Advanced Aqueous Synthesis of Quinoline Piperazine Hydrochloride for Malaria Treatment Intermediates
Advanced Aqueous Synthesis of Quinoline Piperazine Hydrochloride for Malaria Treatment Intermediates
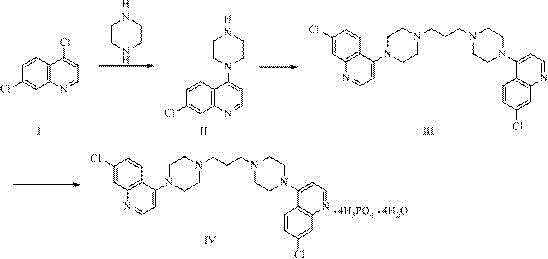
The global fight against malaria continues to drive demand for highly effective antimalarial agents, particularly piperaquine phosphate, which is a cornerstone of Artemisinin-based Combination Therapy (ACT). A critical bottleneck in the supply chain for this life-saving medication has historically been the synthesis of its key intermediate, 7-chloro-4-(1-piperazinyl) quinoline, often referred to as quinoline piperazine. The patent CN110606830B, published in March 2022, introduces a transformative manufacturing methodology that addresses long-standing purity and environmental challenges. This technical disclosure outlines a novel aqueous-phase synthesis route that achieves product purity greater than or equal to 99.5 percent while eliminating the need for toxic organic solvents. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediates supplier, this innovation represents a significant leap forward in process chemistry, offering a pathway to reduce impurity profiles below the stringent 0.1 percent threshold required by international markets.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of quinoline piperazine has been plagued by inefficient purification protocols and hazardous reaction conditions. Prior art methods, such as those disclosed in US20060270852 and CN101440063, typically rely on refluxing 4,7-dichloroquinoline with anhydrous piperazine in organic solvents like isopropanol or butanol for extended periods, often exceeding 36 hours. These conventional routes suffer from severe drawbacks, including the formation of difficult-to-remove by-products, specifically the bis-quinolinepiperazine impurity (Formula II-a), which is known to possess toxicity concerns. Furthermore, the workup procedures in these legacy processes are notoriously cumbersome, requiring multiple extraction steps, vacuum distillation, and the use of large excesses of piperazine that are difficult to recover economically. The reliance on organic solvents not only increases the cost reduction in API manufacturing but also generates significant hazardous waste, complicating environmental compliance and supply chain sustainability.
The Novel Approach
In stark contrast, the method described in patent CN110606830B utilizes water as the primary reaction medium, fundamentally altering the thermodynamics and kinetics of the nucleophilic substitution. By carefully controlling the pH of the aqueous system between 6.0 and 8.0 using hydrochloric acid, the process selectively promotes the mono-substitution of 4,7-dichloroquinoline while suppressing the formation of the di-substituted impurity. This approach eliminates the need for complex liquid-liquid extractions; instead, the product precipitates directly from the reaction mixture upon cooling. A standout feature of this novel approach is the strategy of mother liquor recycling. Rather than expending energy to separate and recover unreacted piperazine from the aqueous filtrate, the process directly applies the mother liquor to the next production batch. This closed-loop system not only simplifies the operational workflow but also ensures consistent yield improvements over successive cycles, making it an ideal candidate for the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Aqueous Nucleophilic Substitution
The core chemical transformation involves the nucleophilic attack of the piperazine nitrogen on the C-4 position of the 4,7-dichloroquinoline ring. In traditional organic solvents, the high reactivity of piperazine often leads to over-alkylation, where a second molecule of dichloroquinoline reacts with the remaining nitrogen of the piperazine ring. This side reaction generates the bis-quinolinepiperazine by-product, which is structurally similar to the target molecule and difficult to separate via standard crystallization. The innovation in this patent lies in the modulation of nucleophilicity through pH control in an aqueous environment. By maintaining the pH between 6.5 and 7.0, a portion of the piperazine exists in its protonated form, which effectively reduces its nucleophilic strength just enough to prevent the second substitution event while still allowing the primary reaction to proceed efficiently at elevated temperatures of 90-100°C.
Furthermore, the solubility characteristics of the reactants and products in water play a crucial role in impurity control. The target product, quinoline piperazine hydrochloride, exhibits specific solubility properties that allow it to crystallize out of the hot aqueous solution upon cooling to 45-65°C, while many polar impurities and excess salts remain in the mother liquor. This in-situ purification mechanism is far superior to the extraction-based methods of the past. The structural integrity of the final product is maintained without the degradation often seen in harsh alkaline conditions used in older patents. By avoiding the formation of the toxic bis-substituted impurity shown in the structural analysis below, the process ensures that the intermediate meets the rigorous quality standards necessary for downstream conversion into piperaquine phosphate.
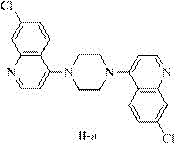
How to Synthesize Quinoline Piperazine Hydrochloride Efficiently
The implementation of this synthesis route requires precise control over reaction parameters to maximize yield and purity. The process begins with the preparation of an aqueous piperazine solution, where the stoichiometry is carefully managed to ensure a molar ratio of 4,7-dichloroquinoline to piperazine between 1:3 and 1:8. The reaction temperature is a critical variable, with the optimal range identified between 90°C and 100°C to ensure complete conversion within a reasonable timeframe of 4 to 5 hours. Following the reaction, the mixture is cooled to induce crystallization, and the solid product is isolated via filtration. The detailed standardized synthetic steps, including specific equipment setups and safety protocols for handling hydrochloric acid and hot slurries, are outlined in the guide below.
- Prepare the reaction system by mixing water and anhydrous piperazine, adjusting pH to 6.0-8.0 with hydrochloric acid before heating.
- Add 4,7-dichloroquinoline at 80-100°C and maintain reaction temperature for 4-5 hours to ensure complete conversion.
- Cool the mixture to 45-65°C, filter while hot to collect the product, and retain the mother liquor for the next batch cycle.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this aqueous-based methodology offers substantial strategic benefits beyond mere technical performance. The elimination of volatile organic compounds (VOCs) from the process significantly reduces the regulatory burden associated with solvent handling, storage, and disposal. This shift aligns perfectly with modern green chemistry initiatives and helps manufacturing sites maintain continuous operation without interruptions due to environmental compliance audits. Moreover, the simplicity of the workup—essentially a heat-cool-filter sequence—drastically reduces the labor and utility costs associated with distillation and extraction units. This operational efficiency translates directly into a more robust and resilient supply chain for high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The most significant economic driver of this technology is the innovative mother liquor recycling protocol. In traditional processes, recovering unreacted piperazine requires energy-intensive evaporation of water or complex distillation of organic solvents. By directly reusing the filtrate for up to four consecutive batches, the facility eliminates the capital and operational expenditure associated with solvent recovery systems. This approach minimizes raw material waste and lowers the overall cost of goods sold (COGS) without compromising product quality, providing a clear competitive advantage in price-sensitive markets.
- Enhanced Supply Chain Reliability: The reliance on water as a solvent removes the supply chain risks associated with fluctuating prices and availability of specialty organic solvents like butanol or isopropanol. Water is universally available and inexpensive, ensuring that production can continue uninterrupted regardless of petrochemical market volatility. Additionally, the shortened processing time—reducing reaction times from 36 hours in legacy methods to approximately 5 hours—increases the throughput capacity of existing reactor vessels, allowing suppliers to respond more rapidly to surges in demand for antimalarial medications.
- Scalability and Environmental Compliance: Scaling this process from pilot plant to commercial production is straightforward due to the absence of exothermic hazards often associated with organic solvent reactions. The aqueous system has a high heat capacity, providing a natural safety buffer during the exothermic addition of reactants. Furthermore, the wastewater generated is primarily saline and contains minimal organic load compared to solvent-heavy processes, simplifying effluent treatment. This environmental profile facilitates easier permitting for new manufacturing lines and supports the corporate sustainability goals of multinational pharmaceutical partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, offering clarity on how this method outperforms historical benchmarks in terms of purity, yield, and operational simplicity. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this technology for large-scale production.
Q: How does this method improve purity compared to traditional organic solvent routes?
A: By utilizing a controlled aqueous environment with precise pH adjustment (6.0-8.0), the process significantly suppresses the formation of the bis-substituted byproduct (Formula II-a), achieving purity levels exceeding 99.5% without complex extraction steps.
Q: What are the economic benefits of the mother liquor recycling technique?
A: The process allows the direct reuse of the filtrate (mother liquor) containing unreacted piperazine for up to four subsequent batches, eliminating the energy-intensive distillation steps typically required for solvent recovery and drastically reducing raw material consumption.
Q: Is this process scalable for industrial production of piperaquine phosphate intermediates?
A: Yes, the method avoids toxic organic solvents and cumbersome extraction procedures, relying instead on simple filtration and water-based chemistry, which simplifies waste treatment and facilitates safe scale-up from laboratory to multi-ton commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Quinoline Piperazine Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize that the quality of the final antimalarial drug is inextricably linked to the purity of its intermediates. Our technical team has extensively analyzed the process described in CN110606830B and possesses the expertise to implement this advanced aqueous synthesis on an industrial scale. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that every batch of quinoline piperazine hydrochloride meets stringent purity specifications of greater than 99.5 percent. Our rigorous QC labs are equipped to detect trace impurities, including the problematic bis-substituted by-products, guaranteeing that our materials are fully compliant with international pharmacopoeia standards.
We invite global pharmaceutical partners to collaborate with us to optimize their supply chains for piperaquine phosphate production. By leveraging our manufacturing capabilities, you can secure a stable source of high-quality intermediates while benefiting from the cost efficiencies of this green chemistry approach. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our optimized processes can enhance your production efficiency and reduce lead time for high-purity pharmaceutical intermediates.
