Advanced Manufacturing of N-Benzylhydroxylamine Hydrochloride via Safe Oxidation and In-Situ Crystallization
Introduction to Patent CN113292446B
The pharmaceutical industry constantly seeks robust synthetic routes for critical intermediates like N-benzylhydroxylamine hydrochloride, a key building block for constructing isoxazoline rings found in bioactive molecules such as the antiplatelet drug ticagrelor. Patent CN113292446B introduces a transformative methodology that addresses longstanding safety and efficiency bottlenecks in the production of this valuable compound. By shifting from traditional volatile solvents to high-flash-point alternatives and implementing a novel dual-oxidation strategy, this technology achieves a substantial leap in both operational safety and chemical yield. The innovation lies not merely in changing reagents but in a fundamental re-engineering of the reaction pathway to capture intermediates that were previously lost as waste. For global supply chain leaders and R&D directors, this patent represents a viable pathway to secure, high-volume manufacturing of high-purity pharmaceutical intermediates with reduced environmental and safety liabilities.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of N-benzylhydroxylamine hydrochloride has been plagued by significant economic and safety drawbacks that hinder scalable commercial production. Early methods, such as those described in CN101429136A, relied on the reduction of benzaldehyde oximes using sodium cyanoborohydride (NaBH3CN), a reagent that is not only prohibitively expensive for large-scale operations but also introduces toxic cyanide handling risks. Subsequent improvements attempted to utilize dibenzylamine oxidation with hydrogen peroxide in methanol, as seen in CN104529814A; however, these processes suffered from inherent instability due to the low flash point of methanol (11°C), creating a severe flash explosion hazard during the exothermic oxidation phase. Furthermore, these conventional oxidative routes typically stalled at conversion rates yielding only 60% to 65% of the desired nitrone intermediate, leaving nearly 40% of the starting material as difficult-to-separate impurities or unreacted intermediates, which drastically inflated the cost of goods sold and complicated downstream purification workflows.
The Novel Approach
The methodology disclosed in CN113292446B fundamentally resolves these issues through a sophisticated two-stage oxidation protocol coupled with a strategic solvent switch. Instead of hazardous methanol, the process utilizes ethylene glycol, which boasts a flash point of 111°C, effectively eliminating the risk of flash explosions during the critical hydrogen peroxide addition. More importantly, the inventors identified that the crude reaction mixture contained approximately 30% of an intermediate species, N,N-dibenzylhydroxylamine, which was previously discarded. By introducing a secondary oxidation step using sodium hypochlorite (NaClO), this intermediate is quantitatively converted into the desired C-phenyl-N-benzyl nitrone, boosting the overall yield by a remarkable 20-25%. This approach transforms a linear, loss-prone process into a convergent, high-efficiency system that maximizes atom economy while ensuring the safety required for multi-ton manufacturing.

Mechanistic Insights into Tungsten-Catalyzed Oxidation and Impurity Conversion
The core of this technological breakthrough relies on a precise tungsten-catalyzed oxidation mechanism that operates under mild yet effective conditions. In the initial step, dibenzylamine is treated with hydrogen peroxide in the presence of a sodium tungstate catalyst within an ethylene glycol medium. The tungsten species likely forms a peroxo-tungstate complex that acts as the active oxygen transfer agent, selectively oxidizing the amine to the nitrone functionality. However, kinetic analysis revealed that this oxidation is not absolute under standard conditions, leading to the accumulation of the N,N-dibenzylhydroxylamine intermediate. Rather than viewing this as a failure, the new process leverages this chemical behavior by introducing a second oxidant, sodium hypochlorite, in a dichloromethane phase. This secondary oxidation specifically targets the hydroxylamine impurity, driving the equilibrium towards the fully oxidized nitrone product. This dual-oxidant strategy ensures that the crude nitrone obtained possesses a purity of over 98%, significantly reducing the burden on subsequent purification steps and minimizing the formation of complex byproduct profiles that often plague amine oxidations.
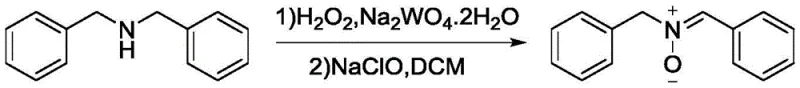
Following the high-yield generation of the nitrone intermediate, the process employs a unique crystallization-driven reduction to secure final product quality. The nitrone is reacted with hydroxylamine hydrochloride in a carefully balanced mixture of ethanol and methyl tert-butyl ether (MTBE). The solubility parameters of this solvent system are tuned such that the reactants remain in solution while the product, N-benzylhydroxylamine hydrochloride, precipitates out as needle-like crystals immediately upon formation. This phenomenon, known as reactive crystallization or in-situ crystallization, serves a dual purpose: it drives the reaction equilibrium forward by removing the product from the solution phase, and it acts as an intrinsic purification step. The resulting crystals exclude soluble impurities, achieving an initial purity of 98.2% to 98.5% without the need for energy-intensive recrystallization. A final slurry with dichloromethane further refines the crystal lattice, pushing the purity to ≥99.0%, which is critical for pharmaceutical applications where impurity profiles must be strictly controlled to meet regulatory standards.
How to Synthesize N-Benzylhydroxylamine Hydrochloride Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for transitioning from laboratory discovery to commercial manufacturing, emphasizing temperature control and phase management. The process begins with the careful mixing of dibenzylamine, ethylene glycol, and the tungsten catalyst, followed by a controlled temperature drop to 0°C to manage the exotherm of hydrogen peroxide addition. After the initial oxidation and subsequent hypochlorite treatment to maximize nitrone yield, the organic phase is isolated and concentrated. The critical second stage involves dissolving this concentrate in the ethanol/MTBE mixture and reacting with hydroxylamine hydrochloride at 35°C. The detailed standardized synthesis steps, including specific molar ratios, stirring speeds, and filtration parameters required for GMP-compliant production, are provided in the technical guide below.
- Oxidize dibenzylamine using hydrogen peroxide and sodium tungstate in ethylene glycol at controlled temperatures to form C-phenyl-N-benzyl nitrone.
- Treat the crude nitrone with sodium hypochlorite to convert residual N,N-dibenzylhydroxylamine intermediates into the desired nitrone product.
- React the purified nitrone with hydroxylamine hydrochloride in an ethanol/MTBE mixture to induce in-situ crystallization of the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology offers profound advantages in terms of cost stability and logistical safety. The elimination of expensive reducing agents like sodium cyanoborohydride and the replacement of hazardous methanol with high-flash-point ethylene glycol directly translate to lower raw material costs and reduced insurance premiums associated with flammable solvent storage. Furthermore, the significant yield improvement of 20-25% means that less starting material is required to produce the same amount of final product, effectively lowering the cost per kilogram and reducing the volume of chemical waste that requires disposal. This efficiency gain is particularly vital in the current market environment where raw material price volatility can severely impact margins, making processes with high atom economy and minimal waste generation far more attractive for long-term contracts.
- Cost Reduction in Manufacturing: The process eliminates the need for costly noble metal catalysts or expensive hydride reducers, relying instead on abundant and inexpensive tungsten salts and bleach. By converting what was previously a 30% waste stream (the N,N-dibenzylhydroxylamine intermediate) into valuable product, the effective cost of goods is drastically reduced. This optimization allows manufacturers to offer competitive pricing for high-purity pharmaceutical intermediates while maintaining healthy margins, providing a distinct advantage in tender negotiations for bulk API ingredients.
- Enhanced Supply Chain Reliability: Switching from methanol to ethylene glycol mitigates the risks associated with transporting and storing highly flammable Class 3 liquids, simplifying logistics and warehousing requirements. The robustness of the reaction conditions, which tolerate slight variations in temperature without catastrophic failure, ensures consistent batch-to-batch quality. This reliability reduces the frequency of failed batches and production delays, ensuring a steady flow of materials to downstream API manufacturers and preventing costly stoppages in the broader pharmaceutical supply chain.
- Scalability and Environmental Compliance: The in-situ crystallization technique simplifies the isolation process, removing the need for complex distillation or multiple recrystallization steps that consume large amounts of energy and solvent. The use of sodium hypochlorite as a secondary oxidant generates benign salt byproducts, easing the burden on wastewater treatment facilities compared to processes generating heavy metal waste or cyanide residues. This alignment with green chemistry principles facilitates easier regulatory approval and supports corporate sustainability goals, making the facility more resilient to tightening environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this high-yield synthesis route. These answers are derived directly from the experimental data and beneficial effects described in patent CN113292446B, providing clarity on how this technology compares to legacy methods. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this process for their own manufacturing lines or for procurement teams assessing the quality claims of potential suppliers.
Q: Why is ethylene glycol preferred over methanol in this synthesis?
A: Ethylene glycol has a significantly higher flash point (111°C) compared to methanol (11°C), drastically reducing the risk of flash explosions during the exothermic hydrogen peroxide oxidation step, thereby enhancing industrial safety.
Q: How does the new method improve yield compared to conventional routes?
A: The process identifies that approximately 30% of the intermediate remains as unconverted N,N-dibenzylhydroxylamine. By introducing a sodium hypochlorite treatment step, this impurity is oxidized into the desired nitrone, increasing total yield by 20-25%.
Q: What purity levels can be achieved without recrystallization?
A: Through the use of an ethanol/methyl tert-butyl ether mixed solvent system, the product crystallizes directly from the reaction mixture as needle crystals, achieving purities of 98.2-98.5% initially and over 99.0% after a simple dichloromethane slurry.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Benzylhydroxylamine Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires deep technical expertise and rigorous process control. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this high-yield oxidation route are fully realized in practice. Our state-of-the-art facilities are equipped to handle the specific solvent systems and temperature controls required for this synthesis, and our stringent purity specifications are enforced through rigorous QC labs that utilize advanced HPLC and NMR techniques to verify every batch. We are committed to delivering N-benzylhydroxylamine hydrochloride that meets the highest international standards, supporting your drug development timelines with reliable, high-quality intermediates.
We invite you to engage with our technical procurement team to discuss how this advanced manufacturing route can optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic benefits of switching to this safer, higher-yield process for your specific application. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that enhance both the quality and profitability of your pharmaceutical manufacturing operations.
