Advanced Synthesis of 3-(2,3-Dihydrobenzofuran-5-yl)propionic Acid for Commercial API Manufacturing
The pharmaceutical landscape for melatonin receptor agonists relies heavily on the efficient synthesis of key intermediates like 3-(2,3-dihydrobenzofuran-5-yl)propionic acid, a critical precursor for the sleep disorder therapeutic Ramelteon. Patent CN101648931A introduces a transformative methodology that shifts the synthetic paradigm from expensive, specialized starting materials to commodity chemicals like p-cresol, fundamentally altering the economic viability of this supply chain. This strategic pivot addresses critical bottlenecks in the production of complex heterocycles, offering a pathway that is not only chemically robust but also economically superior for large-scale manufacturing operations. By leveraging classic organic transformations such as Friedel-Crafts cyclization and catalytic hydrogenation, the disclosed process minimizes environmental impact while maximizing yield through optimized reaction conditions. For R&D directors and procurement specialists, understanding this route is essential for securing a competitive edge in the production of high-value pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Conventional syntheses, as illustrated in the prior art reaction schemes, typically commence with 2,3-dihydrobenzofuran-5-carbaldehyde, a substrate that commands a premium price due to its complex preparation requirements and limited commercial availability. 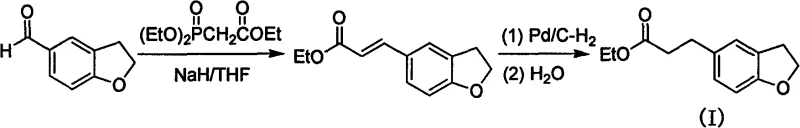 . These legacy methods often necessitate harsh basic conditions using reagents like sodium hydride or potassium tert-butoxide, which pose significant safety hazards and complicate waste disposal protocols on an industrial scale due to the generation of reactive byproducts. Furthermore, the reliance on phosphonate esters for chain extension introduces additional stoichiometric waste streams that require rigorous downstream processing to remove phosphorus residues, increasing both operational complexity and cost. The cumulative effect of these factors results in a manufacturing process that is fragile, expensive, and difficult to scale without incurring substantial penalties in yield and purity.
. These legacy methods often necessitate harsh basic conditions using reagents like sodium hydride or potassium tert-butoxide, which pose significant safety hazards and complicate waste disposal protocols on an industrial scale due to the generation of reactive byproducts. Furthermore, the reliance on phosphonate esters for chain extension introduces additional stoichiometric waste streams that require rigorous downstream processing to remove phosphorus residues, increasing both operational complexity and cost. The cumulative effect of these factors results in a manufacturing process that is fragile, expensive, and difficult to scale without incurring substantial penalties in yield and purity.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes p-cresol, a ubiquitous and low-cost petrochemical derivative, as the foundational building block for constructing the entire molecular architecture. 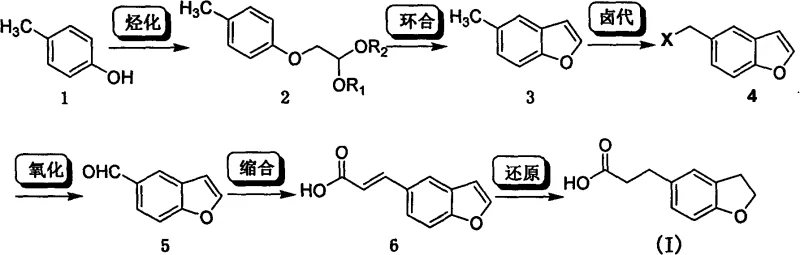 . This new trajectory constructs the benzofuran core in situ through an intramolecular Friedel-Crafts reaction, effectively bypassing the need for pre-functionalized heterocyclic aldehydes and drastically simplifying the overall material flow. The route employs a logical sequence of etherification, cyclization, halogenation, and oxidation, allowing for precise control over the substitution pattern on the aromatic ring. By deferring the formation of the sensitive dihydrobenzofuran ring until the final hydrogenation step, the process avoids the stability issues associated with handling reduced heterocycles in early stages, thereby enhancing overall process robustness and operator safety.
. This new trajectory constructs the benzofuran core in situ through an intramolecular Friedel-Crafts reaction, effectively bypassing the need for pre-functionalized heterocyclic aldehydes and drastically simplifying the overall material flow. The route employs a logical sequence of etherification, cyclization, halogenation, and oxidation, allowing for precise control over the substitution pattern on the aromatic ring. By deferring the formation of the sensitive dihydrobenzofuran ring until the final hydrogenation step, the process avoids the stability issues associated with handling reduced heterocycles in early stages, thereby enhancing overall process robustness and operator safety.
Mechanistic Insights into Friedel-Crafts Cyclization and Hydrogenation
The heart of this synthetic innovation lies in the precise execution of the Friedel-Crafts cyclization, where a 2-(4-methylphenoxy)acetaldehyde derivative undergoes ring closure under Lewis or Brønsted acid catalysis to form the benzofuran skeleton. This step is critical for establishing the core structure with high regioselectivity, ensuring that the methyl group is positioned correctly for subsequent functionalization at the 5-position without forming unwanted isomers. Following halogenation and oxidation to generate the aldehyde moiety using reagents like DMSO or hexamethylenetetramine, the process employs a Knoevenagel condensation with malonic acid, a reaction known for its atom economy and ease of purification compared to phosphonate-based alternatives. The choice of acid catalyst, ranging from polyphosphoric acid to boron trifluoride etherate, allows for fine-tuning of the reaction kinetics to minimize polymerization side reactions.
The final transformation involves a catalytic hydrogenation using palladium on carbon or Raney nickel, which simultaneously reduces the olefinic double bond and saturates the furan ring to yield the dihydrobenzofuran system. This dual-reduction capability is particularly advantageous as it consolidates two potential steps into one operational unit, thereby reducing reactor occupancy time and solvent consumption significantly. Impurity profiles are tightly controlled through the crystallization of the acrylic acid intermediate, which serves as an effective purge point for non-volatile byproducts before the final reduction step. The use of mild hydrogenation conditions ensures that the carboxylic acid functionality remains intact while achieving complete saturation of the carbon-carbon double bond, resulting in a high-purity final product suitable for direct use in API synthesis.
How to Synthesize 3-(2,3-Dihydrobenzofuran-5-yl)propionic Acid Efficiently
The synthesis of this valuable intermediate begins with the etherification of p-cresol followed by a cascade of functional group transformations that build complexity efficiently. The detailed standardized synthesis steps involve specific molar ratios and temperature controls to ensure maximum conversion at each stage, from the initial alkylation to the final catalytic reduction. Operators must pay close attention to the purification of the benzofuran-5-carbaldehyde intermediate, as its quality directly impacts the yield of the subsequent condensation reaction. For a comprehensive breakdown of the exact reaction parameters, solvent choices, and workup procedures required to replicate this high-yielding process, please refer to the structured guide below.
- Condense p-cresol with 2-haloacetaldehyde under alkaline conditions to form the ether intermediate.
- Perform Friedel-Crafts cyclization using Lewis or Brønsted acid to construct the 5-methylbenzofuran core.
- Oxidize the halogenated intermediate to an aldehyde, condense with malonic acid, and finalize via catalytic hydrogenation.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the substitution of specialized heterocyclic aldehydes with p-cresol represents a monumental shift in cost structure, decoupling production expenses from the volatile pricing of niche fine chemicals. The elimination of phosphonate reagents and strong bases like sodium hydride further contributes to substantial cost savings by reducing the burden on waste treatment facilities and lowering the requirement for specialized corrosion-resistant equipment. Supply chain reliability is significantly enhanced because p-cresol is produced in massive volumes globally, ensuring consistent availability even during market fluctuations that might affect smaller volume intermediates. The process design inherently supports scalability, as the reaction conditions ranging from ambient temperature etherification to moderate pressure hydrogenation are well within the operational parameters of standard multipurpose chemical plants. Environmental compliance is streamlined due to the generation of fewer hazardous byproducts, aligning with increasingly stringent global regulations on pharmaceutical manufacturing emissions and sustainability goals.
- Cost Reduction in Manufacturing: The utilization of commodity raw materials eliminates the markup associated with custom-synthesized starting materials, leading to a drastic reduction in the overall cost of goods sold. By avoiding expensive coupling reagents and simplifying the purification workflow, the process minimizes solvent usage and energy consumption per kilogram of product. This efficiency translates directly into improved margins for manufacturers and more competitive pricing for downstream API producers seeking reliable sources.
- Enhanced Supply Chain Reliability: Sourcing strategies are simplified as the primary feedstock is a widely available industrial chemical with a robust global supply network. This reduces the risk of production stoppages caused by shortages of exotic reagents, ensuring continuous operation and on-time delivery for critical pharmaceutical projects. The resilience of the supply chain is further bolstered by the use of common catalysts and solvents that can be sourced from multiple vendors without compromising quality.
- Scalability and Environmental Compliance: The synthetic route is designed with green chemistry principles in mind, generating minimal waste and utilizing recyclable solvents where possible. This facilitates easier permitting and regulatory approval for new manufacturing sites, accelerating the time to market for commercial production. The ability to scale from pilot plant to multi-ton production without significant process re-engineering makes this an ideal candidate for long-term supply agreements.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route for 3-(2,3-dihydrobenzofuran-5-yl)propionic acid. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on process capabilities and limitations. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this methodology for their specific manufacturing needs. For further technical discussions or custom optimization requests, our team is available to provide deeper insights.
Q: What are the primary cost advantages of this new synthesis route?
A: The primary advantage is the replacement of expensive 2,3-dihydrobenzofuran-5-carbaldehyde with commodity p-cresol, significantly lowering raw material costs and simplifying the supply chain.
Q: How does this method improve environmental compliance?
A: By avoiding phosphonate reagents and strong bases like sodium hydride, the process generates less hazardous waste and reduces the burden on wastewater treatment systems.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the reaction conditions utilize standard solvents and catalysts like Pd/C or Raney Ni, making it highly scalable for multi-ton annual production in existing facilities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-(2,3-Dihydrobenzofuran-5-yl)propionic Acid Supplier
Partnering with NINGBO INNO PHARMCHEM provides access to this advanced synthetic technology, positioning your organization as a leader in the efficient production of Ramelteon intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and risk-mitigated. We maintain stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of 3-(2,3-dihydrobenzofuran-5-yl)propionic acid meets the exacting standards required for GMP pharmaceutical applications. Our commitment to quality and consistency makes us the preferred partner for global pharmaceutical companies seeking to optimize their supply chains.
We invite you to contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements and production timelines. By collaborating with us, you can secure specific COA data and route feasibility assessments that demonstrate the tangible economic benefits of adopting this superior manufacturing process. Let us help you achieve cost reduction in pharmaceutical intermediate manufacturing while ensuring the highest levels of quality and supply security for your critical drug development programs.
