Advanced Carbonylation Strategy for High-Purity 4,4-Dimethylisoxazol-3-one Manufacturing
The global demand for high-efficiency herbicides continues to drive innovation in intermediate synthesis, particularly for key scaffolds like 4,4-dimethylisoxazol-3-one, a critical precursor for the widely used herbicide Clomazone. A significant technological breakthrough in this domain is detailed in patent CN112174906B, which outlines a novel preparation method utilizing 3-chloro-2-methylpropene and carbon monoxide as primary feedstocks. This patent represents a paradigm shift from traditional acyl chloride-based routes to a more atom-economical carbonylation strategy. By leveraging high-pressure carbonylation followed by precise hydroxylamination and cyclization, this method addresses long-standing industry pain points regarding impurity profiles and purification complexity. For R&D directors and procurement specialists seeking a reliable agrochemical intermediate supplier, understanding the mechanistic advantages of this route is essential for securing a stable, high-quality supply chain for next-generation crop protection agents.
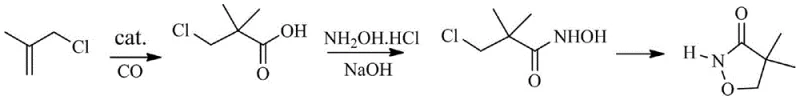
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of 4,4-dimethylisoxazol-3-one has relied heavily on the chloropivaloyl chloride method, which involves the reaction of chloropivaloyl chloride with hydroxylamine hydrochloride. While this traditional pathway offers mild reaction conditions and utilizes water as a solvent, it suffers from inherent chemical inefficiencies that impact final product quality. The preparation of the starting material, chloropivaloyl chloride, typically requires thionyl chloride, a reagent known for generating substantial amounts of acidic waste and sulfur-containing by-products that are environmentally burdensome. More critically, the subsequent ring-closing step in the conventional process is plagued by competitive side reactions that generate difficult-to-remove impurities. These impurities not only lower the overall yield, often capping around 93%, but also necessitate rigorous and costly purification steps to meet the stringent purity specifications required for high-performance herbicide formulations. Consequently, manufacturers face challenges in maintaining consistent batch-to-batch quality while managing escalating waste disposal costs.
The Novel Approach
In stark contrast, the innovative methodology disclosed in the patent introduces a streamlined three-step sequence that fundamentally alters the synthetic logic. The process initiates with the direct carbonylation of 3-chloro-2-methylpropene using carbon monoxide in the presence of specific acid catalysts, such as boron trifluoride hydrates or sulfuric acid hydrates. This step efficiently constructs the carbon skeleton and introduces the carboxylic acid functionality in a single operation, bypassing the need for pre-formed acyl chlorides entirely. As illustrated in the reaction scheme, this is followed by a controlled hydroxylamination to form the hydroxamic acid intermediate, and finally, a base-mediated cyclization. This novel approach not only eliminates the use of thionyl chloride, aligning with green chemistry principles, but also significantly simplifies the post-reaction workup. The result is a process characterized by high conversion rates, minimal by-product formation, and a final product purity that consistently exceeds 99%, offering a compelling value proposition for cost reduction in herbicide manufacturing.
Mechanistic Insights into Acid-Catalyzed Carbonylation and Cyclization
The cornerstone of this advanced synthesis is the initial carbonylation step, where 3-chloro-2-methylpropene reacts with carbon monoxide under high pressure (10-100 MPa) and elevated temperature (120-150°C). The choice of catalyst is pivotal; the patent specifies the use of hydrated Lewis or Brønsted acids like BF3·(H2O)2 or H2SO4·(H2O)x. These catalysts facilitate the electrophilic addition of the protonated alkene to carbon monoxide, generating a stable acylium ion intermediate that is subsequently trapped by water to yield 3-chloropivalic acid. The use of hydrated catalysts is particularly ingenious as it provides the necessary proton source while moderating the acidity to prevent polymerization of the alkene substrate. This precise control over the reaction environment ensures high selectivity for the linear acid product over potential branched isomers or oligomers, establishing a clean foundation for the subsequent transformation steps.
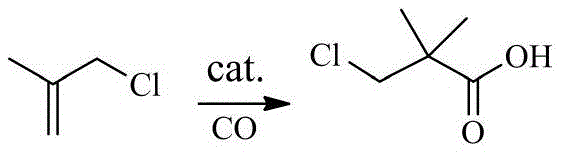
Following the formation of the acid, the process moves to hydroxylamination and cyclization, where pH control becomes the dominant factor in determining product quality. During the hydroxylamination phase, the reaction mixture is maintained at a near-neutral pH of 7.0-7.5. This specific window is critical because it promotes the formation of the hydroxamic acid while preventing the premature hydrolysis of the chloro-group or the acid itself, which would occur in strongly alkaline conditions. The final cyclization step is then executed by carefully raising the pH to 8.5-9.5 at moderate temperatures (35-45°C). This mild alkaline environment facilitates the intramolecular nucleophilic attack of the hydroxamic oxygen on the adjacent carbon bearing the chlorine atom, closing the isoxazole ring. By avoiding the harsh conditions typical of older methods, this mechanism effectively suppresses the formation of ring-opened by-products and polymeric tars, ensuring that the final 4,4-dimethylisoxazol-3-one is obtained with exceptional clarity and purity.
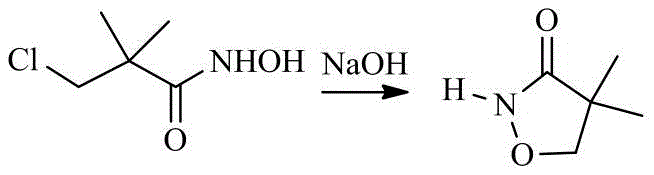
How to Synthesize 4,4-Dimethylisoxazol-3-one Efficiently
Implementing this synthesis route requires careful attention to reaction engineering, particularly regarding pressure management and pH titration. The process begins with the charging of the carbonylation reactor with the chosen acid catalyst and the introduction of carbon monoxide to reach the target pressure before heating. Once the 3-chloropivalic acid is isolated via steam distillation and extraction, it is immediately subjected to the hydroxylamination conditions using hydroxylamine hydrochloride and a phase transfer catalyst system like KI/polyethylene glycol. The transition to the cyclization phase must be gradual, with continuous monitoring of the pH to ensure it remains within the narrow 8.5-9.5 optimal range. Detailed standardized operating procedures are essential to replicate the high yields reported in the patent examples. For a comprehensive breakdown of the specific reagent quantities, temperature ramps, and workup protocols, please refer to the technical guide below.
- Perform acid-catalyzed carbonylation of 3-chloro-2-methylpropene with CO at 120-150°C and 10-100 MPa to form 3-chloropivalic acid.
- Conduct hydroxylamination of the acid using hydroxylamine hydrochloride and base at pH 7.0-7.5 to generate the hydroxamic acid intermediate.
- Execute ring-closure under controlled alkaline conditions (pH 8.5-9.5) at 35-45°C to yield the final isoxazolone product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this carbonylation-based route offers substantial strategic benefits for procurement managers and supply chain heads focused on long-term stability and cost efficiency. The most immediate advantage lies in the raw material profile; by utilizing 3-chloro-2-methylpropene and carbon monoxide, the process relies on abundant, commodity-grade chemicals rather than specialized, expensive acyl chlorides. This shift drastically reduces the exposure to price volatility associated with niche reagents and simplifies sourcing logistics. Furthermore, the elimination of thionyl chloride from the process flow removes a significant environmental liability, thereby reducing the costs associated with hazardous waste treatment and regulatory compliance. The simplified purification process, which avoids complex chromatographic separations or multiple recrystallizations, translates directly into reduced processing time and lower utility consumption per kilogram of product, driving down the overall cost of goods sold.
- Cost Reduction in Manufacturing: The economic benefits of this novel pathway are driven primarily by the simplification of the synthetic sequence and the reduction of waste streams. By removing the need for thionyl chloride and its associated scrubbing systems, manufacturers can achieve significant operational expenditure savings. Additionally, the high selectivity of the carbonylation and cyclization steps minimizes the loss of valuable starting materials to side products, effectively improving the mass balance of the entire process. The ability to isolate the final product with high purity through simple extraction and crystallization further reduces the burden on downstream processing units, allowing for faster batch turnover and increased plant throughput without the need for capital-intensive equipment upgrades.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the use of robust, widely available feedstocks. Unlike complex intermediates that may be sourced from a limited number of specialized vendors, 3-chloro-2-methylpropene and carbon monoxide are produced on a massive industrial scale, ensuring a consistent and reliable supply even during market fluctuations. The process tolerance described in the patent, which allows for variations in catalyst hydration states and pressure ranges, adds another layer of robustness, making the manufacturing process less susceptible to minor deviations in raw material specifications. This reliability is crucial for maintaining uninterrupted production schedules for downstream herbicide formulations, safeguarding against stockouts that could impact agricultural seasons.
- Scalability and Environmental Compliance: The design of this synthesis is inherently scalable, making it ideal for commercial scale-up of complex agrochemical intermediates. The reaction conditions, while requiring high-pressure equipment for the first step, utilize standard unit operations such as distillation and liquid-liquid extraction in subsequent steps, which are easily adaptable to multi-ton reactors. From an environmental standpoint, the process aligns perfectly with modern green chemistry mandates by minimizing halogenated waste and avoiding the generation of sulfur dioxide. This eco-friendly profile not only facilitates easier permitting in regions with strict environmental regulations but also enhances the sustainability credentials of the final herbicide product, a factor increasingly valued by global agrochemical companies and end-users.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for evaluating the technology's feasibility. Understanding these details is vital for technical teams assessing the risk and reward profile of adopting this new manufacturing method. The answers highlight the specific operational parameters and quality outcomes that differentiate this process from legacy technologies.
Q: How does this carbonylation route improve purity compared to the chloropivaloyl chloride method?
A: The novel route bypasses the use of thionyl chloride and harsh acylation conditions, significantly reducing halogenated by-products. Furthermore, the controlled pH cyclization minimizes side reactions, resulting in assay values exceeding 99% without complex refining.
Q: What are the critical safety parameters for the carbonylation step?
A: The reaction operates at high pressures (10-100 MPa) and elevated temperatures (120-150°C). Strict control of CO feed rates and the use of specialized high-pressure reactors are mandatory to ensure operational safety and prevent runaway exotherms.
Q: Is this process scalable for industrial clomazone production?
A: Yes, the process utilizes commodity raw materials like carbon monoxide and simple alkenes. The elimination of difficult-to-remove impurities simplifies downstream processing, making it highly suitable for multi-ton commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4,4-Dimethylisoxazol-3-one Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the efficacy and safety of modern crop protection solutions. Our technical team has extensively analyzed the carbonylation route described in CN112174906B and possesses the expertise to translate this laboratory-scale innovation into robust commercial reality. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot plant to full-scale manufacturing is seamless and efficient. Our facilities are equipped with state-of-the-art high-pressure reactors and rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 4,4-dimethylisoxazol-3-one delivered meets the exacting standards required for premium herbicide synthesis.
We invite you to collaborate with us to leverage this advanced technology for your supply chain. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical needs. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us demonstrate how our commitment to process innovation and quality assurance can drive value and reliability in your agrochemical manufacturing operations.
