Advanced Catalytic Synthesis of Thiotriazinone for Commercial Scale API Production
The pharmaceutical landscape is constantly evolving, demanding more efficient and cost-effective pathways for critical active pharmaceutical ingredient (API) intermediates. Patent CN102558080B introduces a groundbreaking method for synthesizing Thiotriazinone, a pivotal building block in the production of Ceftriaxone Sodium. This innovation addresses long-standing inefficiencies in heterocyclic chemistry by employing a specific catalytic system that enhances both yield and operational throughput. For global procurement leaders and R&D directors, understanding this technological shift is essential for securing a reliable pharmaceutical intermediate supplier capable of meeting rigorous quality standards. The transition from traditional thermal methods to this acid-catalyzed approach represents a significant leap forward in process chemistry, offering a robust solution for the commercial scale-up of complex heterocycles required in modern antibiotic manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Thiotriazinone has been plagued by suboptimal reaction kinetics and economic inefficiencies that hinder large-scale production capabilities. Conventional non-catalytic routes often suffer from prolonged reaction times, necessitating extended heating periods that drive up energy consumption and operational costs significantly. Furthermore, these traditional methods frequently result in lower conversion rates, leading to substantial waste of valuable raw materials such as 2-methylthiosemicarbazone and diethyl oxalate. The accumulation of by-products in these slower reactions complicates downstream purification, often requiring multiple recrystallization steps or expensive chromatographic techniques to achieve the necessary purity levels for API synthesis. These bottlenecks create supply chain vulnerabilities, making it difficult for manufacturers to guarantee consistent delivery schedules and competitive pricing structures in a volatile market.
The Novel Approach
The innovative methodology detailed in the patent data revolutionizes this synthesis by introducing a dual-component catalyst system comprising ammonium chloride and a 10 weight percent hydrochloric acid solution. This strategic addition fundamentally alters the reaction pathway, facilitating a much faster cyclization process that drastically reduces the overall reaction time while simultaneously boosting the final yield. By optimizing the molar ratios of reactants and utilizing ethanol as a solvent, the process achieves a highly efficient conversion that minimizes the formation of unwanted impurities. The subsequent purification step utilizes water for recrystallization, which is not only environmentally benign but also significantly reduces solvent recovery costs compared to organic solvent systems. This streamlined approach ensures a more stable and predictable manufacturing process, directly addressing the needs of a reliable pharmaceutical intermediate supplier seeking to optimize cost reduction in API manufacturing.
Mechanistic Insights into Acid-Catalyzed Cyclization
The core of this technological advancement lies in the precise mechanistic role played by the ammonium chloride and hydrochloric acid mixture during the condensation reaction. The acidic environment generated by the catalyst promotes the protonation of the carbonyl oxygen in diethyl oxalate, thereby increasing its electrophilicity and making it more susceptible to nucleophilic attack by the amino groups of 2-methylthiosemicarbazone. This activation lowers the energy barrier for the initial condensation steps, allowing the reaction to proceed rapidly even at moderate temperatures ranging from 30 to 50 degrees Celsius during the addition phase. As the reaction progresses to reflux, the catalyst continues to facilitate the intramolecular cyclization and dehydration steps required to form the stable triazine ring structure. This controlled catalytic environment prevents the degradation of sensitive functional groups, ensuring that the structural integrity of the final product is maintained throughout the synthesis.
Controlling the impurity profile is paramount for any intermediate destined for beta-lactam antibiotic production, and this method offers superior selectivity. The specific acidity provided by the 10wt% HCl and NH4Cl combination suppresses side reactions that typically lead to polymeric by-products or open-chain oligomers. Following the reaction, the crude product is subjected to a recrystallization process using distilled water, potentially aided by activated carbon to remove colored impurities. This step leverages the differential solubility of the target Thiotriazinone versus residual starting materials and salts in hot versus cold water. The result is a high-purity crystalline powder with a melting point exceeding 245 degrees Celsius, meeting the stringent specifications required for downstream coupling reactions in Ceftriaxone Sodium synthesis.
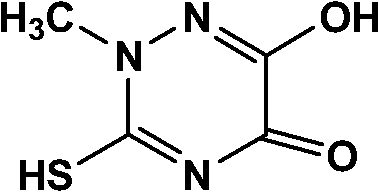
How to Synthesize Thiotriazinone Efficiently
Implementing this synthesis route requires careful attention to stoichiometry and temperature control to maximize the benefits of the catalytic system. The process begins with the preparation of the reaction mass, where 2-methylthiosemicarbazone is suspended in ethanol along with the specific catalyst mixture. Operators must maintain strict temperature control during the dropwise addition of diethyl oxalate to prevent exothermic runaway and ensure uniform mixing. Once the addition is complete, the mixture is heated to reflux for a defined period to drive the reaction to completion. The detailed standardized synthetic steps, including exact gram quantities and specific timing for filtration and drying, are outlined in the technical guide below for immediate implementation by process engineers.
- Combine 2-methylthiosemicarbazone, ethanol, and a catalyst mixture of ammonium chloride and 10wt% hydrochloric acid in a reactor.
- Control temperature between 30-50°C while dropwise adding diethyl oxalate, then heat to reflux for 3 hours.
- Cool the reaction mixture, filter the crude product, and recrystallize using water to obtain the final high-purity Thiotriazinone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this catalytic synthesis method translates into tangible strategic advantages that extend beyond simple unit cost metrics. The elimination of harsh reaction conditions and the reduction in processing time directly correlate to increased plant throughput, allowing manufacturers to respond more agilely to market demand fluctuations. By simplifying the operational workflow and reducing the number of unit operations, the risk of human error and batch-to-batch variability is significantly minimized. This reliability is crucial for maintaining uninterrupted supply lines for critical antibiotics, ensuring that downstream API manufacturers can adhere to their own production schedules without delay. Furthermore, the use of common, inexpensive reagents like ammonium chloride and hydrochloric acid mitigates the risk associated with sourcing specialized or volatile catalysts.
- Cost Reduction in Manufacturing: The implementation of this catalytic system drives down manufacturing expenses through multiple mechanisms, primarily by enhancing the overall atom economy and yield of the reaction. By avoiding the need for expensive transition metal catalysts or exotic reagents, the raw material bill of materials is substantially optimized. Additionally, the shortened reaction cycle time reduces utility consumption, including steam for heating and electricity for agitation, leading to lower overhead costs per kilogram of product. The ability to use water as a recrystallization solvent further eliminates the high costs associated with purchasing, recovering, and disposing of large volumes of organic solvents, resulting in comprehensive cost reduction in pharmaceutical intermediate manufacturing.
- Enhanced Supply Chain Reliability: Securing a steady supply of high-quality intermediates is often challenged by the complexity of synthesis routes, but this method simplifies the dependency on fragile supply chains. The raw materials required, specifically 2-methylthiosemicarbazone and diethyl oxalate, are commodity chemicals with robust global availability, reducing the risk of shortages. The robustness of the reaction conditions means that production is less susceptible to minor variations in environmental factors or equipment performance, ensuring consistent output quality. This stability allows supply chain planners to forecast inventory levels with greater accuracy and reduces the need for excessive safety stock, thereby freeing up working capital and improving overall logistics efficiency.
- Scalability and Environmental Compliance: As regulatory pressures regarding industrial emissions and waste disposal intensify, this synthesis route offers a distinct advantage in environmental compliance and scalability. The replacement of toxic organic solvents with ethanol and water for the workup phase significantly reduces the volatile organic compound (VOC) load released into the atmosphere. The aqueous waste streams generated are easier to treat in standard effluent treatment plants compared to those containing heavy metals or halogenated solvents. This green chemistry profile facilitates smoother regulatory approvals for capacity expansion, enabling the commercial scale-up of complex heterocycles from pilot plant quantities to multi-ton annual production without encountering significant environmental bottlenecks.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of Thiotriazinone using this patented methodology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity for technical teams evaluating this route. Understanding these specifics helps in aligning internal quality standards with the capabilities of potential manufacturing partners. The answers reflect the practical realities of scaling this chemistry from the laboratory bench to full-scale industrial reactors.
Q: What are the key advantages of the new catalytic method for Thiotriazinone?
A: The novel method utilizes a cost-effective ammonium chloride and hydrochloric acid catalyst system which significantly improves reaction yield and shortens reaction time compared to conventional non-catalytic processes.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the process is specifically designed for industrial suitability, featuring simplified operation steps, reduced energy consumption through optimized reflux conditions, and the use of water for recrystallization which minimizes environmental impact.
Q: What is the primary application of Thiotriazinone in the pharmaceutical industry?
A: Thiotriazinone serves as a critical pharmaceutical intermediate primarily used in the synthesis of Ceftriaxone Sodium, a widely prescribed third-generation cephalosporin antibiotic.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Thiotriazinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the global pharmaceutical supply chain, particularly for life-saving antibiotics like Ceftriaxone. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale optimization to full manufacturing is seamless and efficient. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which utilize advanced analytical techniques to verify every batch against international pharmacopoeia standards. Our facility is equipped to handle the specific requirements of acid-catalyzed cyclizations, providing a safe and compliant environment for the synthesis of sensitive heterocyclic compounds.
We invite you to collaborate with us to leverage these technological advancements for your specific supply chain needs. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis that details how switching to this optimized route can impact your bottom line. We encourage you to contact us today to obtain specific COA data for our current stock and to discuss route feasibility assessments tailored to your long-term production goals. Let us be your trusted partner in driving efficiency and quality in your pharmaceutical manufacturing operations.
