Scalable Synthesis of Imidazole-4-Carboxylates Using Glycine Ester for Pharmaceutical Applications
The pharmaceutical industry continuously seeks robust and scalable synthetic routes for critical heterocyclic building blocks, particularly imidazole derivatives which serve as foundational structures for numerous active pharmaceutical ingredients (APIs). Patent CN101693694B discloses a groundbreaking preparation method for glyoxaline formic ether (imidazole-4-carboxylate) and its derivatives, addressing long-standing challenges in yield and environmental safety. This innovation leverages glycine as a primary feedstock, transforming it through a sequence of esterification, acylation, cyclization, and oxidation to achieve high-purity targets. The significance of this technology lies in its ability to bypass toxic reagents and complex purification steps inherent in legacy processes, thereby offering a streamlined pathway for the production of histidine analogs and related therapeutic agents. For R&D teams and procurement strategists, this represents a pivotal shift towards greener, more cost-effective manufacturing paradigms that align with modern regulatory standards.
The Limitations of Conventional Methods vs. The Novel Approach
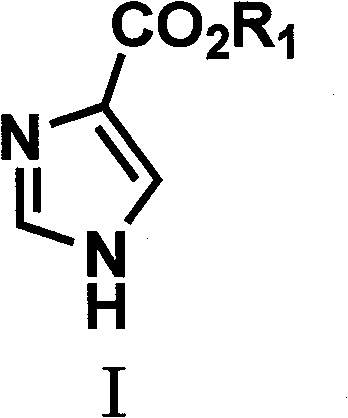
The Limitations of Conventional Methods
Historically, the synthesis of imidazole-4-carboxylic acid and its derivatives has been plagued by significant operational hurdles and safety concerns that hinder large-scale adoption. Traditional routes often rely on hazardous starting materials such as diamino maleonitrile or methyl bromoacetate, which pose severe toxicity risks and require stringent handling protocols to ensure worker safety and environmental compliance. As illustrated in prior art reaction schemes, these conventional pathways frequently involve multi-step sequences with harsh reaction conditions, leading to the formation of complex impurity profiles that are difficult to separate. The use of strong acids, heavy metal catalysts, or controlled substances further complicates the supply chain, increasing both the direct cost of goods sold and the indirect costs associated with waste disposal and regulatory reporting. Consequently, these factors render many existing methods economically unviable for commercial-scale production, limiting the availability of high-quality intermediates for downstream drug development.
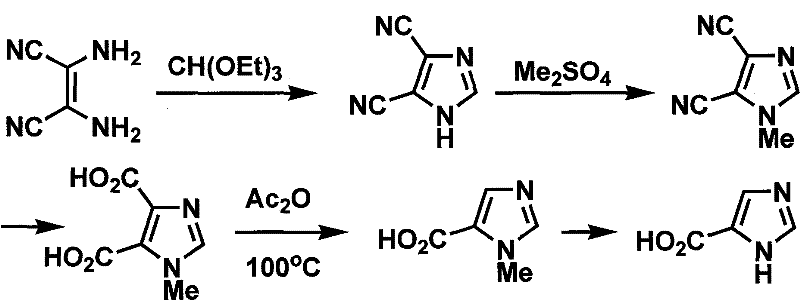
The Novel Approach
In stark contrast, the novel methodology detailed in the patent utilizes glycine, an abundant and benign amino acid, as the cornerstone of the synthesis, fundamentally altering the economic and safety landscape of imidazole production. This approach replaces dangerous precursors with easily accessible alcohols and formates, conducting reactions under moderate temperatures and controllable pH environments that significantly reduce energy consumption and equipment corrosion. The strategic design of the reaction sequence minimizes side reactions, particularly during the critical cyclization and oxidation stages, ensuring that the crude product profile is clean and amenable to simple purification techniques like recrystallization. By eliminating the need for exotic catalysts and toxic solvents, this process not only lowers the barrier to entry for manufacturers but also enhances the overall sustainability of the supply chain, making it an ideal candidate for reliable pharmaceutical intermediate supplier networks seeking to optimize their portfolios.
Mechanistic Insights into Glycine-Based Cyclization and Oxidation
The core of this synthetic strategy relies on a precise four-step transformation that maximizes atom economy while maintaining strict control over regioselectivity. The process initiates with the esterification of glycine to form a glycine ester derivative, typically achieved by refluxing glycine with an appropriate alcohol in the presence of an acid catalyst such as sulfuric acid or thionyl chloride. This step is crucial as it protects the amine functionality and sets the stage for subsequent acylation. Following this, the glycine ester undergoes acylation to generate an N-acylated intermediate, a reaction that can be facilitated by bases like triethylamine or DMAP in solvents such as ethyl acetate. The subsequent condensation with ethyl formate under alkaline conditions, followed by acid-catalyzed cyclization in the presence of a sulfur source like potassium thiocyanate, constructs the imidazole ring with high fidelity. This specific sequence ensures that the carboxylate group is positioned correctly at the 4-position of the ring, a structural feature essential for biological activity in many target molecules.
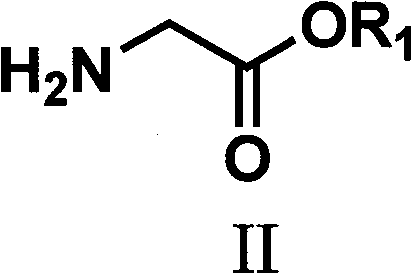
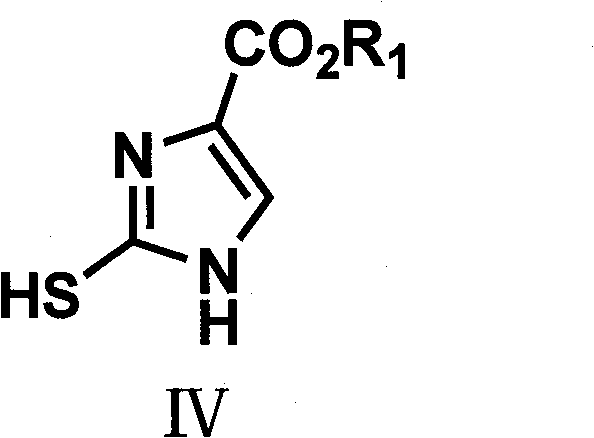
The final and perhaps most critical mechanistic step involves the oxidative desulfurization of the thiol-substituted imidazole intermediate to yield the desired imidazole-4-carboxylate. This transformation is elegantly executed using hydrogen peroxide as the oxidant, which selectively removes the sulfur moiety without compromising the integrity of the ester group or the aromatic imidazole ring. The reaction proceeds smoothly at temperatures ranging from 0°C to 50°C, demonstrating remarkable chemoselectivity that prevents over-oxidation or ring degradation. This mild oxidative condition is a key differentiator, as it avoids the use of heavy metal oxidants that often leave trace contaminants requiring expensive removal processes. The resulting product, characterized by its high purity and defined crystal structure, serves as a versatile platform for further derivatization into acids, alcohols, or aldehydes, thereby expanding the utility of this synthetic route across various therapeutic classes.
How to Synthesize Imidazole-4-Carboxylate Efficiently
To implement this advanced synthesis in a laboratory or pilot plant setting, operators must adhere to a standardized protocol that emphasizes temperature control and reagent stoichiometry to maximize yield and purity. The process begins with the careful preparation of the glycine ester, followed by sequential acylation and cyclization steps that require precise pH adjustments to drive the ring closure effectively. The final oxidation step demands attention to the rate of oxidant addition to manage exothermicity and ensure complete conversion of the thiol intermediate. While the general workflow is straightforward, specific parameters such as solvent choice and reaction times may need optimization based on the specific ester group desired. For a comprehensive guide on the exact operational parameters, please refer to the detailed standard operating procedures below.
- Perform esterification of glycine with suitable alcohol under catalytic conditions to obtain glycine ester.
- Conduct acylation reaction on the glycine ester to form the N-acylated intermediate.
- Condense the acylated intermediate with ethyl formate under alkaline conditions, followed by cyclization in acid to form the thiol-substituted imidazole.
- Oxidize the thiol-substituted intermediate using an oxidant like hydrogen peroxide to yield the final imidazole-4-carboxylate.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this glycine-based synthesis route offers profound benefits for procurement managers and supply chain directors aiming to stabilize costs and secure long-term material availability. The shift away from volatile and regulated raw materials to commodity chemicals like glycine and ethanol drastically reduces the risk of supply disruptions caused by regulatory changes or raw material shortages. Furthermore, the simplified reaction workup and purification processes translate directly into lower operational expenditures, as less time and resources are required for waste treatment and product isolation. This efficiency gain allows manufacturers to offer more competitive pricing structures without sacrificing quality, creating a compelling value proposition for downstream pharmaceutical clients who are increasingly pressure-sensitive regarding their input costs.
- Cost Reduction in Manufacturing: The elimination of expensive and toxic reagents, combined with the use of inexpensive bulk chemicals, leads to a substantial decrease in the overall cost of goods. By avoiding complex purification steps associated with heavy metal catalysts, manufacturers can significantly reduce processing time and utility consumption, resulting in a leaner and more profitable production model that enhances margin potential for all stakeholders in the value chain.
- Enhanced Supply Chain Reliability: Sourcing glycine and common alcohols is far more stable than relying on specialized heterocyclic precursors that may have limited global suppliers. This diversification of the raw material base mitigates the risk of single-source dependency, ensuring consistent delivery schedules and reducing lead times for high-purity pharmaceutical intermediates. The robustness of the supply chain is further bolstered by the process tolerance, which allows for flexibility in sourcing grades of reagents without compromising final product specifications.
- Scalability and Environmental Compliance: The mild reaction conditions and aqueous workup possibilities make this process inherently scalable from kilogram to multi-ton production levels with minimal engineering modifications. Additionally, the reduced generation of hazardous waste aligns with increasingly stringent environmental regulations, lowering the compliance burden and potential liability for manufacturing sites. This environmental stewardship not only safeguards the company's reputation but also future-proofs the production capability against evolving green chemistry mandates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this novel synthetic route. These insights are derived directly from the patent specifications and are intended to clarify the operational feasibility and strategic advantages for potential partners. Understanding these details is crucial for making informed decisions about integrating this technology into existing production frameworks or new product development pipelines.
Q: What are the primary advantages of the glycine-based route over traditional methods?
A: The glycine-based route utilizes inexpensive, non-toxic raw materials compared to hazardous precursors like diamino maleonitrile. It features milder reaction conditions, fewer side reactions, and simpler purification processes, making it highly suitable for industrial scale-up.
Q: How is the purity of the final imidazole-4-carboxylate ensured?
A: Purity is ensured through the selective oxidation of the thiol intermediate using hydrogen peroxide, which minimizes byproduct formation. The process allows for effective recrystallization steps, resulting in high-purity products suitable for sensitive pharmaceutical applications.
Q: Can this method be adapted for different ester derivatives?
A: Yes, the method is versatile. By varying the alcohol used in the initial esterification step (e.g., methanol, ethanol, benzyl alcohol), different ester derivatives (R1 groups) can be synthesized. Furthermore, the final ester can be hydrolyzed or reduced to access acids, alcohols, or aldehydes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Imidazole-4-Carboxylate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this glycine-based synthesis technology and are fully equipped to leverage it for your specific project needs. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab bench to market is seamless and efficient. Our state-of-the-art facilities are designed to handle the specific requirements of heterocyclic chemistry, featuring rigorous QC labs and stringent purity specifications that guarantee the highest quality standards for every batch produced. We are committed to delivering not just a chemical product, but a comprehensive solution that optimizes your supply chain and accelerates your time to market.
We invite you to engage with our technical team to explore how this innovative route can be tailored to your unique requirements. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits specific to your volume needs. We encourage you to contact our technical procurement team today to索取 specific COA data and route feasibility assessments, allowing us to demonstrate our capability as your trusted partner in advancing pharmaceutical innovation.
