Scalable Synthesis of 2-Hydroxy-4-Amino-5-Methylpyridine: A Cost-Effective Route for Finerenone Intermediates
The pharmaceutical industry is constantly seeking robust and scalable pathways for critical API intermediates, particularly for emerging therapies like Finerenone (BAY 94-8862), a non-steroidal selective mineralocorticoid receptor antagonist. Patent CN115340492A introduces a groundbreaking preparation method for 2-hydroxy-4-amino-5-methylpyridine, a pivotal building block in the synthesis of this life-saving medication. This novel methodology addresses the historical challenges associated with synthesizing this specific pyridine derivative, which has traditionally been plagued by complex multi-step sequences and prohibitive costs. By leveraging a strategic N-oxide activation approach, the disclosed process achieves high regioselectivity and operational simplicity, offering a compelling value proposition for global supply chains. The route is characterized by mild reaction conditions, the use of readily available industrial reagents, and a significant reduction in environmental impact compared to legacy methods. For R&D directors and procurement specialists alike, this patent represents a viable pathway to secure a stable supply of high-purity intermediates essential for the treatment of chronic kidney disease and cardiovascular impairments in diabetic patients.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of substituted aminopyridines has presented formidable challenges regarding regiocontrol and functional group tolerance. Conventional routes often rely on harsh halogenation conditions or expensive transition metal-catalyzed cross-coupling reactions that require stringent anhydrous environments and inert atmospheres. These traditional methods frequently suffer from poor atom economy, generating substantial quantities of hazardous waste streams that complicate disposal and increase the overall carbon footprint of the manufacturing process. Furthermore, the introduction of substituents at the 4-position of the pyridine ring is notoriously difficult due to the electron-deficient nature of the heterocycle, often leading to mixtures of isomers that require costly and yield-eroding purification steps. The reliance on precious metal catalysts not only inflates the raw material costs but also introduces the risk of heavy metal contamination in the final product, necessitating additional downstream processing to meet rigorous pharmaceutical purity specifications. These cumulative inefficiencies have historically resulted in a fragile supply chain for 2-hydroxy-4-amino-5-methylpyridine, characterized by long lead times and price volatility that threatens the commercial viability of downstream drug products.
The Novel Approach
In stark contrast to these legacy limitations, the methodology described in patent CN115340492A employs a clever N-oxide activation strategy that fundamentally alters the electronic properties of the pyridine ring to facilitate efficient functionalization. By initially oxidizing 2-chloro-5-methylpyridine to its corresponding N-oxide, the process activates the ring towards electrophilic aromatic substitution, allowing for the highly regioselective introduction of a nitro group at the desired 4-position using standard nitrating agents. This approach bypasses the need for complex directing groups or exotic catalysts, utilizing instead common industrial acids like sulfuric and nitric acid under controlled thermal conditions. The subsequent reduction step is equally innovative, as it simultaneously reduces both the nitro group and the N-oxide functionality back to the amine and pyridine forms, respectively, streamlining the synthesis into fewer operational units. This telescoping of chemical transformations not only reduces the total number of isolation steps but also minimizes solvent consumption and waste generation. The final hydrolysis step utilizes strong bases in high-boiling solvents like ethylene glycol to effect the displacement of the chloro group, completing the synthesis with remarkable efficiency and yielding a product that is exceptionally well-suited for further elaboration into Finerenone.
Mechanistic Insights into N-Oxide Activated Regioselective Nitration
The core chemical innovation of this process lies in the manipulation of the pyridine ring's electron density through N-oxidation. In its native state, the pyridine ring is electron-deficient due to the electronegative nitrogen atom, making it resistant to electrophilic attack and prone to nucleophilic substitution primarily at the 2- and 4-positions under extreme conditions. However, upon oxidation to the N-oxide, the oxygen atom donates electron density into the ring system through resonance, significantly activating the 4-position towards electrophilic substitution. This electronic modulation allows the nitration reaction to proceed with high specificity, ensuring that the nitro group is installed exclusively at the 4-position relative to the nitrogen, while the chloro and methyl groups direct further reactivity. This mechanistic understanding is crucial for R&D teams aiming to replicate or optimize the process, as it highlights the importance of maintaining the integrity of the N-oxide intermediate prior to nitration to avoid the formation of undesired regioisomers.
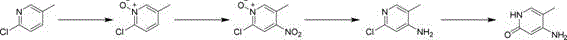
Following the nitration, the reduction step serves a dual purpose, acting as a deoxygenation of the N-oxide and a reduction of the nitro group to an amine. The use of iron powder in acetic acid provides a source of electrons that facilitates this dual transformation efficiently. The mechanism likely involves the formation of iron complexes that transfer electrons to the nitrogen-oxygen bonds, cleaving them to restore the aromatic pyridine system while converting the nitro group to the amino functionality. This step is critical for impurity control, as incomplete reduction could lead to hydroxylamine intermediates or residual N-oxides that are difficult to separate. The final substitution of the chloro group with a hydroxyl group proceeds via a nucleophilic aromatic substitution mechanism, facilitated by the electron-withdrawing nature of the adjacent amino group and the high temperature provided by the ethylene glycol solvent. This sequence ensures that the final product, 2-hydroxy-4-amino-5-methylpyridine, is obtained with high structural fidelity and minimal byproduct formation, meeting the stringent quality requirements for pharmaceutical intermediates.
How to Synthesize 2-Hydroxy-4-Amino-5-Methylpyridine Efficiently
The synthesis of this critical intermediate is achieved through a logical four-step sequence that balances chemical efficiency with operational practicality. The process begins with the oxidation of the starting material, followed by regioselective nitration, simultaneous reduction, and final hydrolysis. Each step has been optimized to maximize yield and minimize the formation of difficult-to-remove impurities. For detailed operational parameters, including specific stoichiometry, temperature profiles, and workup procedures, please refer to the standardized synthesis guide below which outlines the exact protocol derived from the patent examples.
- Oxidize 2-chloro-5-methylpyridine using m-CPBA or hydrogen peroxide to form the N-oxide derivative.
- Perform regioselective nitration using sulfuric and nitric acid at elevated temperatures to introduce the nitro group at the 4-position.
- Reduce both the nitro group and the N-oxide simultaneously using iron powder or catalytic hydrogenation in acetic acid.
- Execute nucleophilic substitution with strong base (KOH) in ethylene glycol to replace the chloro group with a hydroxyl group.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented route offers substantial advantages that directly address the pain points of procurement managers and supply chain directors. The primary benefit stems from the utilization of commodity chemicals as reagents, such as m-CPBA, sulfuric acid, nitric acid, and iron powder, which are globally available and subject to minimal price volatility compared to specialized organometallic catalysts. This reliance on bulk chemicals insulates the manufacturing process from supply shocks and ensures a consistent cost base, allowing for more accurate long-term budgeting and pricing strategies for the final API. Furthermore, the elimination of precious metals like palladium or platinum from the reduction step removes a significant cost driver and simplifies the regulatory burden associated with heavy metal limits in drug substances. The process design inherently supports green chemistry principles by reducing the number of unit operations and avoiding the generation of toxic heavy metal waste streams, which translates to lower waste disposal costs and a reduced environmental compliance burden for manufacturing sites.
- Cost Reduction in Manufacturing: The economic viability of this route is underpinned by the high overall yield and the low cost of goods sold (COGS) associated with the reagents. By avoiding expensive catalytic systems and utilizing high-yielding transformations, the process minimizes the amount of starting material required per kilogram of final product. The ability to perform the reduction using iron powder rather than catalytic hydrogenation or hydride reagents represents a significant saving in both reagent costs and equipment capital expenditure, as high-pressure hydrogenation reactors are not required. Additionally, the use of ethylene glycol as a solvent for the final step allows for high-temperature reactions without the need for pressurized vessels, further reducing infrastructure costs. These factors combine to create a manufacturing process that is inherently lean and cost-effective, providing a competitive edge in the marketplace for Finerenone intermediates.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the use of widely available raw materials that are not subject to geopolitical constraints or single-source dependencies. 2-Chloro-5-methylpyridine is a mature chemical commodity with multiple global suppliers, ensuring that production can be scaled up rapidly to meet surging demand without bottlenecking at the raw material stage. The robustness of the chemical steps, which tolerate minor variations in conditions without catastrophic failure, adds another layer of reliability, reducing the risk of batch failures and production delays. This stability allows supply chain planners to maintain lower safety stock levels while still ensuring continuity of supply, optimizing working capital and reducing inventory holding costs. The simplified purification requirements also mean that cycle times are shorter, enabling faster turnover and more responsive delivery schedules to downstream API manufacturers.
- Scalability and Environmental Compliance: The process is designed with scale-up in mind, utilizing reaction conditions that are easily transferable from laboratory to pilot and commercial scales. The exothermic nature of the oxidation and nitration steps is manageable with standard cooling systems, and the heterogeneous reduction step is straightforward to filter and work up on a large scale. From an environmental standpoint, the absence of heavy metal catalysts simplifies wastewater treatment and reduces the toxicity of solid waste, aligning with increasingly stringent global environmental regulations. The high atom economy of the nitration and reduction steps minimizes the volume of organic solvents required, further reducing the facility's environmental footprint. This alignment with sustainability goals not only mitigates regulatory risk but also enhances the brand value of the supply chain partners by supporting the production of greener pharmaceuticals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of 2-hydroxy-4-amino-5-methylpyridine. These answers are derived directly from the technical specifications and advantageous effects detailed in the patent literature, providing clarity on the feasibility and benefits of this specific synthetic route for potential partners and stakeholders.
Q: How does the N-oxide strategy improve regioselectivity in this synthesis?
A: The formation of the pyridine N-oxide activates the ring towards electrophilic substitution, specifically directing the nitro group to the 4-position, which is otherwise difficult to achieve on the unoxidized pyridine ring.
Q: What are the cost advantages of using iron powder for reduction?
A: Using iron powder as a reducing agent eliminates the need for expensive precious metal catalysts like palladium, significantly lowering raw material costs and simplifying the removal of metal residues from the final API intermediate.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process utilizes common industrial reagents like sulfuric acid, nitric acid, and ethylene glycol, and operates at manageable temperatures, making it highly scalable for metric-ton production without requiring specialized high-pressure equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Hydroxy-4-Amino-5-Methylpyridine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development and commercialization of novel therapeutics like Finerenone. Our team of expert chemists has thoroughly analyzed the pathway described in CN115340492A and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this process to fruition. We are committed to delivering 2-hydroxy-4-amino-5-methylpyridine with stringent purity specifications, utilizing our rigorous QC labs to ensure that every batch meets the exacting standards required for GMP manufacturing. Our facility is equipped to handle the specific reagents and conditions of this N-oxide route safely and efficiently, ensuring a seamless transition from process development to full-scale supply.
We invite pharmaceutical companies and contract manufacturers to collaborate with us to leverage this cost-effective and scalable technology. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can optimize your supply chain for this vital pharmaceutical intermediate.
