Advanced Synthesis of 7-Methoxycoumarin-3-Carboxylic Acid for Commercial Scale-Up
Advanced Synthesis of 7-Methoxycoumarin-3-Carboxylic Acid for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking robust, high-yield synthetic routes for complex heterocyclic compounds, particularly those serving as critical intermediates for therapeutic agents. Patent CN102924415A introduces a groundbreaking preparation method for 7-methoxycoumarin-3-carboxylic acid, also known as ayapanin-3-carboxylic acid, a compound with significant potential in the treatment of gout and related inflammatory conditions. This technical insight report analyzes the novel methodology disclosed in the patent, highlighting its superiority over traditional extraction methods which often suffer from batch variability and low purity. By leveraging a direct condensation strategy, this process offers a streamlined pathway that addresses the critical pain points of reproducibility and scalability faced by modern reliable pharmaceutical intermediate supplier networks globally.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the acquisition of bioactive coumarin derivatives like ayapanin has relied heavily on the extraction of natural sources, such as the bark of ash trees, or through convoluted multi-step organic syntheses that are inefficient for industrial application. As noted in the background technology of the patent, traditional Chinese medicine extracts containing total coumarins face severe challenges regarding quality control due to variations in raw material origin and batch differences, making it difficult to standardize clinical efficacy. Furthermore, existing synthetic patents, such as those describing the synthesis of furan-tonka bean camphor derivatives, often require harsh phase-transfer catalysts, strong alkalis, or complex purification sequences that generate substantial chemical waste. These conventional approaches frequently result in lower overall yields and impurity profiles that are difficult to characterize, posing significant risks for cost reduction in pharmaceutical intermediates manufacturing where consistency is paramount.
The Novel Approach
In stark contrast to these legacy methods, the disclosed invention utilizes a highly efficient one-pot condensation reaction that dramatically simplifies the production workflow while maximizing output. The novel approach employs 4-methoxysalicylaldehyde and a cyclic active methylene compound (specifically a malonate derivative) reacting in the presence of piperidine within an acetonitrile solvent system. This strategic selection of reagents allows for the direct formation of the coumarin core with a carboxylic acid functionality already in place, eliminating the need for subsequent hydrolysis steps often required when using ester precursors. The process is characterized by a remarkably high yield of 93%, achieved through a controlled temperature profile starting at 0°C and progressing to reflux, ensuring that the reaction kinetics are optimized for maximum conversion. This method represents a paradigm shift towards high-purity pharmaceutical intermediates by providing a scientifically reasonable and reproducible protocol that minimizes side reactions.
Mechanistic Insights into Piperidine-Catalyzed Cyclization
The core of this synthetic breakthrough lies in the base-catalyzed Knoevenagel condensation followed by an intramolecular transesterification or cyclization event. The reaction initiates with the deprotonation of the active methylene group on the cyclic malonate derivative by piperidine, generating a nucleophilic enamine or carbanion species. This nucleophile attacks the electrophilic carbonyl carbon of the 4-methoxysalicylaldehyde, forming a beta-hydroxy intermediate which subsequently undergoes dehydration to establish the carbon-carbon double bond characteristic of the coumarin scaffold. The presence of the phenolic hydroxyl group ortho to the aldehyde is crucial, as it facilitates the ring closure to form the lactone moiety of the coumarin system. Understanding this mechanistic pathway is essential for commercial scale-up of complex pharmaceutical intermediates, as it highlights the sensitivity of the reaction to stoichiometry and temperature control.
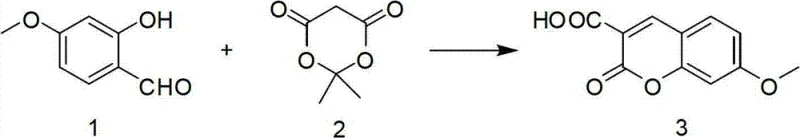
Furthermore, the choice of piperidine as the catalyst is instrumental in controlling the impurity profile of the final product. Unlike stronger inorganic bases that might promote polymerization or degradation of the sensitive aldehyde starting material, piperidine offers a moderate basicity that drives the condensation forward without compromising the structural integrity of the reactants. The subsequent reflux step ensures the completion of the cyclization and the removal of volatile by-products, such as acetone if a Meldrum's acid derivative is used, driving the equilibrium towards the product. The final recrystallization from methanol serves as a critical purification step, effectively removing unreacted starting materials and oligomeric by-products, thereby ensuring the stringent purity specifications required for downstream drug synthesis. This level of mechanistic control is what distinguishes a laboratory curiosity from a viable industrial process.
How to Synthesize 7-Methoxycoumarin-3-Carboxylic Acid Efficiently
Implementing this synthesis requires precise adherence to the molar ratios and thermal conditions outlined in the patent to replicate the reported 93% yield. The process begins with the dissolution of the aldehyde and the cyclic malonate in acetonitrile under cooling, followed by the careful addition of the amine catalyst to manage the exotherm and precipitation. Detailed standardized operating procedures are critical for maintaining batch-to-batch consistency, particularly during the reflux and filtration stages where product loss can occur if not managed correctly. For a comprehensive guide on the exact operational parameters, please refer to the technical steps below.
- Weigh 4-methoxysalicylaldehyde and the cyclic malonate derivative (molar ratio approx 1: 1.3) and dissolve in acetonitrile at 0°C.
- Add piperidine catalyst to the mixture, stir to induce precipitation, and react for one hour before refluxing overnight.
- Filter the resulting precipitate, dry the solid, and purify via recrystallization with methanol to obtain the final high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this patented methodology offers transformative advantages that directly impact the bottom line and operational reliability. By shifting from extraction-based or multi-step synthetic routes to this direct condensation method, manufacturers can significantly reduce the complexity of their supply chains and the associated logistical overheads. The use of commodity chemicals like 4-methoxysalicylaldehyde and acetonitrile ensures that raw material sourcing is stable and less susceptible to the geopolitical or agricultural fluctuations that affect natural extracts. This stability is crucial for reducing lead time for high-purity pharmaceutical intermediates, allowing for more accurate demand forecasting and inventory management.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the exceptional yield of 93%, which drastically reduces the cost per kilogram of the final active ingredient compared to lower-yielding alternatives. Additionally, the elimination of expensive transition metal catalysts or specialized phase-transfer agents simplifies the bill of materials and reduces the cost of goods sold. The simplified workup procedure, involving merely filtration and recrystallization rather than complex chromatographic separations, further lowers processing costs by reducing solvent consumption and labor hours. These factors combine to create a highly economical production model that enhances competitiveness in the global market.
- Enhanced Supply Chain Reliability: The reliance on widely available, commercially produced starting materials mitigates the risk of supply disruptions that are common with botanical extracts subject to seasonal harvest variations. The robustness of the chemical synthesis allows for production to be scheduled independently of agricultural cycles, ensuring a continuous flow of materials to downstream customers. Moreover, the scalability of the reaction, which proceeds efficiently in standard glass-lined or stainless steel reactors, means that production capacity can be ramped up quickly to meet surges in demand without requiring significant capital investment in new equipment.
- Scalability and Environmental Compliance: The process is inherently greener than many traditional methods due to the high atom economy of the condensation reaction and the ability to recover and recycle the acetonitrile solvent. The absence of heavy metal contaminants simplifies wastewater treatment protocols and ensures compliance with increasingly stringent environmental regulations regarding effluent discharge. The solid nature of the product allows for easy isolation and drying, minimizing the energy footprint associated with solvent removal and facilitating safe, dust-controlled handling in large-scale manufacturing facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 7-methoxycoumarin-3-carboxylic acid. These answers are derived directly from the technical specifications and beneficial effects described in patent CN102924415A, providing clarity for stakeholders evaluating this technology for integration into their supply chains. Understanding these details is vital for making informed decisions about sourcing strategies and process validation.
Q: What is the expected yield of this synthesis method?
A: According to patent CN102924415A, the optimized process achieves a yield of approximately 93%, which is significantly higher than many traditional extraction or multi-step synthetic routes.
Q: What are the key raw materials required?
A: The process utilizes commercially available 4-methoxysalicylaldehyde and a cyclic malonate derivative, along with piperidine as a catalyst and acetonitrile as the solvent.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the patent explicitly states that the method provides detailed theory and practice guidance for expanded production, citing simple workup procedures like filtration and recrystallization.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 7-Methoxycoumarin-3-Carboxylic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of next-generation therapeutics for gout and inflammation. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of 7-methoxycoumarin-3-carboxylic acid performs consistently in your downstream synthesis.
We invite you to collaborate with us to leverage this advanced synthetic route for your specific applications. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can optimize your supply chain and accelerate your time to market.
