Scalable Synthesis of Imidazole-4-Carboxylates via Glycine Esterification for Pharmaceutical Applications
Scalable Synthesis of Imidazole-4-Carboxylates via Glycine Esterification for Pharmaceutical Applications
The pharmaceutical industry continuously seeks robust and cost-effective pathways for synthesizing critical heterocyclic building blocks, particularly imidazole derivatives which serve as foundational structures for numerous active pharmaceutical ingredients (APIs). A significant technological advancement in this domain is detailed in Chinese Patent CN101693694B, which discloses a novel preparation method for glyoxaline formic ether and its derivatives. This patent introduces a streamlined four-step synthesis starting from glycine, bypassing the complexities and toxicological hazards associated with legacy methods. The core innovation lies in the strategic use of esterification, acylation, condensation, and oxidation to construct the imidazole ring with high regioselectivity and yield. By leveraging readily available amino acid precursors, this methodology addresses the growing demand for a reliable pharmaceutical intermediate supplier capable of delivering high-purity materials at scale.
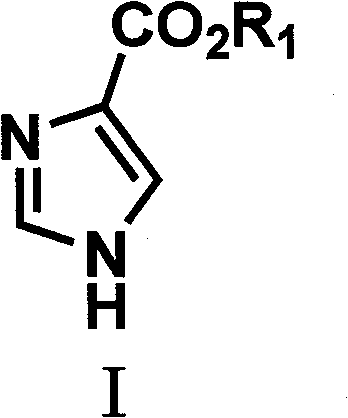
The target compounds, represented generally by Formula (I) in the patent documentation, are pivotal intermediates for drugs treating conditions ranging from allergic rhinitis to parasitic infections. The structural versatility of the imidazole ring allows for diverse functionalization, making these intermediates essential for the development of next-generation therapeutics. The patent emphasizes that the R1 group can be varied across C1-6 alkyl chains or substituted aryl groups, offering flexibility for downstream medicinal chemistry campaigns. This adaptability ensures that the process is not merely a single-product solution but a platform technology for cost reduction in API manufacturing, enabling producers to tailor the ester moiety to specific solubility or reactivity requirements without altering the core synthetic logic.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the synthesis of imidazole-4-carboxylic acid and its derivatives was plagued by significant operational challenges and safety concerns. Historical literature, such as the work by Connell et al. in 1988, relied on sarcosine as a starting material, necessitating a multi-step sequence involving esterification, N-formylation, ring closure, and desulfurization. These traditional routes often suffered from tedious operational complexity and low overall yields due to the accumulation of impurities at each stage. Furthermore, alternative pathways utilizing diamino maleonitrile required the use of triethyl orthoformate for ring closure, followed by difficult selective decarboxylation steps.
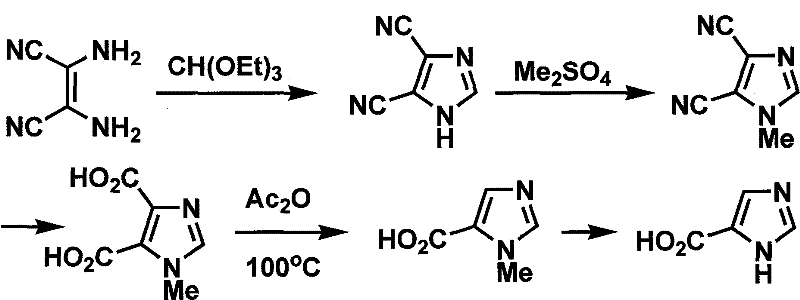
Another prevalent method involved methyl bromoacetate, which introduced severe handling risks due to the lachrymatory and toxic nature of the alkylating agent. These conventional approaches frequently generated substantial amounts of hazardous waste and required stringent safety controls, thereby inflating production costs and extending lead times. The side reactions inherent in these older chemistries often resulted in product mixtures that were difficult to purify, limiting their applicability for industrial-scale production. Consequently, the industry faced a bottleneck in securing a consistent supply of high-quality imidazole intermediates, driving the urgent need for a more efficient and environmentally benign synthetic strategy.
The Novel Approach
The methodology outlined in CN101693694B represents a paradigm shift by utilizing glycine, a ubiquitous and inexpensive amino acid, as the primary feedstock. This novel approach simplifies the synthetic tree into four distinct, controllable stages that avoid the use of highly toxic controlled substances found in prior art. The initial esterification of glycine proceeds under moderate thermal conditions (80-120°C) using common mineral acids or solid acid catalysts, ensuring high conversion rates without the need for exotic reagents. Subsequent acylation and condensation steps are designed to maximize atom economy, directly constructing the imidazole core with minimal byproduct formation.
Crucially, the new route eliminates the need for dangerous alkyl halides like methyl bromoacetate, significantly enhancing workplace safety and reducing the regulatory burden associated with hazardous material handling. The process conditions are described as moderate and easily controllable, allowing for precise management of reaction kinetics to suppress side reactions. This level of control translates directly into improved product purity, reducing the need for extensive downstream purification such as column chromatography. By streamlining the workflow and utilizing commodity chemicals, this method offers a viable pathway for the commercial scale-up of complex heterocycles, aligning perfectly with modern green chemistry principles and economic efficiency goals.
Mechanistic Insights into Glycine-Based Imidazole Construction
The mechanistic elegance of this synthesis lies in its stepwise construction of the heterocyclic ring, beginning with the activation of glycine. In the first stage, glycine undergoes esterification with a C1-6 alcohol in the presence of catalysts such as sulfuric acid, thionyl chloride, or strong-acid ion exchange resins. This generates the glycine ester intermediate (Formula II), which serves as the nucleophilic foundation for the subsequent ring closure. The choice of catalyst is critical; for instance, using sodium pyrosulfate or hydrogen chloride gas allows for the efficient generation of the ester hydrochloride salt, which stabilizes the amine group against premature side reactions during the acylation phase.
Following esterification, the intermediate undergoes acylation to protect the nitrogen atom, followed by a condensation reaction with ethyl formate under alkaline conditions. This step generates an enolate species that is primed for cyclization. The introduction of a thiocyanate source, such as potassium thiocyanate (KSCN), in an acidic environment triggers the formation of the thiol-substituted imidazole intermediate (Formula IV). This specific cyclization mechanism is highly advantageous as it directs the substitution pattern exclusively to the 4-position of the imidazole ring, avoiding the formation of regioisomers that complicate purification. The final oxidative step utilizes hydrogen peroxide or similar oxidants to convert the thiol group into the desired functionality, completing the synthesis of the target imidazole-4-carboxylate with high fidelity.
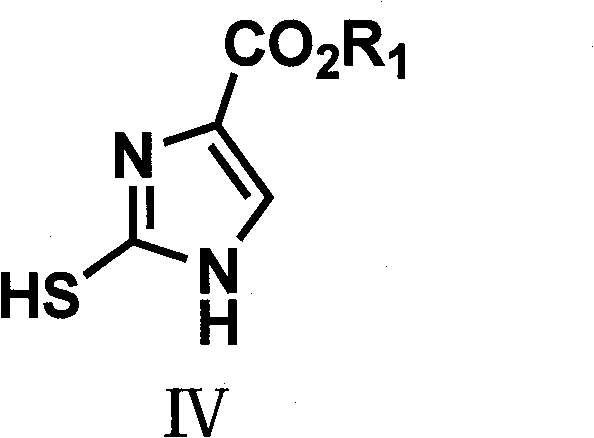
Impurity control is inherently built into this mechanistic pathway. The oxidation of the thiol intermediate is a clean transformation that typically yields water as the only byproduct when using hydrogen peroxide, minimizing organic waste. Furthermore, the patent details specific workup procedures, such as neutralization with saturated sodium carbonate and recrystallization from water or ethanol, which effectively remove inorganic salts and unreacted starting materials. The ability to isolate the thiol intermediate as a solid precipitate before oxidation provides an additional checkpoint for quality control, allowing manufacturers to verify intermediate purity before committing to the final step. This robustness ensures that the final API intermediate meets stringent purity specifications required by global regulatory bodies.
How to Synthesize Imidazole-4-Carboxylate Efficiently
The synthesis of these valuable intermediates follows a logical progression that balances reaction efficiency with operational simplicity. The process begins with the preparation of the glycine ester, followed by protection, ring closure, and final oxidation. Each step has been optimized in the patent examples to demonstrate reproducibility across different ester variants (methyl, ethyl, benzyl). For a comprehensive understanding of the specific stoichiometric ratios, temperature profiles, and workup procedures required to replicate this high-yielding process, please refer to the standardized synthesis guide below.
- Perform esterification of glycine with a C1-6 alcohol under acidic catalysis at 80-120°C to obtain the glycine ester intermediate.
- Conduct an acylation reaction on the glycine ester using appropriate solvents and bases to generate the N-acylated derivative.
- Condense the acylated intermediate with ethyl formate under alkaline conditions, followed by cyclization in an acidic environment with thiocyanate to form the thiol-substituted imidazole.
- Execute an oxidation reaction on the thiol-substituted intermediate using an oxidant like hydrogen peroxide to yield the final imidazole-4-carboxylate product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this glycine-based synthetic route offers transformative benefits regarding cost stability and supply continuity. The primary driver of cost reduction in manufacturing stems from the substitution of expensive or hazardous starting materials with glycine, one of the most abundant and affordable amino acids available globally. Unlike sarcosine or specialized nitriles, glycine is produced on a massive scale for various industries, ensuring a stable price point and shielding buyers from volatile raw material markets. Additionally, the elimination of toxic alkylating agents reduces the costs associated with specialized containment equipment, hazardous waste disposal, and regulatory compliance reporting.
Enhanced supply chain reliability is another critical advantage, as the reagents required for this process—such as ethyl formate, potassium thiocyanate, and common mineral acids—are commodity chemicals with multiple qualified suppliers worldwide. This diversification of the supply base mitigates the risk of production stoppages due to single-source bottlenecks. The simplicity of the reaction conditions, which do not require cryogenic temperatures or high-pressure vessels, allows for production in standard multipurpose reactors found in most fine chemical facilities. This flexibility enables rapid technology transfer and reduces the lead time for high-purity intermediates, allowing pharmaceutical companies to accelerate their own drug development timelines without waiting for complex custom synthesis campaigns.
Scalability and environmental compliance are seamlessly integrated into this process design. The use of aqueous workups and the generation of benign byproducts like water and salts simplify wastewater treatment, lowering the environmental footprint of the manufacturing site. The process is inherently safe, avoiding exothermic runaways associated with more aggressive cyclization methods. From a strategic perspective, this means that production can be scaled from pilot plant quantities to multi-ton annual capacities with minimal engineering changes. The robustness of the chemistry ensures consistent batch-to-batch quality, which is essential for maintaining long-term contracts with major pharmaceutical partners who demand unwavering supply security and adherence to strict environmental, social, and governance (ESG) criteria.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing clarity on the feasibility and advantages of the technology. Understanding these details is crucial for technical teams evaluating the integration of this intermediate into their broader supply chain strategy.
Q: What are the primary advantages of the glycine-based route over traditional sarcosine methods?
A: The glycine-based route utilizes widely available and low-cost raw materials compared to sarcosine or diamino maleonitrile. It avoids the use of highly toxic reagents often found in older methodologies, resulting in a safer production environment and simplified waste treatment protocols.
Q: How does the process ensure high purity of the final imidazole derivative?
A: The process incorporates a specific oxidation step that converts the thiol-substituted intermediate into the target ester. This transformation, combined with controlled crystallization and neutralization steps described in the patent, effectively minimizes side products and facilitates the removal of impurities.
Q: Can this synthetic route be adapted to produce other imidazole derivatives?
A: Yes, the patent explicitly outlines downstream modifications. The resulting imidazole-4-carboxylate can be hydrolyzed to imidazole-4-carboxylic acid, reduced to 4-hydroxymethylimidazole, or further oxidized to imidazole-4-carbaldehyde, providing a versatile platform for various pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Imidazole-4-Carboxylic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality heterocyclic intermediates play in the development of life-saving medications. Our technical team has extensively analyzed the glycine-based route described in CN101693694B and possesses the expertise to implement this chemistry at an industrial level. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from laboratory benchtop to full-scale manufacturing. Our facilities are equipped with rigorous QC labs and advanced analytical instrumentation to guarantee that every batch of imidazole-4-carboxylate meets stringent purity specifications, free from the regioisomeric impurities common in older synthetic methods.
We invite you to collaborate with us to leverage this cost-effective and environmentally friendly technology for your upcoming projects. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this glycine-derived route for your specific application. We encourage you to contact us today to request specific COA data and route feasibility assessments tailored to your volume requirements. Let us help you secure a sustainable and competitive supply of this essential pharmaceutical building block.
