Revolutionizing Cephalosporin Intermediates: Safe Scale-Up of 1-Tetrazole Acetate
Introduction to Advanced Tetrazole Chemistry
The pharmaceutical industry's relentless pursuit of safer, more efficient synthetic routes for critical antibiotic intermediates has led to significant breakthroughs in heterocyclic chemistry, specifically regarding the production of 1-tetrazole acetate. As detailed in the pioneering patent CN111393382A, a novel preparation method has been established that fundamentally shifts the paradigm away from hazardous reagents toward a greener, more sustainable manufacturing protocol. This technology addresses the critical need for high-purity intermediates used in the synthesis of cephalosporin antibiotics such as cefazolin and ceftezole, which are vital for global healthcare infrastructure. By replacing traditional, high-risk starting materials with stable aminoguanidine carbonate, this process not only mitigates safety hazards associated with explosive compounds but also streamlines the purification workflow, resulting in a product with superior impurity profiles. For R&D directors and procurement specialists alike, understanding the mechanistic advantages of this route is essential for securing a resilient supply chain capable of meeting the rigorous quality standards of modern GMP manufacturing environments.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of tetrazole acetic acid derivatives has been plagued by significant safety and environmental challenges, primarily due to the reliance on sodium azide as a key nitrogen source. As illustrated in the reaction pathways of prior art, the condensation of glycine with formic acid or triethyl orthoformate followed by reaction with sodium azide presents severe operational risks. 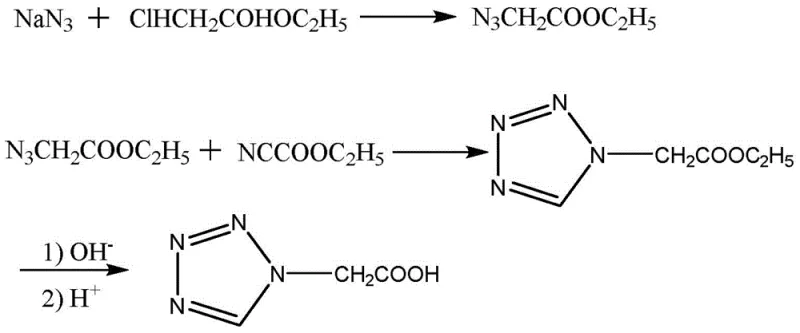 These conventional routes often involve the use of hypertoxic compounds like methyl chloroformate or ethyl cyanoformate, which are strictly regulated due to their high toxicity and potential for misuse, thereby complicating logistics and storage. Furthermore, the use of hydrazine hydrate in alternative pathways introduces extreme toxicity concerns, with an LD50 as low as 129mg/kg, posing unacceptable risks to personnel and requiring elaborate waste treatment systems to prevent environmental contamination. The inherent instability of azide intermediates also necessitates specialized equipment and rigorous temperature controls to prevent runaway exothermic reactions, driving up capital expenditure and operational costs significantly. Additionally, these older methods frequently suffer from poor regioselectivity during the ring-closing steps, leading to complex mixtures of isomers that are difficult and costly to separate, ultimately reducing the overall yield and purity of the final API intermediate.
These conventional routes often involve the use of hypertoxic compounds like methyl chloroformate or ethyl cyanoformate, which are strictly regulated due to their high toxicity and potential for misuse, thereby complicating logistics and storage. Furthermore, the use of hydrazine hydrate in alternative pathways introduces extreme toxicity concerns, with an LD50 as low as 129mg/kg, posing unacceptable risks to personnel and requiring elaborate waste treatment systems to prevent environmental contamination. The inherent instability of azide intermediates also necessitates specialized equipment and rigorous temperature controls to prevent runaway exothermic reactions, driving up capital expenditure and operational costs significantly. Additionally, these older methods frequently suffer from poor regioselectivity during the ring-closing steps, leading to complex mixtures of isomers that are difficult and costly to separate, ultimately reducing the overall yield and purity of the final API intermediate.
The Novel Approach
In stark contrast to these hazardous legacy processes, the innovative methodology disclosed in patent CN111393382A utilizes aminoguanidine carbonate as a stable, non-explosive starting material, effectively eliminating the need for handling bulk sodium azide or hydrazine hydrate. This new route initiates with a controlled diazotization and cyclization sequence that generates 5-aminotetrazole under relatively mild acidic and then alkaline conditions, ensuring a much safer reaction profile. 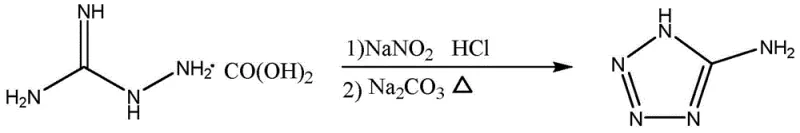 By shifting the synthetic strategy to build the tetrazole ring first and then performing the alkylation, the process leverages the electronic properties of the amino group to enhance regioselectivity, a critical factor in minimizing isomeric impurities. The elimination of highly regulated dangerous chemicals simplifies the regulatory compliance burden and reduces the need for specialized hazardous material storage facilities, offering immediate logistical advantages. Moreover, the aqueous nature of several steps in this new protocol aligns perfectly with green chemistry principles, drastically reducing the volume of organic solvent waste and lowering the environmental footprint of the manufacturing process. This approach not only safeguards the workforce but also ensures a more consistent and reliable production output, making it an ideal candidate for large-scale commercial adoption in the competitive pharmaceutical intermediate market.
By shifting the synthetic strategy to build the tetrazole ring first and then performing the alkylation, the process leverages the electronic properties of the amino group to enhance regioselectivity, a critical factor in minimizing isomeric impurities. The elimination of highly regulated dangerous chemicals simplifies the regulatory compliance burden and reduces the need for specialized hazardous material storage facilities, offering immediate logistical advantages. Moreover, the aqueous nature of several steps in this new protocol aligns perfectly with green chemistry principles, drastically reducing the volume of organic solvent waste and lowering the environmental footprint of the manufacturing process. This approach not only safeguards the workforce but also ensures a more consistent and reliable production output, making it an ideal candidate for large-scale commercial adoption in the competitive pharmaceutical intermediate market.
Mechanistic Insights into Diazotization and Regioselective Alkylation
The core chemical innovation of this process lies in the precise control of reaction conditions during the formation of the tetrazole ring and the subsequent N-alkylation step. In the first stage, aminoguanidine carbonate is dissolved in hydrochloric acid and cooled to below 10°C before the dropwise addition of sodium nitrite, a critical exothermic diazotization step that must be carefully managed to prevent the decomposition of the diazonium intermediate. Following this, the pH is adjusted to above 9 using sodium carbonate, which triggers the intramolecular cyclization to form 5-aminotetrazole; maintaining a pH greater than 9 is essential to maximize precipitation and drive the equilibrium toward the desired product. The second stage involves the nucleophilic substitution of 5-aminotetrazole with chloroacetic acid, where the presence of the amino group plays a pivotal role in directing the alkylation. Due to the resonance structures of the tetrazole ring, the pKa values of the nitrogen atoms at the 1 and 2 positions are typically very similar, often leading to a roughly 1:1 mixture of isomers in unsubstituted systems. However, the electron-donating effect of the 5-amino substituent alters the electron density distribution across the ring, effectively differentiating the nucleophilicity of the nitrogen atoms and improving the selectivity for the 1-position substituted product from a baseline of approximately 50% to an optimized level of around 75%. This mechanistic advantage significantly reduces the burden on downstream purification units, as fewer isomeric impurities need to be removed to meet stringent pharmaceutical specifications.
Furthermore, the final deamination step employs a reductive strategy using agents such as hypophosphorous acid or lower alcohols in the presence of concentrated sulfuric acid to remove the amino group without disrupting the sensitive tetrazole ring. This reduction is conducted at low temperatures, typically between 0°C and 5°C, to ensure the stability of the intermediate and prevent side reactions that could compromise the integrity of the heterocyclic core. The use of hypophosphorous acid is particularly advantageous as it acts as a clean reducing agent that generates water-soluble byproducts, facilitating easy separation of the organic product via extraction with solvents like ethyl acetate. The entire sequence is designed to minimize the formation of tarry byproducts and colored impurities, which are common pitfalls in tetrazole chemistry, thereby yielding a white crystalline powder with high optical purity. By understanding these mechanistic nuances, process chemists can better optimize parameters such as addition rates, temperature gradients, and stoichiometric ratios to further enhance the robustness of the synthesis for multi-ton scale production.
How to Synthesize 1-Tetrazole Acetate Efficiently
The synthesis of 1-tetrazole acetate via this patented route involves a streamlined three-step sequence that prioritizes safety and yield optimization at every stage. The process begins with the conversion of aminoguanidine carbonate into 5-aminotetrazole through a diazotization-cyclization cascade, followed by a regioselective N-alkylation with chloroacetic acid, and concludes with a reductive deamination to furnish the final target molecule. Each step has been rigorously tested to ensure reproducibility, with specific attention paid to pH control and temperature management to maximize conversion rates. For detailed operational parameters, including exact molar ratios, stirring speeds, and crystallization protocols, please refer to the standardized synthesis guide below which outlines the critical process parameters (CPPs) identified during the development of this technology.
- Dissolve aminoguanidine carbonate in hydrochloric acid, cool below 10°C, and add sodium nitrite solution dropwise. Adjust pH to alkaline (>9) and reflux to cyclize into 5-aminotetrazole.
- React 5-aminotetrazole with chloroacetic acid under alkaline conditions (using sodium bicarbonate) to perform N-alkylation, yielding 1-acetic acid-5-aminotetrazole with improved regioselectivity.
- Perform reductive deamination on the amino-intermediate using hypophosphorous acid or alcohol reducing agents in acidic media to obtain the final 1-tetrazole acetate product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route offers profound strategic benefits that extend far beyond simple chemical transformation. By eliminating the dependency on sodium azide and hydrazine hydrate, manufacturers can drastically reduce the costs associated with hazardous material handling, specialized storage insurance, and complex waste disposal compliance. The use of readily available and stable raw materials like aminoguanidine carbonate ensures a more resilient supply chain that is less susceptible to the regulatory bottlenecks and transportation restrictions often imposed on explosive precursors. This stability translates directly into more predictable lead times and a lower risk of production stoppages due to raw material shortages or safety audits. Furthermore, the improved regioselectivity achieved in the alkylation step means that less material is lost during purification, effectively increasing the overall mass balance and reducing the cost of goods sold (COGS) per kilogram of finished product. The greener nature of the process also aligns with the increasingly strict environmental, social, and governance (ESG) mandates of multinational pharmaceutical companies, making suppliers who adopt this technology more attractive partners for long-term contracts.
- Cost Reduction in Manufacturing: The elimination of expensive and dangerous reagents like sodium azide removes the need for costly safety infrastructure and specialized waste treatment protocols, leading to substantial operational savings. Additionally, the higher selectivity of the alkylation step reduces the consumption of solvents and energy required for chromatographic purification or repeated recrystallization, further driving down production costs. The ability to use standard stainless steel reactors without the need for exotic lining materials required for highly corrosive or explosive mixtures also lowers capital depreciation costs over time.
- Enhanced Supply Chain Reliability: Sourcing aminoguanidine carbonate and sodium nitrite is significantly more straightforward than procuring regulated explosives, ensuring a continuous flow of materials even during periods of tight regulatory scrutiny. The reduced hazard profile simplifies logistics, allowing for broader transportation options and faster customs clearance, which is critical for maintaining just-in-time inventory levels for downstream API manufacturers. This reliability minimizes the risk of stockouts and enables more accurate forecasting for global demand fluctuations in the cephalosporin market.
- Scalability and Environmental Compliance: The aqueous-based nature of the initial steps and the use of benign reducing agents make this process highly scalable from pilot plant to multi-ton commercial production without significant re-engineering. The reduction in toxic waste generation simplifies effluent treatment, ensuring compliance with increasingly stringent environmental regulations in major manufacturing hubs like China and India. This environmental compatibility future-proofs the supply chain against potential regulatory crackdowns on polluting chemical processes, securing long-term operational continuity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route, derived directly from the experimental data and beneficial effects described in the patent literature. These insights are intended to clarify the practical implications of switching from traditional azide-based methods to this safer, amine-based protocol for both technical teams and business stakeholders. Understanding these details is crucial for evaluating the feasibility of technology transfer and the potential impact on existing manufacturing workflows.
Q: Why is the aminoguanidine carbonate route safer than traditional sodium azide methods?
A: Traditional methods rely on sodium azide, which is highly explosive and toxic, requiring stringent safety protocols. The patented route uses aminoguanidine carbonate and sodium nitrite, significantly reducing explosion risks and toxic waste generation while maintaining high yields.
Q: How does this process improve regioselectivity during N-alkylation?
A: By utilizing the electron-donating effect of the amino substituent on the tetrazole ring, the pKa value of the nitrogen atoms is altered. This strategic modification increases the selectivity for the 1-position substitution from approximately 50% to about 75%, simplifying downstream purification.
Q: What are the typical yields for the key intermediates in this synthesis?
A: The process demonstrates robust efficiency, with the initial cyclization step achieving yields up to 97%. The subsequent alkylation and deamination steps maintain competitive yields of approximately 74% and 78% respectively, ensuring overall process viability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-Tetrazole Acetate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to safer, more efficient synthetic routes is not just a technical upgrade but a strategic imperative for the modern pharmaceutical supply chain. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising results seen in patent CN111393382A can be seamlessly translated into robust industrial reality. We are committed to delivering high-purity 1-tetrazole acetate that meets stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify every batch against the highest international standards. Our facility is designed to handle complex heterocyclic chemistry with the utmost safety and precision, guaranteeing a supply of intermediates that empower your antibiotic production lines without interruption.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis method can be integrated into your specific supply requirements. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits tailored to your volume needs. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that enhance both the safety and profitability of your cephalosporin manufacturing operations.
