Advanced 3-Step Synthesis of FR20 Inhibitor for Scalable Pharmaceutical Manufacturing
Advanced 3-Step Synthesis of FR20 Inhibitor for Scalable Pharmaceutical Manufacturing
The pharmaceutical industry is constantly seeking more efficient and environmentally benign pathways for synthesizing complex therapeutic agents, particularly those targeting inflammatory pathways. Patent CN107793367B discloses a groundbreaking method for synthesizing FR20, a potent human microsomal prostaglandin synthase 1 (mPGES-1) inhibitor, which represents a significant advancement over prior art. Unlike traditional non-steroidal anti-inflammatory drugs (NSAIDs) that target COX-2 and often suffer from severe side effects, mPGES-1 inhibitors offer a promising alternative with a potentially improved safety profile. This new synthetic route not only streamlines the production process but also aligns with modern green chemistry standards by eliminating hazardous reagents. The target molecule, characterized by its complex quinazolinone scaffold and biphenyl moiety, is crucial for next-generation anti-inflammatory therapies. 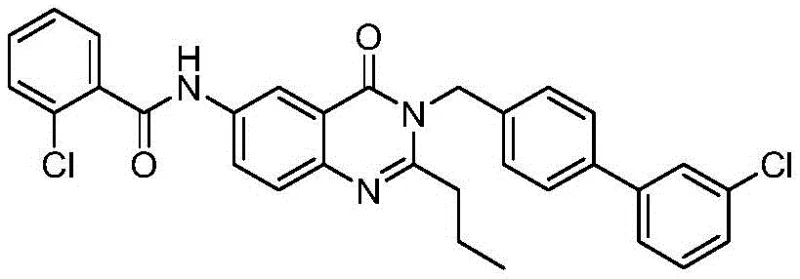
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the synthesis of FR20 and related intermediates was plagued by inefficiencies and safety concerns that hindered large-scale manufacturing. The conventional methodology typically required a four-step sequence, which inherently lowered the overall yield due to material loss at each stage. More critically, the initial step relied heavily on the use of triphenyl phosphite, a highly toxic chemical reagent that poses significant risks to operator safety and environmental compliance. The use of such hazardous materials necessitates expensive containment systems and complex waste treatment protocols, driving up the cost of goods sold. Furthermore, the older routes were associated with low atom economy, generating substantial amounts of chemical waste and by-products that required rigorous purification efforts. 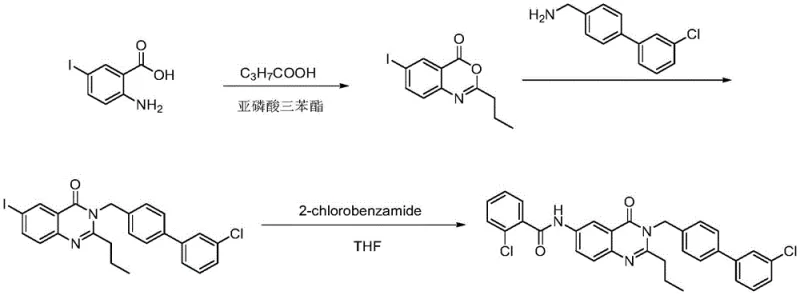
The Novel Approach
In stark contrast, the method disclosed in the patent introduces a streamlined three-step synthesis that dramatically improves both safety and efficiency. By bypassing the need for toxic phosphites, the new route utilizes readily available starting materials such as 5-iodoisatoic anhydride and 4-(3-chlorophenyl)benzylamine hydrochloride. The process leverages advanced transition metal catalysis to achieve cyclization and functionalization in fewer operations, thereby enhancing the overall atom economy. This reduction in step count directly translates to higher throughput and reduced solvent consumption, which are key metrics for sustainable manufacturing. The final product is obtained with high purity through standard chromatographic techniques, demonstrating the robustness of this new chemical strategy for producing high-value pharmaceutical intermediates. 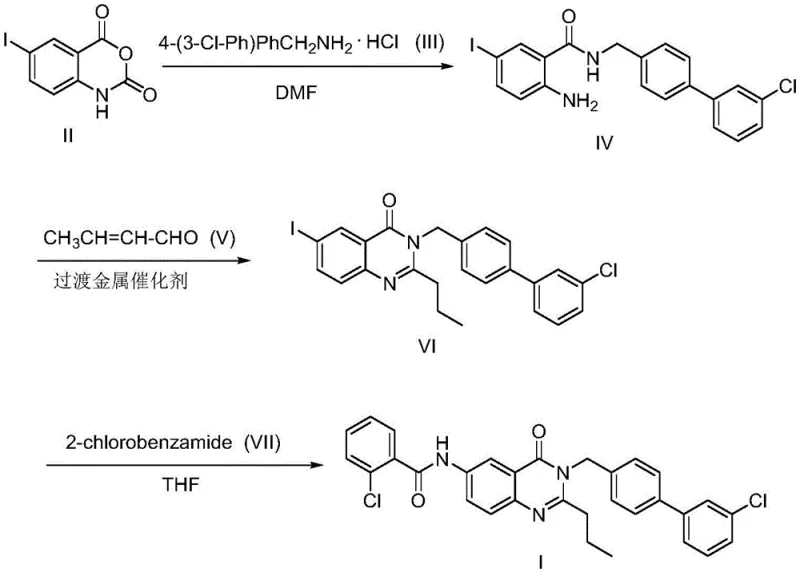
Mechanistic Insights into Transition Metal Catalyzed Cyclization
The core of this synthetic breakthrough lies in the second step, where a sophisticated transition metal-catalyzed transformation occurs. Under nitrogen protection, the intermediate amide undergoes a condensation and dehydrogenation transfer hydrogenation reaction in the presence of an iridium complex catalyst, specifically [Cp*IrCl2]2. This catalytic cycle facilitates the formation of the quinazolinone ring system while simultaneously introducing the propyl group via reaction with crotonaldehyde. The mechanism likely involves the activation of the aldehyde by the iridium center, followed by hydride transfer and subsequent cyclization, which is a testament to the power of modern organometallic chemistry in simplifying complex molecular constructions. The reaction proceeds efficiently at 120°C in toluene, showcasing the thermal stability and effectiveness of the catalyst system in driving the reaction to completion with minimal side reactions.
Impurity control is another critical aspect where this mechanistic approach excels. The high selectivity of the iridium catalyst ensures that the desired cyclization occurs without significant formation of regioisomers or over-reduced by-products. Following this, the final step employs a copper-catalyzed Goldberg amination to attach the 2-chlorobenzamide moiety. This C-N bond formation is facilitated by CuI and a diamine ligand in THF, providing a reliable method to install the final pharmacophore. The combination of these two distinct catalytic cycles—Iridium-mediated cyclization and Copper-mediated amination—creates a orthogonal reactivity profile that minimizes cross-reactivity and ensures a clean impurity profile, which is essential for meeting the stringent quality standards of active pharmaceutical ingredients.
How to Synthesize FR20 Efficiently
Implementing this synthesis requires precise control over reaction parameters to maximize yield and purity. The process begins with the amidation of 5-iodoisatoic anhydride, followed by the critical iridium-catalyzed cyclization, and concludes with the copper-mediated coupling. Each step has been optimized in the patent examples to demonstrate reproducibility, with specific attention paid to stoichiometry and temperature control. For R&D teams looking to adopt this route, it is essential to maintain an inert atmosphere during the catalytic steps to prevent catalyst deactivation. The detailed standardized synthesis steps, including exact reagent quantities and workup procedures, are outlined in the guide below to ensure successful replication in your laboratory.
- React 5-iodoisatoic anhydride with 4-(3-chlorophenyl)benzylamine hydrochloride in DMF with potassium carbonate at 50-60°C to form the N-substituted amide intermediate.
- Perform cyclization and dehydrogenation transfer hydrogenation using a transition metal iridium catalyst and crotonaldehyde in toluene at 120°C to generate the quinazolinone core.
- Execute a Goldberg amination reaction with o-chlorobenzamide using CuI catalyst and potassium phosphate in THF at 110°C to yield the final FR20 compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the shift to this novel synthesis route offers tangible strategic benefits beyond mere technical elegance. By eliminating the reliance on hazardous reagents like triphenyl phosphite, the manufacturing process becomes significantly safer and less regulated, reducing the overhead costs associated with hazardous material handling and disposal. This simplification of the supply chain allows for more flexible sourcing of raw materials, as the new starting materials are commodity chemicals with stable market availability. Furthermore, the reduction in synthetic steps from four to three inherently lowers the operational expenditure by reducing labor hours, energy consumption, and solvent usage per kilogram of product. These factors collectively contribute to a more resilient and cost-effective supply chain capable of withstanding market fluctuations.
- Cost Reduction in Manufacturing: The elimination of toxic reagents and the reduction in step count lead to substantial cost savings in the production of FR20 intermediates. Without the need for specialized equipment to handle hazardous phosphites, capital expenditure is lowered, and the simplified purification processes reduce the consumption of expensive chromatography media and solvents. The high atom economy means that a greater proportion of the starting material mass ends up in the final product, minimizing waste disposal fees and maximizing raw material utilization efficiency.
- Enhanced Supply Chain Reliability: The use of robust, commercially available starting materials such as 5-iodoisatoic anhydride ensures a stable supply base that is less susceptible to shortages. The reaction conditions, while requiring specific catalysts, utilize standard industrial solvents like toluene and THF, which are widely accessible globally. This accessibility reduces the risk of supply chain disruptions and allows for multi-sourcing strategies, ensuring continuous production schedules and reliable delivery timelines for downstream pharmaceutical customers.
- Scalability and Environmental Compliance: Designed with green chemistry principles, this process generates fewer by-products and avoids toxic waste streams, making it easier to scale from pilot plant to commercial production. The reduced environmental footprint simplifies regulatory compliance and permitting processes in various jurisdictions. This scalability ensures that the manufacturing capacity can be rapidly expanded to meet increasing demand for mPGES-1 inhibitors without the bottlenecks typically associated with complex, multi-step syntheses involving hazardous chemicals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of FR20 based on the patented technology. These insights are derived directly from the experimental data and process descriptions provided in the patent documentation, offering clarity on the feasibility and advantages of this route. Understanding these details is crucial for technical teams evaluating the potential for technology transfer or licensing agreements.
Q: What are the primary advantages of this new FR20 synthesis method over conventional routes?
A: The novel method described in patent CN107793367B reduces the synthetic steps from four to three and eliminates the use of highly toxic triphenyl phosphite. This results in significantly higher atom economy, fewer by-products, and a safer, more environmentally compliant manufacturing process suitable for large-scale production.
Q: What specific catalysts are utilized in the key cyclization step?
A: The critical cyclization and hydrogen transfer step utilizes a transition metal iridium complex, specifically [Cp*IrCl2]2, which facilitates the formation of the quinazolinone core with high efficiency under nitrogen protection at elevated temperatures.
Q: Is this synthesis route suitable for commercial scale-up?
A: Yes, the process is designed with green chemistry principles in mind, avoiding toxic reagents and minimizing waste. The use of standard solvents like DMF, toluene, and THF, along with robust reaction conditions, makes it highly adaptable for commercial scale-up from kilogram to multi-ton quantities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable FR20 Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this advanced synthesis route for the production of high-purity FR20 intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project moves seamlessly from the laboratory to the marketplace. Our state-of-the-art facilities are equipped to handle the specific catalytic requirements of this process, including the safe handling of transition metal catalysts and the rigorous purification needed to meet stringent purity specifications. With our dedicated rigorous QC labs, we guarantee that every batch of FR20 intermediate delivered meets the highest international quality standards, providing you with the confidence needed to advance your drug development programs.
We invite you to collaborate with us to leverage this innovative technology for your supply chain needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this greener route can optimize your budget. Please contact us today to request specific COA data for our reference batches and to discuss route feasibility assessments for your upcoming projects. Let us help you secure a reliable, cost-effective, and sustainable supply of this critical pharmaceutical intermediate.
