogen atoms, establishing the conjugated double bond characteristic of the flavone structure found in Diosmetin. This oxidative dehydrogenation is thermodynamically favorable under the specified thermal conditions (70°C to 150°C), allowing the reaction to proceed to completion with minimal formation of over-oxidized byproducts or degradation of the methoxy and hydroxyl substituents on the aromatic rings.
From an impurity control perspective, this mechanism offers distinct advantages over radical-based oxidation methods. The ionic nature of the catalytic cycle ensures high regioselectivity, preventing unwanted oxidation at the electron-rich phenolic positions (3', 5, and 7-hydroxyl groups). The subsequent workup steps are designed to leverage the solubility differences between the product and impurities. The addition of an alkaline solution (ammonia or sodium bicarbonate) neutralizes the acidic reaction medium and precipitates inorganic salts, which are removed via filtration. The following acid reflux step with dilute hydrochloric acid serves a dual purpose: it hydrolyzes any transient intermediates and induces the crystallization of the crude Diosmetin. This multi-stage purification strategy, culminating in ethanol recrystallization, consistently delivers a final product with a purity exceeding 98%, meeting the stringent specifications required for pharmaceutical grade intermediates.
How to Synthesize Diosmetin Efficiently
Implementing this synthesis route requires precise control over reaction parameters to maximize yield and minimize impurity profiles. The process is divided into three critical stages: the oxidative reaction, the isolation of the crude product, and the final purification. Operators must maintain strict temperature control during the dropwise addition of the oxidant mixture to prevent thermal runaway, ensuring the reaction proceeds smoothly over a 2 to 6-hour period. The detailed standardized operating procedures, including specific mass ratios of Hesperetin to DMF (1:2 to 1:5) and catalyst loading (0.005 to 0.05 equivalents), are critical for reproducibility. For a comprehensive breakdown of the exact operational steps and safety protocols required for GMP-compliant manufacturing, please refer to the technical guide below.
- Dissolve Hesperetin in DMF, add trace Potassium Iodide catalyst, and react with a DMSO/Acid mixture at 80-120°C to effect oxidative dehydrogenation.
- Neutralize the reaction mixture with alkaline solution, filter, recover solvent, and perform acid reflux with hydrochloric acid to isolate crude Diosmetin.
- Purify the crude product via ethanol recrystallization, involving dissolution, hot filtration, solvent recovery, cooling, and drying to achieve >98% purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this synthetic route offers transformative economic and logistical benefits. The primary driver of cost reduction is the substitution of expensive, scarce natural extracts with Hesperetin, a derivative of Hesperidin, which is abundantly available as a byproduct of the citrus juice industry. This shift decouples production from agricultural volatility, ensuring a stable, year-round supply of raw materials. Furthermore, the elimination of precious metal catalysts removes the need for costly scavenging resins and extensive analytical testing for heavy metal residues, significantly lowering the Cost of Goods Sold (COGS). The simplified downstream processing, characterized by standard filtration and solvent recovery steps, reduces energy consumption and labor hours, contributing to substantial overall manufacturing efficiency.
- Cost Reduction in Manufacturing: The economic viability of this process is underpinned by the use of commodity chemicals such as DMF, DMSO, and Potassium Iodide, all of which are readily available in the global bulk chemical market at competitive prices. By avoiding the use of stoichiometric amounts of expensive oxidants like DDQ or hypervalent iodine reagents, the raw material cost per kilogram of Diosmetin is drastically reduced. Additionally, the recovery and reuse of solvents like DMF and ethanol further enhance the process economics, creating a lean manufacturing model that allows for aggressive pricing strategies without compromising margin.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly improved by the robustness of the synthetic pathway. Unlike extraction processes that are susceptible to crop failures or seasonal fluctuations in flavonoid content, this chemical synthesis can be ramped up or down based on market demand with short lead times. The use of stable, non-hazardous reagents simplifies logistics and storage requirements, reducing the risk of supply disruptions caused by regulatory restrictions on hazardous materials. This reliability makes it an ideal solution for long-term supply agreements with major pharmaceutical and nutraceutical clients who require guaranteed continuity of supply.
- Scalability and Environmental Compliance: The process is inherently scalable, having been successfully demonstrated in reactor volumes ranging from 500L to 1000L in the patent examples. The absence of heavy metals and the use of a closed-loop solvent recovery system align perfectly with modern Green Chemistry principles and stringent environmental regulations. This reduces the burden of wastewater treatment and hazardous waste disposal, lowering the environmental compliance costs associated with production. The simplicity of the unit operations ensures that technology transfer to larger commercial scales (multi-ton) can be achieved rapidly with minimal engineering hurdles.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of Diosmetin synthesized via this advanced oxidative method. These insights are derived directly from the experimental data and technical specifications outlined in the patent literature, providing a transparent view of the process capabilities. Understanding these details is crucial for R&D teams evaluating route feasibility and procurement officers assessing supplier qualifications.
Q: What are the primary advantages of this synthetic route over natural extraction?
A: Natural extraction yields are extremely low due to the sparse distribution of Diosmetin in plants. This synthetic route utilizes abundant Hesperetin (derived from citrus waste Hesperidin) as a starting material, ensuring consistent supply, higher purity (>98%), and significantly lower production costs suitable for industrial scale.
Q: Does this process utilize hazardous heavy metal catalysts?
A: No. Unlike traditional methods that may rely on expensive or toxic transition metal oxidants, this patented process employs trace amounts of Potassium Iodide (KI) as a catalyst and a DMSO-based system as the oxidant. This eliminates the need for complex heavy metal removal steps, simplifying downstream processing and reducing environmental impact.
Q: Is this synthesis method scalable for commercial tonnage production?
A: Yes. The patent explicitly demonstrates scalability with examples ranging from 500L to 1000L reaction vessels. The process involves standard unit operations such as heating, filtration, solvent recovery, and recrystallization, making it highly adaptable for large-scale commercial manufacturing without requiring specialized high-pressure equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Diosmetin Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of high-value flavonoids depends on more than just a laboratory recipe; it requires a partner with deep process engineering expertise and a commitment to quality. Our technical team has extensively analyzed this KI-catalyzed oxidative pathway and possesses the capability to optimize it for industrial throughput. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot scale to full manufacturing is seamless. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of Diosmetin meets the exacting standards required for pharmaceutical and cosmetic applications.
We invite you to collaborate with us to secure a sustainable and cost-effective supply of this critical intermediate. Whether you are looking to reduce your current procurement costs or need a backup supplier to mitigate supply chain risks, our team is ready to assist. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can support your product development goals.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
