Optimizing Sulfaclozine Sodium Production: A Technical Breakdown for Global Supply Chains
Optimizing Sulfaclozine Sodium Production: A Technical Breakdown for Global Supply Chains
The global demand for effective anticoccidial agents in the poultry industry necessitates robust and scalable manufacturing processes for key active pharmaceutical ingredients. A pivotal advancement in this domain is detailed in patent CN102617489A, which outlines a superior preparation method for Sulfaclozine Sodium. This technical disclosure moves away from archaic high-temperature fusion techniques, introducing a refined solution-phase condensation followed by an innovative binary solvent purification strategy. For R&D directors and procurement specialists, understanding this shift is critical, as it directly impacts the purity profile, environmental footprint, and cost-efficiency of the supply chain. By leveraging phase transfer catalysis and optimized crystallization dynamics, this method addresses the longstanding challenges of by-product formation and saline wastewater generation that have plagued traditional manufacturing routes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
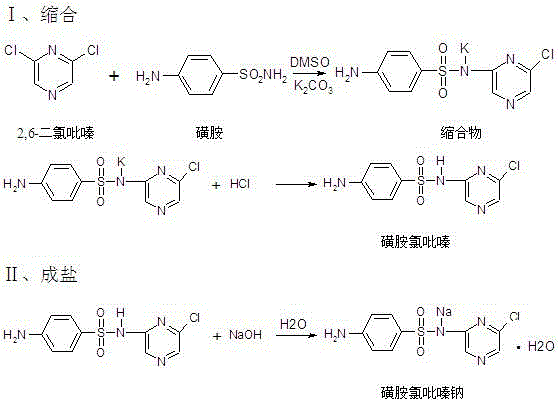
Historically, the industrial synthesis of Sulfaclozine Sodium relied heavily on a dry fusion method, as documented in earlier literature such as US2,475,673. In this legacy process, solid sulfanilamide, 2,6-dichloropyrazine, and anhydrous potassium carbonate were melted together at temperatures escalating rapidly from 123°C to 152°C. This aggressive thermal environment created significant operational hazards and chemical inefficiencies. The high heat promoted severe side reactions, specifically the condensation of one molecule of 2,6-dichloropyrazine with two molecules of sulfanilamide, resulting in the persistent impurity 2,6-bis-p-aminobenzenesulfonamidopyrazine. Furthermore, the purification of the crude product required a tedious 'acid-alkali' cycle involving dissolution in alkali, acidification to precipitate the free acid, and re-salting. This oscillating pH process not only consumed excessive amounts of acid and base but also generated voluminous quantities of saline wastewater, posing a heavy burden on environmental treatment facilities and complicating regulatory compliance.
The Novel Approach
The methodology presented in patent CN102617489A fundamentally re-engineers the synthesis pathway to mitigate these risks. Instead of a solvent-free melt, the reaction is conducted in toluene, a solvent chosen for its ability to form an azeotrope with water, thereby continuously removing the water by-product and driving the equilibrium forward. The introduction of a phase transfer catalyst, such as tetrabutylammonium bromide, facilitates the interaction between the organic reactants and the solid inorganic base, potassium carbonate. This allows the reaction to proceed smoothly at a much milder temperature range of 100-120°C. Consequently, the formation of the troublesome bis-sulfonamide impurity is drastically suppressed. Downstream processing is equally revolutionized; rather than the wasteful acid-alkali swing, the crude sodium salt is purified directly via recrystallization in a specific binary mixed solvent system comprising methanol and dichloromethane. This approach leverages differential solubility characteristics to isolate the target molecule with high efficiency while minimizing liquid waste discharge.
Mechanistic Insights into Phase Transfer Catalyzed Condensation
The core chemical innovation lies in the efficient nucleophilic substitution facilitated by the phase transfer catalyst (PTC). In the absence of a PTC, the reaction between the organic halide (2,6-dichloropyrazine) and the sulfonamide anion generated on the surface of solid potassium carbonate is diffusion-limited and slow. The quaternary ammonium salt acts as a molecular shuttle, transporting the reactive sulfonamide anion into the organic toluene phase where the concentration of the pyrazine derivative is high. This intimate contact accelerates the reaction kinetics without requiring the extreme thermal energy of the dry fusion method. The azeotropic removal of water is another critical mechanistic feature; by continuously stripping water from the reaction mixture via the toluene reflux, the reversible condensation reaction is pushed towards completion, significantly enhancing the conversion ratio of the starting materials.
Controlling the impurity profile is paramount for meeting stringent pharmacopoeial standards, particularly the European Pharmacopoeia requirement for single impurities below 0.2%. The traditional dry method often yielded impurity levels around 0.6%, which were difficult to remove due to similar physical properties. The new process minimizes the generation of this impurity at the source through temperature control. Furthermore, the binary solvent recrystallization exploits the subtle solubility differences between the mono-substituted target and the di-substituted impurity. By optimizing the mass ratio of methanol to dichloromethane at 2:1, the process ensures that the target Sulfaclozine Sodium crystallizes out with high purity (>99.5%), while the impurities remain in the mother liquor. This dual strategy of kinetic control during synthesis and thermodynamic control during purification ensures a robust quality profile suitable for sensitive veterinary applications.
How to Synthesize Sulfaclozine Sodium Efficiently
Implementing this synthesis route requires precise control over reaction parameters and solvent ratios to maximize yield and purity. The process begins with the condensation step in toluene, where maintaining the reflux temperature between 110°C and 115°C is critical for optimal water separation without degrading the reactants. Following the isolation of the intermediate sulfachlorpyrazine, the salt formation is conducted in an aqueous medium, followed by the crucial recrystallization step. The success of the purification hinges on the specific composition of the binary solvent system. Detailed operational protocols, including exact stirring rates, cooling gradients, and filtration parameters, are essential for reproducing the high yields reported in the patent data. For a comprehensive guide on executing these steps in a GMP environment, please refer to the standardized procedure outlined below.
- Condense sulfanilamide and 2,6-dichloropyrazine in toluene using potassium carbonate and a phase transfer catalyst at 100-120°C.
- React the resulting sulfachlorpyrazine intermediate with sodium hydroxide solution to form the crude sodium salt.
- Purify the crude product via recrystallization in a methanol and dichloromethane binary mixed solvent system.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel manufacturing process offers tangible strategic benefits beyond mere technical elegance. The elimination of the high-temperature dry fusion step reduces energy consumption and equipment wear, leading to a more sustainable production lifecycle. More importantly, the shift away from the acid-alkali refining method represents a significant reduction in waste management costs. By avoiding the generation of large volumes of saline wastewater, manufacturers can lower their environmental compliance overhead and reduce the risk of production stoppages due to effluent treatment bottlenecks. This streamlined workflow enhances the overall reliability of the supply chain, ensuring consistent delivery schedules for downstream formulators.
- Cost Reduction in Manufacturing: The adoption of phase transfer catalysis allows for the use of stoichiometric amounts of reactants rather than the large excesses required in dry fusion to drive the reaction. This optimization of raw material usage directly lowers the bill of materials. Additionally, the simplified purification process eliminates the need for multiple pH adjustment cycles and the associated consumption of hydrochloric acid and sodium hydroxide. The reduction in solvent complexity and waste disposal fees contributes to a leaner cost structure, allowing for more competitive pricing in the global veterinary drug market without compromising on quality margins.
- Enhanced Supply Chain Reliability: Traditional methods often suffered from batch-to-batch variability due to the difficulty in controlling the exothermic dry fusion reaction. The new solution-phase process offers superior thermal control and reproducibility, minimizing the risk of batch failures or off-spec material that could disrupt supply. The use of common, commercially available solvents like toluene, methanol, and dichloromethane ensures that raw material sourcing remains stable and unaffected by niche supply shocks. This reliability is crucial for maintaining continuous production lines for critical anticoccidial medications, especially during peak disease seasons in the poultry industry.
- Scalability and Environmental Compliance: Scaling up high-temperature melt reactions presents significant engineering challenges regarding heat dissipation and mixing efficiency. The proposed solvent-based method translates seamlessly from laboratory to pilot and commercial scales, as the heat transfer characteristics of a boiling solvent system are well-understood and easily managed in standard reactors. Furthermore, the drastic reduction in saline wastewater aligns with increasingly stringent global environmental regulations. This future-proofs the manufacturing site against tightening effluent standards, ensuring long-term operational continuity and reducing the likelihood of regulatory fines or mandatory shutdowns for environmental upgrades.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Sulfaclozine Sodium using this advanced methodology. These insights are derived directly from the experimental data and comparative analysis provided in the patent literature. Understanding these nuances helps stakeholders make informed decisions regarding vendor qualification and process validation. The answers reflect the consensus on best practices for achieving high-purity results while maintaining operational efficiency.
Q: How does the new method improve impurity profiles compared to dry fusion?
A: The novel solvent-based method utilizing phase transfer catalysis operates at lower temperatures (100-120°C) compared to the traditional dry fusion method (up to 152°C). This thermal control significantly suppresses the formation of the bis-sulfonamide by-product, ensuring compliance with strict pharmacopoeia limits for single impurities.
Q: What is the environmental advantage of the binary solvent recrystallization?
A: Traditional acid-alkali refining generates substantial volumes of saline wastewater due to repeated pH adjustments. The patented binary mixed solvent recrystallization (methanol/dichloromethane) eliminates the need for acidic precipitation and re-salting, drastically reducing wastewater load and simplifying effluent treatment.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process replaces hazardous high-temperature melt reactions with a controlled solution-phase reaction using common industrial solvents like toluene. The use of solid anhydrous potassium carbonate facilitated by phase transfer catalysts ensures robust mixing and heat transfer, making it highly scalable for multi-ton production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sulfaclozine Sodium Supplier
At NINGBO INNO PHARMCHEM, we recognize that the theoretical advantages of a patent must be translated into practical, commercial reality. Our technical team has extensively analyzed the pathway described in CN102617489A and possesses the expertise to implement this phase transfer catalyzed route at an industrial scale. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of reduced impurities and lower waste are realized in every batch we produce. Our facility is equipped with rigorous QC labs and adheres to stringent purity specifications, guaranteeing that our Sulfaclozine Sodium meets or exceeds the latest international pharmacopoeial requirements for veterinary use.
We invite global partners to collaborate with us to optimize their supply chains for anticoccidial agents. By switching to our optimized manufacturing process, you can secure a stable supply of high-quality intermediates while achieving significant sustainability goals. We encourage you to contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. Our experts are ready to provide specific COA data and route feasibility assessments to demonstrate how our advanced synthesis capabilities can add value to your final formulations.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
