Advanced Synthesis of Polyfluorophenyl Aromatic Diphenols for High-Performance Electronic Polymers
The rapid evolution of the microelectronics and optical communication sectors demands materials that can withstand rigorous thermal environments while exhibiting superior electrical insulation properties. Patent CN102432436A introduces a groundbreaking class of polyfluorophenyl substituted aromatic diphenols that serve as critical monomers for next-generation polyarylether polymers. Unlike traditional bisphenols which often result in rigid, insoluble polymer chains, this invention strategically incorporates bulky trifluoromethyl and polyfluorophenyl side groups into the molecular backbone. This structural innovation effectively increases the free volume within the polymer matrix, thereby solving the longstanding trade-off between thermal stability and processability. The resulting polymers exhibit remarkably low dielectric constants and dielectric loss, making them ideal candidates for high-frequency circuit boards and aerospace insulation applications where signal integrity is paramount.
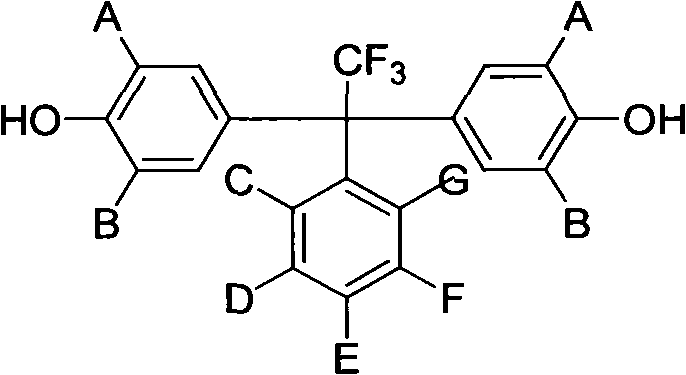
For procurement specialists and supply chain managers seeking a reliable electronic chemical supplier, understanding the synthetic accessibility of these monomers is crucial. The patent outlines a direct condensation route that avoids complex multi-step fluorination sequences, relying instead on the reaction between readily available monophenols and alpha,alpha,alpha-trifluoro-polyfluorinated acetophenones. This streamlined approach not only simplifies the manufacturing workflow but also significantly reduces the dependency on exotic reagents, thereby enhancing supply chain resilience. By leveraging this technology, manufacturers can produce high-purity OLED material precursors and semiconductor process chemicals with greater efficiency, ensuring a steady flow of critical components for the global electronics industry.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of high-performance polyarylether ketones (PAEK) and polyarylether sulfones (PES) has been hindered by the inherent rigidity of their molecular structures. Conventional monomers, such as unsubstituted hydroquinone or 4,4'-biphenol, lead to polymers with highly regular and packed chains. While this confers excellent mechanical strength, it results in poor solubility and high melting points that complicate processing and molding. Furthermore, standard aromatic polymers often suffer from relatively high dielectric constants and significant moisture absorption, which are detrimental characteristics for applications in high-speed optical communication and microelectronics. Attempts to modify these properties often involved post-polymerization modifications or the use of expensive, difficult-to-synthesize fluorinated monomers that drove up production costs and limited commercial scalability.
The Novel Approach
The methodology described in the patent represents a paradigm shift by pre-functionalizing the monomer with specific fluorinated architectures before polymerization. By reacting monophenols with polyfluorinated acetophenones under Lewis acid catalysis, the process installs a central quaternary carbon bearing both a trifluoromethyl group and a polyfluorophenyl ring. This bulky, asymmetric structure acts as a molecular spacer that disrupts chain packing in the final polymer. As illustrated in the reaction scheme below, this diphenol intermediate can then be copolymerized with various dihalogen monomers to create a diverse range of polyarylethers. This approach ensures that the beneficial properties—such as enhanced solubility in common solvents like NMP and DMF, and reduced water uptake—are built directly into the material's foundation, offering a robust solution for cost reduction in electronic chemical manufacturing.
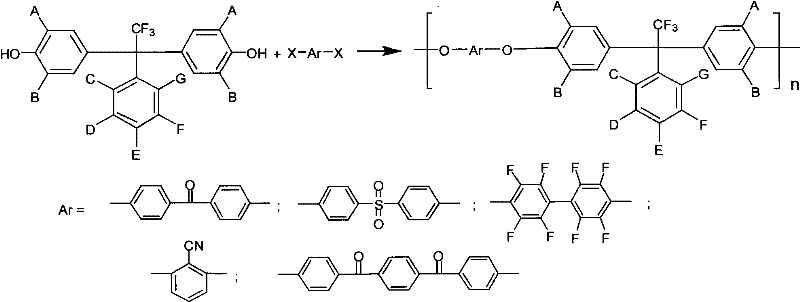
Mechanistic Insights into Lewis Acid-Catalyzed Condensation
The core of this synthesis lies in the electrophilic aromatic substitution facilitated by a Lewis acid catalyst. The reaction initiates with the activation of the carbonyl group in the alpha,alpha,alpha-trifluoro-polyfluorinated acetophenone by the acidic catalyst, such as trifluoroacetic acid or methanesulfonic acid. This activation increases the electrophilicity of the carbonyl carbon, making it susceptible to nucleophilic attack by the electron-rich aromatic ring of the monophenol. The presence of multiple fluorine atoms on the acetophenone ring exerts a strong electron-withdrawing effect, which must be carefully balanced by the choice of catalyst strength and reaction temperature, typically ranging from 70°C to 210°C. The mechanism proceeds through a carbocation intermediate, which is stabilized by the adjacent trifluoromethyl group, ultimately leading to the formation of the diaryl-methane type structure with high regioselectivity at the para-position relative to the hydroxyl group.
Impurity control is meticulously managed through the physical properties of the reaction byproducts and unreacted starting materials. The patent emphasizes a purification strategy that exploits the differential solubility of the product versus the impurities. After the reaction reaches completion, typically within 0.5 to 48 hours depending on the specific substrates, the crude mixture is subjected to vigorous washing with boiling water. This step is critical for removing residual acid catalysts and water-soluble phenolic oligomers. Subsequent recrystallization from binary solvent systems, such as ethanol/water or toluene/n-hexane, further refines the crystal lattice, excluding structurally similar isomers or ortho-substituted byproducts. This rigorous purification protocol ensures that the resulting diphenol meets the stringent purity specifications required for the synthesis of high-molecular-weight polymers with consistent electrical properties.
How to Synthesize Polyfluorophenyl Aromatic Diphenol Efficiently
The synthesis protocol detailed in the patent offers a reproducible pathway for generating these specialized monomers on a commercial scale. The process begins with the precise stoichiometric mixing of the monophenol and the fluorinated acetophenone, typically in a molar ratio between 3:1 and 8:1 to drive the reaction to completion and minimize side reactions. The choice of solvent is often unnecessary as the reactants can form a homogeneous melt upon heating, although careful temperature control is required to prevent thermal degradation. The following guide summarizes the critical operational parameters derived from the experimental examples, providing a framework for process engineers to adapt this chemistry for large-scale production while maintaining high yields and product quality.
- Mix monophenol, alpha,alpha,alpha-trifluoro-polyfluorinated acetophenone, and a Lewis acid catalyst in a reaction vessel, heating gently to ensure complete dissolution into a homogeneous liquid.
- Raise the temperature to between 70°C and 210°C and maintain stirring under the catalytic influence of the Lewis acid for a duration ranging from 0.5 to 48 hours to facilitate the condensation reaction.
- Cool the reaction mixture, wash the resulting phenolic compound thoroughly with boiling water to remove impurities, and finally purify the product via recrystallization using solvents such as ethanol/water or toluene/n-hexane.
Commercial Advantages for Procurement and Supply Chain Teams
For organizations focused on optimizing their supply chain and reducing overall manufacturing expenditures, this technology offers several distinct logistical and economic benefits. The reliance on commodity chemicals like phenol and commercially available fluorinated acetophenones eliminates the need for custom synthesis of complex fluorinated building blocks, which are often bottlenecks in the supply of specialty electronic materials. Furthermore, the reaction conditions do not require extreme pressures or cryogenic temperatures, allowing the process to be conducted in standard glass-lined or stainless steel reactors commonly found in fine chemical facilities. This compatibility with existing infrastructure significantly lowers the barrier to entry for scale-up and reduces capital expenditure requirements for new production lines.
- Cost Reduction in Manufacturing: The synthetic route described eliminates the need for expensive transition metal catalysts or hazardous fluorinating agents that typically drive up the cost of fluorinated intermediates. By utilizing simple Lewis acids like trifluoroacetic acid which can potentially be recovered or neutralized easily, the process minimizes raw material costs. Additionally, the high yield reported in the examples suggests efficient atom economy, meaning less waste is generated per unit of product. This efficiency translates directly into lower production costs, allowing suppliers to offer competitive pricing for high-purity electronic chemical intermediates without compromising on quality margins.
- Enhanced Supply Chain Reliability: The raw materials specified in the patent, including various substituted phenols and fluorinated acetophenones, are produced by multiple global chemical manufacturers, ensuring a diversified and resilient supply base. This reduces the risk of single-source dependency that often plagues the procurement of niche electronic materials. Moreover, the robustness of the reaction conditions means that production schedules are less likely to be disrupted by minor fluctuations in utility supplies or environmental conditions, guaranteeing consistent delivery timelines for downstream polymer manufacturers who rely on just-in-time inventory models.
- Scalability and Environmental Compliance: The workup procedure primarily involves aqueous washing and recrystallization, avoiding the generation of heavy metal waste streams associated with palladium or nickel-catalyzed cross-coupling reactions. This simplifies wastewater treatment and aligns with increasingly strict environmental regulations governing the production of specialty chemicals. The ability to purify the product through crystallization rather than column chromatography makes the process inherently scalable from kilogram to multi-ton quantities. This scalability ensures that as demand for low-dielectric polymers grows in the 5G and aerospace sectors, the supply of the key diphenol monomer can be rapidly expanded to meet market needs.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing clarity on the practical aspects of producing and utilizing these advanced monomers. Understanding these details is essential for R&D teams evaluating the feasibility of integrating these materials into their existing product portfolios.
Q: What are the primary advantages of introducing polyfluorophenyl groups into polyarylether polymers?
A: Introducing bulky polyfluorophenyl side groups significantly increases the free volume of the polymer chain. This structural modification enhances solubility in common organic solvents, reduces the dielectric constant and dielectric loss due to the low polarizability of fluorine atoms, and lowers moisture absorption while maintaining high thermal stability.
Q: Which catalysts are suitable for the synthesis of these aromatic diphenols?
A: The patent specifies the use of strong Lewis acids or protic acids as catalysts. Suitable options include methanesulfonic acid, p-toluenesulfonic acid, trifluoromethanesulfonic acid, trifluoroacetic acid, hydrochloric acid, oleum, and nitric acid, allowing flexibility based on availability and specific reactivity requirements.
Q: How is the purity of the final diphenol product ensured during manufacturing?
A: Purity is achieved through a robust workup procedure involving repeated washing of the crude phenolic compound with boiling deionized water until the product turns white, followed by a final recrystallization step using specific solvent systems like methanol/water, ethanol/water, or toluene/n-hexane to remove residual starting materials and byproducts.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polyfluorophenyl Aromatic Diphenol Supplier
As the demand for advanced materials in the microelectronics and optical communication sectors continues to surge, having a partner with deep technical expertise and proven manufacturing capabilities is essential. NINGBO INNO PHARMCHEM stands at the forefront of this industry, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the specific thermal and corrosive requirements of fluorinated chemistry, ensuring that every batch of polyfluorophenyl aromatic diphenol meets stringent purity specifications. With our rigorous QC labs and commitment to process optimization, we deliver materials that consistently perform in the most demanding polymerization applications, supporting your innovation from pilot scale to full commercialization.
We invite you to collaborate with us to explore how this patented technology can enhance your product line and reduce your overall manufacturing costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and application needs. Please contact us today to request specific COA data for our available grades and to discuss route feasibility assessments for your next generation of high-performance polyarylether polymers. Let us be your trusted partner in navigating the complexities of the global electronic chemical supply chain.
