Advanced Manufacturing Strategy for Evatanepag: Technical Breakthroughs and Commercial Scalability
Advanced Manufacturing Strategy for Evatanepag: Technical Breakthroughs and Commercial Scalability
The pharmaceutical industry is constantly seeking robust and efficient synthetic routes for novel therapeutic agents, particularly in the realm of ophthalmic treatments where precision and purity are paramount. Patent CN114591227A discloses a significant advancement in the preparation method of an EP2 receptor selective prostaglandin E2 agonist, commonly known as Evatanepag. This technology represents a pivotal shift from traditional, often cumbersome synthetic pathways to a more streamlined, cost-effective, and environmentally benign process. For R&D directors and procurement specialists alike, understanding the nuances of this patent is crucial for evaluating potential supply chain partnerships and optimizing manufacturing costs. The disclosed method not only simplifies the operational complexity but also ensures a high degree of control over impurity profiles, which is essential for regulatory compliance in the production of active pharmaceutical ingredients. By leveraging this intellectual property, manufacturers can achieve substantial improvements in yield and process safety, ultimately translating to a more reliable supply of high-purity pharmaceutical intermediates for the global market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of complex prostaglandin analogues has been plagued by several inherent challenges that hinder efficient commercial production. Conventional methods often rely on harsh reaction conditions, including the use of strong acids or bases at elevated temperatures, which can lead to the degradation of sensitive functional groups and the formation of difficult-to-remove by-products. Furthermore, many traditional routes require the use of expensive transition metal catalysts, such as palladium or platinum, which not only inflate the raw material costs but also introduce the risk of heavy metal contamination in the final product. This necessitates additional, costly purification steps to meet stringent regulatory limits for residual metals. Additionally, older processes frequently involve multiple protection and deprotection steps that increase the overall step count, thereby reducing the overall yield and extending the production lead time. These inefficiencies create significant bottlenecks in the supply chain, making it difficult to scale up production to meet commercial demand without compromising on quality or cost-effectiveness.
The Novel Approach
In contrast, the novel approach detailed in the patent data offers a transformative solution by utilizing mild reaction conditions and readily available reagents to construct the target molecule with high efficiency. The process eliminates the need for precious metal catalysts, relying instead on cost-effective bases like potassium carbonate and reducing agents like sodium borohydride. This strategic shift not only drastically reduces the raw material costs but also simplifies the downstream processing by removing the requirement for specialized metal scavenging steps. The synthetic route is designed to be linear and convergent, minimizing the number of isolation steps and thereby reducing material loss at each stage. Moreover, the workup procedures are optimized for simplicity, utilizing standard extraction and crystallization techniques that are easily adaptable to large-scale reactor systems. This approach ensures that the manufacturing process is not only chemically robust but also operationally safe, avoiding the risks associated with explosive or highly toxic reagents often found in legacy synthetic pathways.
Mechanistic Insights into the Four-Step Synthetic Route
The core of this synthetic strategy lies in a meticulously designed four-step sequence that builds the molecular complexity of Evatanepag in a controlled manner. The first step involves an etherification reaction where Compound 1 reacts with Compound 2 in the presence of potassium carbonate in DMF at 100°C. This nucleophilic substitution is critical for establishing the ether linkage that forms the backbone of the intermediate structure. The choice of DMF as a solvent ensures excellent solubility for the reactants, while the moderate temperature facilitates the reaction kinetics without promoting thermal degradation. Following this, the second step employs a reductive amination strategy using sodium borohydride in methanol. This step is pivotal for introducing the amine functionality with high chemoselectivity, ensuring that the aldehyde group is reduced efficiently without affecting other sensitive moieties. The use of ice bath cooling during the addition of the reducing agent further enhances control over the exothermic nature of the reaction, preventing runaway scenarios and ensuring consistent batch-to-batch quality.
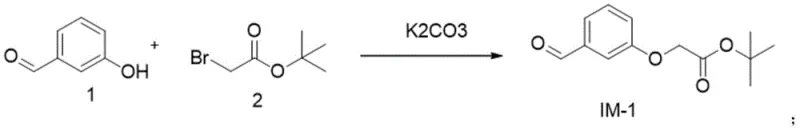
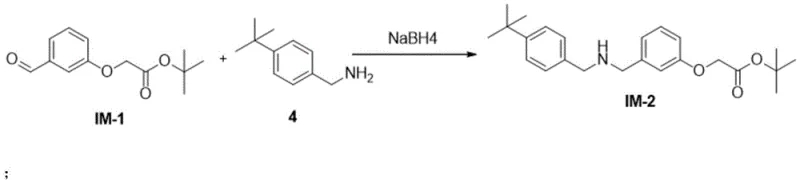
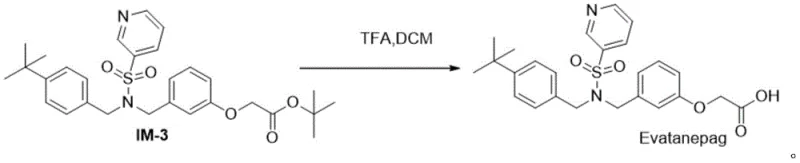
Continuing the synthesis, the third step involves the formation of a sulfonamide bond through the reaction of Intermediate IM-2 with Compound 6 in dichloromethane, facilitated by triethylamine. This coupling reaction is performed under mild conditions, typically starting at 0°C and warming to room temperature, which preserves the stereochemical integrity of the molecule. The final step is a deprotection reaction using trifluoroacetic acid, which cleaves the protecting group to reveal the active carboxylic acid functionality of Evatanepag. A key mechanistic advantage here is the precise pH control during workup; adjusting the pH to 5 induces the precipitation of the product while keeping impurities in solution. This crystallization-driven purification is a powerful tool for impurity rejection, ensuring that the final solid meets the rigorous purity specifications required for pharmaceutical applications without the need for extensive chromatographic purification.
How to Synthesize Evatanepag Efficiently
Implementing this synthetic route requires a thorough understanding of the process parameters to ensure optimal yield and safety. The procedure begins with the preparation of Intermediate IM-1, followed by its conversion to IM-2, then IM-3, and finally the target product. Each step has been optimized to balance reaction rate with selectivity, ensuring that side reactions are minimized. For R&D teams looking to replicate or scale this process, it is essential to adhere to the specified stoichiometry and temperature profiles, particularly during the exothermic reduction and deprotection stages. The detailed standardized synthesis steps provided in the technical documentation below offer a comprehensive guide for laboratory and pilot plant execution. By following these protocols, manufacturers can achieve consistent results and maintain the high quality standards expected in the production of complex pharmaceutical intermediates.
- Perform etherification of Compound 1 with Compound 2 using potassium carbonate in DMF at 100°C to yield IM-1.
- Conduct reductive amination of IM-1 with Compound 4 using sodium borohydride in methanol to generate IM-2.
- React IM-2 with Compound 6 in dichloromethane with triethylamine to form the sulfonamide intermediate IM-3.
- Deprotect IM-3 using trifluoroacetic acid followed by pH adjustment and recrystallization to obtain Evatanepag.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented process offers significant advantages that directly impact the bottom line and supply chain resilience. The elimination of expensive transition metal catalysts results in a substantial reduction in raw material costs, which is a critical factor for procurement managers negotiating supply contracts. Furthermore, the simplified workup procedures reduce the consumption of solvents and utilities, contributing to lower operational expenditures and a smaller environmental footprint. The use of common, commercially available reagents ensures that the supply chain is not vulnerable to shortages of specialized chemicals, thereby enhancing supply continuity. For supply chain heads, the robustness of this process means that production can be scaled up rapidly to meet market demand without the need for specialized equipment or extensive operator training. The overall efficiency of the route translates to shorter production cycles, allowing for faster turnaround times and improved responsiveness to customer needs.
- Cost Reduction in Manufacturing: The process achieves cost optimization primarily by removing the dependency on precious metal catalysts, which are not only expensive to purchase but also costly to dispose of in compliance with environmental regulations. By substituting these with inexpensive inorganic bases and reducing agents, the direct material cost per kilogram of product is significantly lowered. Additionally, the high yields reported in the patent examples indicate efficient atom economy, meaning less raw material is wasted as by-products. This efficiency extends to the purification stage, where the reliance on crystallization rather than preparative chromatography reduces solvent usage and processing time. These factors combined create a compelling economic case for adopting this method over traditional routes, offering significant cost savings in API manufacturing without compromising on product quality.
- Enhanced Supply Chain Reliability: The reliance on readily available starting materials such as potassium carbonate, sodium borohydride, and common organic solvents ensures a stable and resilient supply chain. Unlike processes that depend on custom-synthesized reagents or materials with long lead times, this route can be initiated quickly using stock chemicals available from multiple global suppliers. This reduces the risk of production delays caused by raw material shortages and allows for more flexible inventory management. Furthermore, the mild reaction conditions reduce the wear and tear on manufacturing equipment, leading to lower maintenance costs and higher equipment availability. For supply chain planners, this reliability translates to more accurate forecasting and the ability to commit to tighter delivery schedules with confidence.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, with reaction conditions that are easily transferable from laboratory flasks to industrial reactors. The absence of high-pressure or high-temperature requirements simplifies the engineering controls needed for safe operation, facilitating faster technology transfer and scale-up. From an environmental standpoint, the method aligns with green chemistry principles by minimizing waste generation and avoiding the use of toxic heavy metals. This simplifies waste treatment protocols and reduces the regulatory burden associated with hazardous waste disposal. The ability to produce high-purity products with minimal environmental impact is increasingly important for meeting corporate sustainability goals and complying with evolving environmental regulations in the chemical industry.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and supply of Evatanepag intermediates. These answers are derived directly from the technical specifications and beneficial effects outlined in the patent data, providing clarity on process capabilities and quality assurance measures. Understanding these details is essential for stakeholders evaluating the feasibility of this manufacturing route for their specific applications. The information provided here serves as a foundational reference for further technical discussions and feasibility assessments.
Q: What are the critical purity control points in Evatanepag synthesis?
A: Critical control points include the removal of inorganic salts after the initial etherification and the precise pH adjustment to 5 during the final deprotection step to ensure optimal precipitation of the target product while leaving impurities in the filtrate.
Q: Is the process scalable for industrial production?
A: Yes, the process utilizes readily available reagents like potassium carbonate and sodium borohydride, and avoids hazardous high-pressure conditions, making it highly suitable for commercial scale-up from kilogram to multi-ton batches.
Q: How does this method reduce environmental impact?
A: The method avoids the use of heavy metal catalysts and generates minimal pollutant waste, aligning with green chemistry principles and simplifying waste treatment protocols for manufacturing facilities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Evatanepag Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthetic routes in the modern pharmaceutical landscape. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from development to market. Our commitment to quality is underscored by our stringent purity specifications and rigorous QC labs, which guarantee that every batch of Evatanepag intermediate meets the highest industry standards. We understand the complexities involved in the commercial scale-up of complex pharmaceutical intermediates and are equipped to handle the technical challenges associated with process optimization and impurity control. Partnering with us means gaining access to a team of dedicated scientists and engineers who are committed to delivering excellence in every aspect of the manufacturing process.
We invite you to collaborate with us to optimize your supply chain and achieve your production goals. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage you to reach out to us to request specific COA data and route feasibility assessments that will demonstrate the value of our manufacturing capabilities. By leveraging our expertise and infrastructure, you can ensure a reliable supply of high-purity Evatanepag intermediates that support your drug development timelines and commercial success. Let us help you navigate the complexities of chemical manufacturing with confidence and precision.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
