Advanced Carbonylation Strategy for High-Purity Ibuprofen Impurity F Production
Introduction to Novel Impurity Synthesis
The pharmaceutical industry relies heavily on the availability of high-purity reference standards to ensure the safety and efficacy of active pharmaceutical ingredients (APIs). A significant breakthrough in this domain is documented in patent CN114195633A, which discloses an innovative synthetic method for Ibuprofen Impurity F, chemically known as 3-(4-isobutylphenyl)propionic acid. This impurity is a critical isomer specified in the European Pharmacopoeia, necessitating rigorous control during ibuprofen manufacturing. The disclosed technology utilizes a palladium-catalyzed carbonylation and rearrangement strategy, transforming 1-(4-isobutylphenyl)ethanol directly into the target acid. This approach represents a paradigm shift from traditional multi-step syntheses, offering a streamlined pathway that aligns with modern green chemistry principles while delivering exceptional purity levels suitable for analytical standardization.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the synthesis of Ibuprofen Impurity F was fraught with inefficiencies that hindered large-scale production and increased costs substantially. Existing literature, such as the method described in CN112441902A, relied on a cumbersome three-step sequence involving a Wittig reaction, catalytic hydrogenation, and subsequent hydrolysis. The initial Wittig step is particularly problematic due to the requirement for expensive phosphorus ylides, often needed in three-fold molar excess relative to the aldehyde starting material, which drastically inflates raw material costs. Furthermore, the purification of the final carboxylic acid product in these traditional routes typically necessitates column chromatography, a technique that is notoriously difficult to scale industrially due to high solvent consumption and low throughput. The generation of significant alkaline wastewater during the hydrolysis step also poses environmental compliance challenges, making these legacy processes unsustainable for modern supply chains seeking cost reduction in pharmaceutical intermediate manufacturing.
The Novel Approach
In stark contrast, the methodology presented in CN114195633A simplifies the entire operation into a highly efficient one-pot transformation. By leveraging a carbonylation-rearrangement cascade, the process converts the alcohol precursor directly into the desired propionic acid derivative using carbon monoxide as the C1 source. This eliminates the need for stoichiometric Wittig reagents and avoids the generation of phosphine oxide byproducts. Crucially, the workup procedure is remarkably straightforward; instead of laborious chromatography, the crude product is neutralized and subjected to crystallization, yielding material with purity exceeding 99%. This drastic simplification of the operational workflow not only reduces the physical footprint required for production but also minimizes the exposure of operators to hazardous solvents, thereby enhancing overall process safety and reliability for a reliable pharmaceutical intermediate supplier.
Mechanistic Insights into Pd-Catalyzed Carbonylation and Rearrangement
The core of this technological advancement lies in the sophisticated interplay between the palladium catalyst and the specialized phosphine ligand system. The reaction initiates with the coordination of the palladium species to the benzylic alcohol substrate, facilitated by the acidic environment provided by hydrochloric acid or p-toluenesulfonic acid. Under high pressure of carbon monoxide (5-7 MPa), the palladium center inserts CO into the carbon-metal bond, forming an acyl-palladium intermediate. This step is critical for extending the carbon chain by one unit, effectively converting the ethyl side chain into a propionic acid moiety. The presence of the acid promoter is essential not only for activating the alcohol but also for driving the subsequent rearrangement that ensures the correct regiochemistry of the final isomer. The reaction temperature, maintained between 110-140°C, provides the necessary thermal energy to overcome the activation barrier for this migratory insertion and rearrangement sequence.
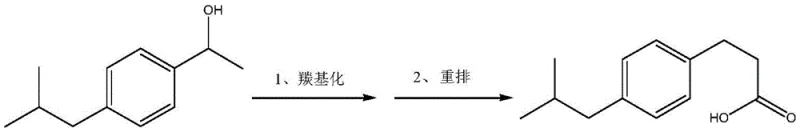
A distinguishing feature of this mechanism is the indispensable role of the sulfonic acid group-modified phosphine ligand, specifically sodium triphenylphosphine tri-m-sulfonate (TPPTS). Comparative data within the patent reveals that substituting this ligand with unmodified triphenylphosphine results in a catastrophic drop in yield from over 28% to less than 1%. This suggests that the sulfonate groups impart crucial water solubility or phase-transfer characteristics that stabilize the active palladium species in the acidic, polar reaction medium. This stabilization prevents the aggregation of palladium black, a common deactivation pathway in carbonylation reactions, thereby maintaining catalytic turnover throughout the prolonged heating period. The precise tuning of the ligand-to-metal ratio (approximately 40:1 to 60:1) further optimizes the electronic environment around the metal center, ensuring high selectivity for the linear acid product over potential branched isomers or oligomerization byproducts.
How to Synthesize 3-(4-isobutylphenyl)propionic Acid Efficiently
Implementing this synthesis requires careful attention to high-pressure safety protocols and reagent quality. The process begins by charging the autoclave with the alcohol substrate, the palladium source (such as palladium acetate), and the sulfonated phosphine ligand in an acetone solvent system. Hydrochloric acid is added to establish the requisite acidic conditions before the vessel is sealed and pressurized with carbon monoxide. The detailed standardized synthesis steps, including specific molar ratios, pressure cycling procedures, and crystallization parameters, are outlined below to ensure reproducibility and safety in your laboratory or pilot plant operations.
- Charge 1-(4-isobutylphenyl)ethanol, palladium acetate, and sodium triphenylphosphine tri-m-sulfonate into a high-pressure autoclave with acetone and hydrochloric acid.
- Replace atmosphere with CO three times, pressurize to 6 MPa, and heat to 125°C until pressure drop ceases, indicating reaction completion.
- Concentrate the reaction mixture, adjust pH to 6-7 with sodium bicarbonate, separate layers, and crystallize the product from cyclohexane to obtain high-purity material.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this carbonylation-based route offers compelling economic and logistical benefits over traditional synthetic pathways. The elimination of column chromatography is perhaps the most significant value driver, as it removes a major bottleneck in production throughput and drastically reduces the consumption of high-purity silica gel and elution solvents. This simplification translates directly into lower operating expenditures (OPEX) and a reduced environmental footprint, aligning with corporate sustainability goals. Furthermore, the use of carbon monoxide as a feedstock is inherently more atom-economical than the multi-carbon reagents used in Wittig chemistry, leading to substantial cost savings in raw material procurement without compromising on the quality of the final reference standard.
- Cost Reduction in Manufacturing: The new process achieves significant cost optimization by removing the need for expensive stoichiometric reagents like Wittig salts, which are not only costly to purchase but also generate large volumes of solid waste that require disposal. By shifting to a catalytic system where the palladium complex is used in minute quantities relative to the substrate, the direct material cost per kilogram of product is markedly decreased. Additionally, the simplified workup involving crystallization rather than chromatography reduces labor hours and utility costs associated with solvent recovery, creating a leaner and more profitable manufacturing model for high-purity pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: Relying on commodity chemicals such as carbon monoxide, acetone, and hydrochloric acid insulates the supply chain from the volatility associated with specialized fine chemical reagents. The robustness of the reaction conditions, which tolerate standard industrial equipment like high-pressure autoclaves, ensures that production can be scaled up rapidly to meet fluctuating market demands. This stability is crucial for maintaining continuous supply lines to regulatory bodies and quality control laboratories that depend on consistent availability of impurity standards for batch release testing.
- Scalability and Environmental Compliance: The transition from batch chromatography to crystallization facilitates seamless scale-up from gram-scale laboratory synthesis to multi-kilogram commercial production. This scalability is supported by the reduced generation of hazardous waste, as the process avoids the massive solvent streams typical of purification columns. The ability to recycle the mother liquor from crystallization further enhances the green profile of the process, making it easier to obtain environmental permits and maintain compliance with increasingly stringent global regulations regarding industrial effluent discharge.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this carbonylation technology. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, offering practical guidance for technical teams evaluating this route for internal production or external sourcing.
Q: Why is the sulfonic acid-modified phosphine ligand critical in this synthesis?
A: The sulfonic acid group on the phosphine ligand (e.g., TPPTS) significantly enhances the solubility and stability of the palladium catalyst in the acidic reaction medium, preventing catalyst precipitation and ensuring high conversion rates compared to unmodified triphenylphosphine.
Q: How does this method improve purification compared to prior art?
A: Unlike previous methods requiring difficult column chromatography for acid products, this carbonylation route allows for simple crystallization from cyclohexane after a basic wash, eliminating silica gel usage and significantly reducing solvent waste.
Q: What are the typical reaction conditions for this carbonylation?
A: The reaction typically proceeds at elevated temperatures between 110-140°C under high carbon monoxide pressure (5-7 MPa), utilizing a strong acid promoter like hydrochloric acid to facilitate the rearrangement step.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-(4-isobutylphenyl)propionic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of having access to well-characterized impurity standards for robust quality control in pharmaceutical development. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistency and precision. We adhere to stringent purity specifications and utilize rigorous QC labs equipped with advanced analytical instrumentation to verify the identity and purity of every batch, guaranteeing that our materials serve as reliable benchmarks for your regulatory submissions.
We invite you to contact our technical procurement team to discuss how we can support your specific project needs. Whether you require a Customized Cost-Saving Analysis for switching to this novel synthetic route or need to request specific COA data and route feasibility assessments for other complex intermediates, our experts are ready to provide the tailored solutions necessary to accelerate your drug development timeline.
