Advanced Synthesis of Griseofulvin Derivatives for Commercial Antitumor Drug Development
Advanced Synthesis of Griseofulvin Derivatives for Commercial Antitumor Drug Development
The pharmaceutical landscape is constantly evolving, driven by the need to repurpose existing natural products into more potent therapeutic agents. A significant breakthrough in this domain is documented in patent CN112979665B, which discloses a novel class of griseofulvin Schmidt rearrangement derivatives. This technology represents a pivotal shift from the traditional antifungal application of griseofulvin to a promising antitumor profile. By leveraging the robust chemistry of the Schmidt rearrangement, researchers have successfully expanded the structural diversity of this spirocyclic benzofuran-3-one natural product. The resulting compounds, characterized by a unique seven-membered lactam ring, demonstrate superior inhibitory effects on tumor cell proliferation compared to the parent molecule. For global procurement and R&D teams, this patent offers a validated pathway for developing next-generation oncology intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, griseofulvin has been utilized almost exclusively as an antifungal antibiotic due to its ability to disrupt fungal mitosis by binding to tubulin. However, its utility in other therapeutic areas, particularly oncology, has been severely limited by its inherent structural rigidity and specific mode of action. Conventional chemical modifications of griseofulvin often struggle to maintain the integrity of the sensitive spirocyclic core while introducing functional groups that could enhance bioactivity against mammalian cells. Traditional synthetic routes frequently involve harsh conditions that lead to decomposition or low yields, making the production of diverse analogues economically unfeasible. Furthermore, the lack of a versatile handle for derivatization on the native structure restricts the ability of medicinal chemists to optimize pharmacokinetic properties. These limitations have hindered the exploration of griseofulvin scaffolds in the high-value anticancer market, leaving a gap for innovative synthetic strategies that can unlock new biological potential without compromising supply chain stability.
The Novel Approach
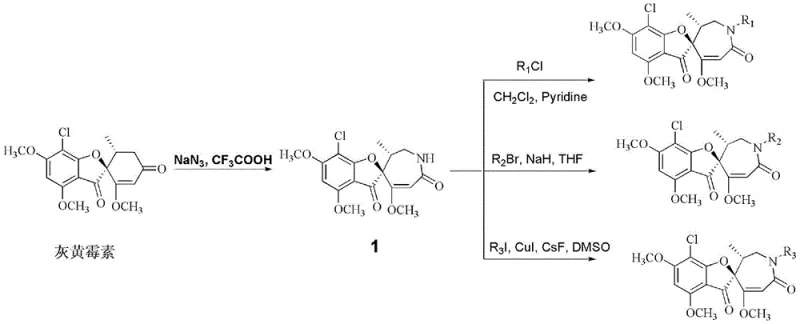
The methodology outlined in patent CN112979665B overcomes these historical barriers through a strategic ring-expansion strategy. By subjecting griseofulvin to a Schmidt rearrangement using sodium azide in a trifluoroacetic acid medium, the process efficiently converts the ketone functionality into a lactam, effectively expanding the ring size and introducing a nitrogen atom. This transformation creates a versatile intermediate (Intermediate 1) that serves as a powerful platform for further functionalization. Unlike previous methods that might degrade the complex stereochemistry, this approach preserves the critical chiral centers while opening up the nitrogen atom for acylation, alkylation, or arylation. The result is a library of derivatives, such as compounds 2a through 2f, which exhibit potent cytotoxicity against a broad spectrum of human cancer cell lines. This novel approach not only revitalizes a cheap and abundant natural product but also provides a reliable pharmaceutical intermediates supplier with a clear route to high-value oncology candidates.
Mechanistic Insights into Schmidt Rearrangement and Functionalization
The core of this synthetic innovation lies in the mechanistic elegance of the Schmidt rearrangement. In the presence of trifluoroacetic acid and sodium azide, the carbonyl group of griseofulvin is activated, facilitating the nucleophilic attack by the azide ion. This leads to the formation of an unstable azidohydrin intermediate, which subsequently undergoes a concerted migration of the adjacent carbon-carbon bond with the expulsion of nitrogen gas. This migration results in the insertion of a nitrogen atom into the carbon skeleton, forming the seven-membered lactam ring characteristic of Intermediate 1. The reaction conditions are meticulously optimized to proceed at room temperature, minimizing thermal degradation and ensuring high stereochemical fidelity. Following this key transformation, the secondary amine within the lactam ring becomes a reactive site for diversification. Whether through nucleophilic substitution with alkyl halides in the presence of strong bases like sodium hydride, or through copper-catalyzed coupling with aryl iodides, the chemistry allows for precise tuning of the molecule's electronic and steric properties. This level of control is essential for generating high-purity griseofulvin derivatives that meet the stringent requirements of drug development.
Impurity control is another critical aspect of this mechanism. The use of mild acidic conditions for the rearrangement step avoids the formation of complex polymeric byproducts often seen in harsher acid-catalyzed reactions. Furthermore, the subsequent derivatization steps utilize standard workup procedures, such as aqueous washes and column chromatography, which are highly effective at removing unreacted starting materials and inorganic salts. The specificity of the Schmidt rearrangement ensures that the major product is the desired ring-expanded lactam, simplifying the purification process significantly. For manufacturing teams, this translates to a cleaner reaction profile and reduced waste generation. The ability to isolate Intermediate 1 as a stable solid allows for a telescoped or stepwise production strategy, providing flexibility in process design. Understanding these mechanistic nuances is vital for scaling the process from laboratory benchtop to commercial scale-up of complex heterocycles, ensuring consistent quality and yield across batches.
How to Synthesize Griseofulvin Derivatives Efficiently
The synthesis protocol described in the patent provides a robust framework for producing these valuable antitumor intermediates. The process begins with the dissolution of griseofulvin in a mixture of trifluoroacetic acid and water, followed by the addition of sodium azide. This initial step is crucial as it sets the stage for the entire synthetic sequence by generating the key lactam scaffold. Once Intermediate 1 is isolated, it can be directed down three distinct pathways depending on the desired final substituent: acylation for acyl derivatives, alkylation for hydrocarbyl derivatives, or cross-coupling for aryl derivatives. Each pathway utilizes readily available reagents and standard solvent systems, making the technology accessible for implementation in various manufacturing settings. The detailed standardized synthesis steps for this efficient route are provided in the guide below.
- Perform Schmidt rearrangement of griseofulvin with sodium azide in trifluoroacetic acid and water at room temperature to obtain ring-expanded intermediate 1.
- Conduct acylation of intermediate 1 with acyl chlorides in pyridine to yield acyl derivatives, or alkylation with alkyl bromides using NaH in THF.
- Execute Buchwald-Hartwig coupling of intermediate 1 with iodoarenes using CuI and CsF in DMSO at elevated temperatures to produce aryl derivatives.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patent offers substantial benefits for organizations looking to optimize their supply chains and reduce manufacturing costs. The reliance on griseofulvin as a starting material is a significant strategic advantage, as it is a commercially available, fermentation-derived natural product produced on a massive scale globally. This abundance ensures a stable and cost-effective raw material supply, mitigating the risks associated with sourcing exotic or synthetic starting materials. The synthetic route itself is designed for efficiency, utilizing common laboratory and industrial reagents such as pyridine, acyl chlorides, and sodium hydride. This compatibility with standard chemical infrastructure means that existing facilities can often be adapted for production without requiring capital-intensive equipment upgrades. Consequently, this leads to cost reduction in API manufacturing by lowering both material and operational expenditures.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts in the initial rearrangement step is a major driver for cost efficiency. While the arylation step does utilize copper iodide, the loading is catalytic and the metal is relatively inexpensive compared to palladium or rhodium alternatives often used in similar couplings. Furthermore, the high yields reported for the derivatization steps minimize material loss, directly impacting the cost of goods sold. By streamlining the synthesis into fewer steps with high atom economy, manufacturers can achieve significant savings. The process avoids the need for cryogenic conditions or ultra-high pressure, further reducing energy consumption and utility costs associated with production.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions contributes to a more reliable supply chain. The tolerance of the Schmidt rearrangement to ambient temperatures reduces the risk of batch failures due to temperature excursions. Additionally, the stability of Intermediate 1 allows it to be stockpiled, acting as a buffer against fluctuations in demand for specific derivatives. This modularity means that a single batch of Intermediate 1 can be diverted to produce different end-products (acyl, alkyl, or aryl) based on real-time market needs. Such flexibility is invaluable for reducing lead time for high-purity intermediates, allowing suppliers to respond quickly to client requests without restarting the entire synthesis from the natural product.
- Scalability and Environmental Compliance: The scalability of this process is supported by the use of conventional solvents like ethyl acetate, tetrahydrofuran, and dimethyl sulfoxide, which are well-understood in terms of safety and waste management. The workup procedures involve standard aqueous extractions and drying, which are easily translated to large-scale separatory equipment. From an environmental standpoint, the avoidance of heavy metal waste in the primary rearrangement step simplifies effluent treatment. The overall green chemistry profile of the route, characterized by mild conditions and efficient transformations, aligns with modern regulatory standards for sustainable pharmaceutical manufacturing, facilitating smoother approval processes for new drug applications.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these griseofulvin derivatives. The answers are derived directly from the experimental data and technical specifications provided in patent CN112979665B. They are intended to clarify the feasibility of the synthesis and the potential applications of the resulting compounds in drug discovery. Understanding these details is essential for stakeholders evaluating the technology for licensing or contract manufacturing opportunities.
Q: What is the primary biological advantage of these griseofulvin derivatives?
A: Unlike native griseofulvin which is primarily antifungal, these Schmidt rearrangement derivatives exhibit significantly enhanced cytotoxic activity against various human tumor cell lines, including esophageal, gastric, and lung cancer cells.
Q: How does the Schmidt rearrangement modify the griseofulvin structure?
A: The reaction expands the original spirocyclic ketone ring into a seven-membered lactam ring system, introducing a nitrogen atom that allows for diverse N-substitution, thereby altering the pharmacological profile.
Q: Are the reaction conditions suitable for large-scale manufacturing?
A: Yes, the process utilizes common reagents like sodium azide, acyl chlorides, and standard solvents such as THF and DMSO, avoiding exotic catalysts and enabling scalable production under controlled conditions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Griseofulvin Derivatives Supplier
The technological advancements presented in patent CN112979665B highlight the immense potential of griseofulvin derivatives in the fight against cancer. At NINGBO INNO PHARMCHEM, we recognize the complexity involved in translating such innovative academic research into commercial reality. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the specific requirements of Schmidt rearrangement chemistry, ensuring stringent purity specifications and rigorous QC labs validate every batch. We are committed to delivering high-quality intermediates that accelerate your drug development timelines while maintaining the highest standards of safety and compliance.
We invite you to collaborate with us to explore the full potential of this synthetic route. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and target specifications. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you secure a competitive edge in the oncology market with our reliable supply of advanced griseofulvin intermediates.
