Scalable Metal-Free Synthesis of Thiosulfonates Using DABSO for Pharmaceutical Applications
Introduction to Advanced Thiosulfonate Manufacturing
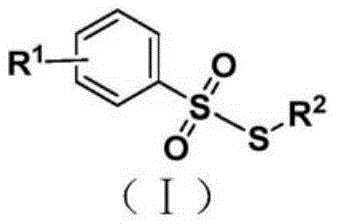
The landscape of organosulfur chemistry is undergoing a significant transformation driven by the demand for safer and more efficient synthetic methodologies. Patent CN109553557B introduces a groundbreaking approach for the synthesis of thiosulfonate compounds, utilizing a sulfur dioxide insertion strategy that fundamentally alters the traditional risk profile of these reactions. This technology leverages DABSO (1,4-diazabicyclo[2.2.2]octane bis(sulfur dioxide)) as a stable, solid surrogate for gaseous sulfur dioxide, enabling the construction of valuable S-S bonds under remarkably mild conditions. For R&D directors and procurement specialists in the pharmaceutical and agrochemical sectors, this represents a pivotal shift towards sustainable manufacturing practices that do not compromise on yield or purity. The method employs readily available aryl diazo tetrafluoroborate and thiophenol derivatives, catalyzed by trifluoroacetic acid in a mixed solvent system, ensuring high functional group tolerance.
Furthermore, the strategic implementation of this synthesis route addresses critical supply chain vulnerabilities associated with hazardous gas handling. By replacing toxic SO2 gas with a bench-stable solid, the process facilitates easier logistics and storage, which is a paramount concern for supply chain heads managing global inventory. The reaction proceeds efficiently under a nitrogen atmosphere at temperatures ranging from 40°C to 80°C, completing within short timeframes of 20 to 60 minutes. This efficiency translates directly into reduced energy consumption and increased throughput capacity for commercial production facilities. As we delve deeper into the technical specifics, it becomes evident that this patent offers a robust platform for generating high-purity pharmaceutical intermediates with a significantly reduced environmental footprint compared to legacy oxidation methods.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of thiosulfonate backbones has been plagued by significant synthetic challenges that hinder large-scale adoption in fine chemical manufacturing. The classical approach typically involves the oxidation of disulfides to their corresponding thiosulfonates, a pathway that suffers from severe selectivity issues, particularly when asymmetric disulfides are employed. This lack of regiocontrol inevitably leads to the formation of complex isomeric mixtures, necessitating costly and time-consuming purification processes that erode overall process economics. Additionally, alternative literature methods often rely on the use of benzenesulfonyl hydrazides or sodium sulfinates, which can introduce impurities that are difficult to remove to the stringent standards required for active pharmaceutical ingredients. Many of these traditional routes also mandate the use of transition metal catalysts, introducing the risk of heavy metal contamination that requires additional scavenging steps, further driving up production costs and extending lead times.
The Novel Approach
In stark contrast, the methodology disclosed in the patent data presents a streamlined, one-pot solution that circumvents these historical bottlenecks through a clever sulfur dioxide insertion mechanism. By utilizing DABSO as the sulfur source, the reaction avoids the generation of isomeric byproducts common in disulfide oxidation, ensuring a cleaner reaction profile and higher crude purity. The metal-free nature of this catalytic system is a distinct advantage, as it eliminates the need for expensive palladium or copper catalysts and the associated regulatory burden of residual metal testing. The use of trifluoroacetic acid as an additive promotes the reaction under mild thermal conditions, preserving sensitive functional groups that might otherwise degrade under harsh oxidative environments. This novel approach not only simplifies the operational workflow but also enhances the overall atom economy, making it an ideal candidate for cost reduction in pharmaceutical intermediates manufacturing where margin pressure is intense.
Mechanistic Insights into Sulfur Dioxide Insertion Strategy
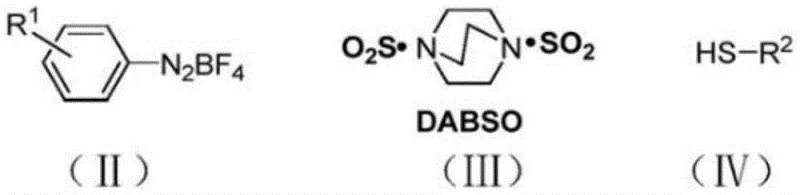
The core innovation of this synthesis lies in the mechanistic pathway where DABSO serves as a controlled release vehicle for sulfur dioxide within the reaction matrix. Upon heating in the presence of trifluoroacetic acid and the diazonium salt, DABSO liberates SO2 in situ, which then inserts into the transient aryl species generated from the diazonium precursor. This insertion forms a reactive sulfonyl intermediate that subsequently couples with the thiol nucleophile to forge the target S-S bond. The beauty of this mechanism is its reliance on the inherent reactivity of the diazonium salt, which acts as a potent electrophile, thereby negating the need for external oxidative promoters or metal catalysts to drive the transformation. The reaction environment, maintained under an inert nitrogen atmosphere, prevents unwanted side reactions such as homocoupling of the thiol or oxidation of the sulfinate species, ensuring that the majority of the starting material is channeled towards the desired thiosulfonate product.
From an impurity control perspective, this mechanism offers superior predictability compared to radical-based oxidations. The defined stoichiometry between the aryldiazonium tetrafluoroborate, DABSO, and the thiol allows for precise tuning of the reaction to minimize excess reagents that could complicate downstream processing. The compatibility with a wide range of substituents, including electron-withdrawing halogens and electron-donating alkyl groups on both the diazonium and thiol components, underscores the robustness of the catalytic cycle. This broad substrate scope is critical for R&D teams looking to diversify their chemical libraries without needing to re-optimize reaction conditions for every new analog. The stability of the intermediates formed during the SO2 insertion ensures that the reaction can be scaled up with confidence, as the exothermic profiles are manageable and the risk of runaway reactions is significantly mitigated by the solid-state nature of the sulfur source.
How to Synthesize Thiosulfonates Efficiently
Executing this synthesis on a laboratory or pilot scale requires strict adherence to the optimized parameters outlined in the patent to ensure maximum yield and reproducibility. The process begins with the careful preparation of the reaction vessel, which must be purged with nitrogen to create an oxygen-free environment essential for preventing thiol oxidation. Reagents are added in a specific sequence, typically starting with the acid additive and the solid components, followed by the injection of the solvent mixture to initiate the reaction upon heating. The choice of solvent, a blend of 1,2-dichloroethane and toluene, is crucial for solubilizing both the organic salts and the DABSO complex while maintaining the appropriate boiling point for the desired reaction temperature window. Operators must monitor the reaction progress closely, as the conversion is typically rapid, occurring within 20 to 60 minutes depending on the electronic nature of the substrates involved.
- Prepare the reaction vessel by filling it with nitrogen and adding trifluoroacetic acid, aryldiazonium tetrafluoroborate, thiol compound, and DABSO.
- Add a mixed solvent of DCE and toluene under nitrogen atmosphere and stir the mixture in an oil bath at 40°C to 80°C for 20 to 60 minutes.
- Upon completion, cool the solution, remove solvents via rotary evaporation, and purify the residue using silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this DABSO-mediated synthesis route offers tangible strategic benefits that extend beyond simple chemical yield. The elimination of gaseous sulfur dioxide from the supply chain removes a major logistical hazard, reducing insurance costs and simplifying regulatory compliance regarding hazardous material transport and storage. Furthermore, the metal-free protocol drastically simplifies the purification workflow, as there is no need for specialized resin columns or complex extraction procedures to remove trace transition metals, which directly translates to lower operational expenditures and faster batch release times. The use of commercially available and stable starting materials ensures a reliable supply chain, minimizing the risk of production delays caused by the scarcity of exotic catalysts or unstable reagents. This reliability is essential for maintaining continuous manufacturing operations in the highly regulated pharmaceutical sector.
- Cost Reduction in Manufacturing: The economic impact of switching to this metal-free methodology is profound, primarily driven by the removal of expensive transition metal catalysts and the associated waste disposal costs. Traditional methods often require stoichiometric amounts of oxidants or precious metal complexes, which represent a significant portion of the raw material bill; by contrast, this method utilizes cost-effective organic salts and a reusable amine-SO2 complex. Additionally, the simplified workup procedure reduces solvent consumption and labor hours required for purification, leading to substantial cost savings in the overall production budget. The high atom economy of the insertion strategy ensures that a greater proportion of the input mass is converted into saleable product, further enhancing the financial viability of the process for large-scale commercialization.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the use of DABSO, a shelf-stable solid that can be sourced reliably without the volatility associated with compressed gas cylinders. This stability allows for bulk purchasing and long-term storage without degradation, providing a buffer against market fluctuations and supply disruptions. The robustness of the reaction conditions, which tolerate a wide variety of functional groups, means that the same protocol can be applied to synthesize a diverse range of intermediates without requiring extensive re-validation or equipment changes. This flexibility enables manufacturers to respond quickly to changing market demands and custom synthesis requests, ensuring that delivery schedules are met consistently even when dealing with complex molecular architectures.
- Scalability and Environmental Compliance: Scaling this process from gram to tonnage levels is facilitated by the mild thermal requirements and the absence of hazardous gas feeds, which simplifies reactor design and safety engineering. The reduced environmental pollution profile aligns perfectly with modern green chemistry initiatives, as the process generates less hazardous waste and avoids the emission of toxic sulfur gases into the atmosphere. This compliance with stringent environmental regulations reduces the administrative burden on EHS teams and minimizes the risk of fines or shutdowns due to non-compliance. The ability to run the reaction in standard glass-lined or stainless steel reactors without special corrosion-resistant coatings for SO2 gas further lowers the capital expenditure required for facility upgrades, making it an attractive option for contract manufacturing organizations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this sulfur dioxide insertion strategy in industrial settings. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear understanding of the method's capabilities and limitations. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios. The answers reflect the consensus on safety, efficiency, and applicability across different classes of thiosulfonate derivatives.
Q: What are the primary safety advantages of using DABSO over sulfur dioxide gas?
A: DABSO acts as a solid surrogate for sulfur dioxide, eliminating the need for handling toxic and corrosive SO2 gas, thereby significantly enhancing operational safety and simplifying reactor requirements.
Q: Does this synthesis method require expensive transition metal catalysts?
A: No, the method described in patent CN109553557B is entirely metal-free, which reduces raw material costs and eliminates the need for complex heavy metal removal steps during purification.
Q: What is the substrate scope for this thiosulfonate synthesis strategy?
A: The protocol demonstrates excellent functional group compatibility, accommodating various substituted aryldiazonium salts and thiols, including those with halogen, alkyl, and alkoxy groups.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Thiosulfonates Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the DABSO-based sulfur dioxide insertion strategy for producing high-value organosulfur compounds. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale discovery to full-scale manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped to handle the specific requirements of metal-free synthesis, featuring rigorous QC labs that enforce stringent purity specifications to meet the exacting standards of the global pharmaceutical industry. We are committed to delivering consistent quality and supply continuity, leveraging our deep technical expertise to optimize every step of the production process for maximum yield and minimal environmental impact.
We invite you to collaborate with us to leverage this advanced synthetic technology for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and target specifications. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to innovation and quality can drive value for your organization. Together, we can accelerate the development of next-generation therapeutics and agrochemicals using safer, more sustainable chemistry.
