Advanced Catalytic Oxidation for High-Purity 3-Cyano-Pyridine N-Oxide Manufacturing
The pharmaceutical and agrochemical industries rely heavily on the efficient synthesis of pyridine derivatives, particularly 3-cyano-pyridine N-oxide, which serves as a critical precursor for high-value active ingredients. A pivotal advancement in this domain is documented in patent CN103570617A, which introduces a highly selective oxidation protocol utilizing heteropoly acid catalysts. This technology addresses long-standing challenges in the production of 2-chloronicotinic acid intermediates, which are essential for synthesizing herbicides like nicosulfuron and diflufenican, as well as pharmaceutical agents such as nevirapine and niflumic acid. By shifting away from traditional transition metal catalysts towards robust heteropoly acid systems, manufacturers can achieve unprecedented levels of purity and yield. This report analyzes the technical merits of this innovation, providing R&D directors and procurement specialists with a clear roadmap for integrating this superior synthetic route into their global supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial oxidation of nicotinonitrile to its N-oxide form has been plagued by selectivity issues and the formation of stubborn byproducts. Traditional methods often employ catalysts such as sodium tungstate or sodium molybdate in aqueous media. While these systems can facilitate oxidation, they frequently suffer from competitive hydrolysis reactions where the nitrile group is inadvertently converted into an amide group. This results in the co-production of nicotinamide-N-oxide, a structural analog that is difficult to separate and detrimental to downstream processing. Furthermore, alternative high-selectivity catalysts like methyl rhenium trioxide, while effective in laboratory settings, are prohibitively expensive and toxic, rendering them unsuitable for large-scale commercial manufacturing. These conventional limitations lead to increased purification costs, lower overall atom economy, and significant bottlenecks in the production of downstream chlorinated intermediates.
The Novel Approach
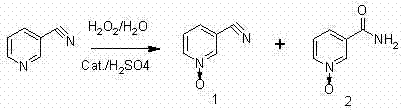
The innovative methodology described in the referenced patent overcomes these deficiencies by employing a specific class of heteropoly acids, such as silicomolybdic acid, phosphomolybdic acid, or phosphotungstic acid, in conjunction with sulfuric acid as a co-catalyst. This dual-catalyst system creates a highly acidic environment that favors the oxidation of the pyridine nitrogen while effectively suppressing the hydrolysis of the cyano group. The result is a dramatic improvement in product selectivity, with the content of the unwanted nicotinamide-N-oxide impurity reduced to negligible levels. This high-purity output not only simplifies the isolation process but also enhances the efficiency of subsequent chlorination steps required to produce 2-chloronicotinic acid. The process utilizes water as the primary solvent and hydrogen peroxide as a green oxidant, aligning with modern principles of sustainable chemical manufacturing.
Mechanistic Insights into Heteropoly Acid-Catalyzed Oxidation
The core of this technological breakthrough lies in the unique electronic properties of heteropoly acids acting in synergy with strong mineral acids. In this catalytic cycle, the heteropoly acid functions as a potent oxygen transfer agent, activating the hydrogen peroxide to generate reactive peroxo-species capable of oxidizing the pyridine ring nitrogen. Simultaneously, the presence of sulfuric acid ensures a sufficiently low pH, which protonates the pyridine nitrogen and modulates the electron density of the ring. This protonation is crucial as it deactivates the cyano group towards nucleophilic attack by water molecules, thereby preventing the hydrolysis pathway that leads to amide formation. The reaction is typically conducted at elevated temperatures between 75°C and 95°C, a range that provides sufficient kinetic energy for the oxidation while maintaining thermal stability of the catalyst system. This precise control over reaction conditions allows for the selective formation of the N-oxide bond without compromising the integrity of the nitrile functionality.
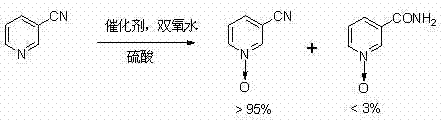
From an impurity control perspective, the mechanism inherently minimizes the generation of side products that complicate downstream purification. In conventional processes, the presence of amide impurities can interfere with the subsequent chlorination reaction using phosphorus oxychloride, leading to lower yields of the final 2-chloro derivative. By ensuring that the starting N-oxide material possesses a purity exceeding 95% with minimal amide contamination, the new catalytic system streamlines the entire synthetic sequence. The catalyst loading is remarkably low, typically ranging from 0.5% to 0.9% by weight relative to the substrate, which further reduces the burden on waste treatment facilities. The robustness of heteropoly acids allows them to maintain activity throughout the extended addition period of the oxidant, ensuring consistent conversion rates even on a multi-ton scale.
How to Synthesize 3-Cyano-Pyridine N-Oxide Efficiently
Implementing this synthesis route requires careful attention to the order of addition and temperature profiling to maximize safety and yield. The process begins with the preparation of the catalytic solution, followed by the controlled addition of the oxidant to manage the exothermic nature of the reaction. Detailed operational parameters, including specific stirring rates and cooling capacities, are critical for maintaining the narrow temperature window required for optimal selectivity. The following guide outlines the standardized procedure derived from the patent embodiments, serving as a foundational reference for process engineers looking to adopt this technology.
- Charge the reactor with water, sulfuric acid, and the selected heteropoly acid catalyst (e.g., silicomolybdic acid).
- Add nicotinonitrile and heat the mixture to a controlled temperature range of 75-95°C.
- Slowly drip 30% hydrogen peroxide over 8-12 hours, maintain temperature, then cool and filter the product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this heteropoly acid-catalyzed oxidation process offers tangible strategic benefits beyond mere technical superiority. The shift to this methodology directly addresses key cost drivers in fine chemical manufacturing, specifically regarding raw material efficiency and waste management. By utilizing water as the solvent instead of organic solvents, the process eliminates the need for costly solvent recovery systems and reduces the environmental footprint associated with volatile organic compound emissions. Furthermore, the high selectivity of the reaction means that less raw material is wasted on byproduct formation, effectively increasing the yield of the desired intermediate per batch. This efficiency translates into a more reliable supply of high-purity precursors, reducing the risk of production delays caused by quality failures or extensive reprocessing requirements.
- Cost Reduction in Manufacturing: The elimination of expensive noble metal catalysts, such as rhenium-based systems, results in a substantial decrease in direct material costs. Additionally, the simplified work-up procedure, which involves merely cooling, standing, and filtering the product, removes the need for complex extraction or chromatographic purification steps. This reduction in unit operations lowers both energy consumption and labor costs, contributing to a more competitive pricing structure for the final intermediate. The ability to recycle the aqueous mother liquor or treat it with lower complexity further enhances the economic viability of the process on an industrial scale.
- Enhanced Supply Chain Reliability: The robustness of the catalyst system ensures consistent batch-to-batch reproducibility, a critical factor for maintaining uninterrupted supply lines to downstream API manufacturers. The use of commodity chemicals like sulfuric acid and hydrogen peroxide mitigates the risk of supply disruptions associated with specialized or imported reagents. Moreover, the process has been validated at the 2000L scale, demonstrating that it can be seamlessly transferred from pilot plants to full commercial production without significant loss of efficiency. This scalability assures partners of a stable long-term supply of critical agrochemical and pharmaceutical intermediates.
- Scalability and Environmental Compliance: The process aligns with stringent environmental regulations by minimizing hazardous waste generation. The primary byproduct of the oxidation is water, and the absence of heavy metal contaminants simplifies effluent treatment protocols. This environmental compatibility facilitates easier permitting and regulatory approval for manufacturing sites, reducing the administrative burden on compliance teams. The high purity of the product also reduces the load on downstream waste streams, as fewer impurities need to be removed in subsequent synthetic steps, creating a cleaner and more sustainable value chain overall.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this oxidation technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent literature, providing clarity on performance metrics and operational feasibility. Understanding these details is essential for technical teams evaluating the integration of this process into existing manufacturing portfolios.
Q: What is the primary advantage of using heteropoly acids over sodium tungstate?
A: Heteropoly acids combined with sulfuric acid significantly suppress the formation of nicotinamide-N-oxide byproducts, achieving yields over 95% compared to traditional methods.
Q: Is this process scalable for industrial production?
A: Yes, the patent explicitly demonstrates successful scale-up in 2000L glass-lined reactors with consistent yields above 95% and high purity.
Q: How does this method impact downstream chlorination?
A: By minimizing amide impurities, the resulting N-oxide is more suitable for subsequent chlorination reactions, improving the overall yield of 2-chloronicotinic acid derivatives.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Cyano-Pyridine N-Oxide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of life-saving medicines and crop protection solutions. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 3-cyano-pyridine N-oxide meets the exacting standards required for sensitive downstream transformations. Our commitment to excellence ensures that our partners receive materials that consistently perform in their own synthetic processes.
We invite global pharmaceutical and agrochemical companies to collaborate with us to optimize their supply chains through advanced manufacturing technologies. By leveraging our expertise in catalytic oxidation, we can help you achieve significant operational efficiencies and cost advantages. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our capabilities can support your long-term strategic goals.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
