Advanced Manufacturing of 4,4'-Dihydroxy-diazobenzophenone for High-Performance Optical Materials
The landscape of functional organic materials is constantly evolving, driven by the demand for compounds with precise photoisomerization characteristics. Patent CN113461564A introduces a significant advancement in this field by disclosing a robust preparation method for 4,4'-dihydroxy-diazobenzophenone, a sophisticated bisazo compound. This molecule is not merely a chemical intermediate; it represents a critical building block for next-generation optical information storage systems, photochromic devices, and nano-engines. The patent outlines a streamlined synthetic route that leverages classical diazotization chemistry but optimizes it for modern manufacturing standards, focusing on aqueous systems and accessible raw materials. For R&D directors and procurement specialists alike, understanding the nuances of this synthesis is vital for securing a reliable supply chain of high-performance optical materials.
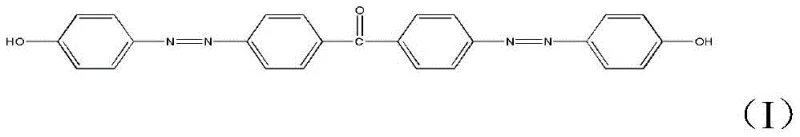
At the heart of this innovation lies the unique structural architecture of the target molecule, which combines the rigidity of a benzophenone core with the dynamic photo-responsive properties of dual azo linkages. The presence of terminal hydroxyl groups further enhances its utility, allowing for potential derivatization or integration into polymer matrices. As industries pivot towards smarter materials capable of responding to light stimuli, the ability to produce such complex bisazo structures efficiently becomes a strategic asset. The methodology described in the patent moves away from exotic reagents, favoring a pathway that is both chemically elegant and commercially viable, setting a new benchmark for the synthesis of disazo-containing materials.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of complex bisazo compounds has been fraught with challenges that hinder large-scale adoption. Conventional routes often rely on organic solvents that are expensive, toxic, and difficult to recover, creating significant environmental burdens and safety hazards in a production facility. Furthermore, older methodologies frequently suffer from poor regioselectivity, leading to mixtures of ortho- and para-substituted isomers that require energy-intensive purification steps to separate. The instability of diazonium intermediates in non-aqueous or poorly controlled thermal environments often results in premature decomposition, drastically reducing overall yields and generating hazardous nitrogen oxide byproducts. These factors collectively inflate the cost of goods sold and introduce volatility into the supply chain, making it difficult for manufacturers to guarantee consistent quality and delivery timelines for high-value optical intermediates.
The Novel Approach
In stark contrast, the approach detailed in CN113461564A utilizes a predominantly aqueous system that mitigates many of these historical pain points. By employing water as the primary solvent medium for the formation of the amine salt and the subsequent diazotization, the process inherently improves thermal management, which is critical for controlling exothermic reactions. The strategy involves a sequential addition protocol where the diazonium salt is generated in situ at low temperatures and immediately coupled with a phenolate solution, minimizing the residence time of the unstable intermediate. This "generate-and-consume" tactic significantly suppresses side reactions and decomposition pathways. Moreover, the final purification via ethanol recrystallization offers a green alternative to chromatographic separations, aligning with modern sustainability goals while ensuring the high purity required for electronic and optical applications.
Mechanistic Insights into Diazotization and Electrophilic Azo Coupling
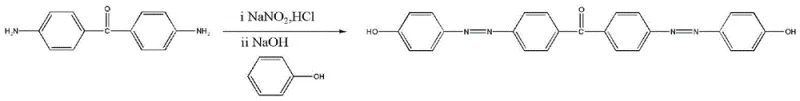
The core of this synthesis relies on the precise manipulation of reaction kinetics during the diazotization phase. The process begins with the protonation of 4,4'-diaminobenzophenone using hydrochloric acid to form a soluble amine salt. This step is crucial as it activates the amine groups for nucleophilic attack by nitrous acid, which is generated in situ from sodium nitrite. The maintenance of the reaction temperature between 0-5°C is not merely a suggestion but a thermodynamic necessity; at higher temperatures, the aryl diazonium ion becomes unstable and decomposes into phenols and nitrogen gas, destroying the synthetic pathway. The patent emphasizes a controlled dropwise addition of sodium nitrite over 10-15 minutes, ensuring that the concentration of nitrous acid never exceeds the consumption rate, thereby preventing the accumulation of excess oxidant which could degrade the sensitive aromatic rings.
Following the formation of the bis-diazonium salt, the mechanism shifts to an electrophilic aromatic substitution known as azo coupling. Here, the phenol is first converted into a phenolate anion using sodium hydroxide. This deprotonation dramatically increases the electron density of the aromatic ring, specifically activating the para-position relative to the hydroxyl group. The electron-rich phenolate acts as a potent nucleophile, attacking the electrophilic terminal nitrogen of the diazonium species. The patent specifies adjusting the pH to neutral during this phase, a delicate balance that ensures the phenol remains activated as a phenolate while preventing the decomposition of the diazonium salt which occurs in highly alkaline conditions. This mechanistic precision ensures that the coupling occurs exclusively at the desired positions, yielding the symmetric 4,4'-dihydroxy-diazobenzophenone with minimal impurity formation.
How to Synthesize 4,4'-Dihydroxy-diazobenzophenone Efficiently
Executing this synthesis requires strict adherence to the thermal and stoichiometric parameters outlined in the patent to ensure reproducibility and safety. The process is designed to be scalable, moving from laboratory beakers to industrial reactors without fundamental changes to the chemistry. Operators must prioritize temperature control equipment capable of maintaining the cryogenic 0-5°C window throughout the diazotization and coupling stages. The following guide summarizes the critical operational phases derived from the patent data, serving as a foundational reference for process engineers looking to implement this route.
- Prepare amine salt solution by dissolving 4,4'-diaminobenzophenone in water and adding hydrochloric acid at room temperature.
- Generate diazonium salt by adding sodium nitrite solution at 0-5°C under strict temperature control.
- Perform azo coupling by adding the diazonium salt to a phenolate solution, adjusting pH to neutral, and recrystallizing from ethanol.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the shift towards the methodology described in CN113461564A offers tangible strategic benefits beyond mere chemical yield. The reliance on commodity chemicals such as hydrochloric acid, sodium hydroxide, sodium nitrite, and phenol means that the raw material supply chain is robust and less susceptible to the geopolitical fluctuations that often plague specialty reagent markets. By utilizing water as the primary reaction medium, the process significantly reduces the volume of volatile organic compounds (VOCs) emitted, simplifying regulatory compliance and lowering the costs associated with solvent recovery and waste disposal. This transition to a greener, aqueous-based workflow directly translates to a more sustainable manufacturing profile, which is increasingly becoming a prerequisite for partnerships with major multinational corporations.
- Cost Reduction in Manufacturing: The elimination of expensive organic solvents during the reaction phase drastically lowers the raw material expenditure per kilogram of product. Furthermore, the simplified workup procedure, which avoids complex chromatographic purification in favor of straightforward filtration and recrystallization, reduces labor hours and energy consumption. The use of ethanol for recrystallization is also economically favorable as it is a widely available, recyclable solvent with a lower toxicity profile compared to alternatives like dichloromethane or DMF. These cumulative efficiencies result in a substantially lower cost base, allowing for more competitive pricing in the global market for optical intermediates.
- Enhanced Supply Chain Reliability: Since all key reagents are bulk industrial chemicals, the risk of supply disruption is minimized. Unlike specialized catalysts or ligands that may have single-source suppliers, hydrochloric acid and phenol are produced globally at massive scales. This abundance ensures that production schedules can be maintained consistently, reducing lead times for high-purity fine chemical intermediates. Additionally, the stability of the final product and the simplicity of its storage requirements facilitate easier logistics and inventory management, ensuring that stock can be held safely without degradation over extended periods.
- Scalability and Environmental Compliance: The aqueous nature of the reaction mixture makes heat transfer more efficient on a large scale, mitigating the risks of thermal runaway that are common in exothermic diazotizations performed in organic solvents. This inherent safety feature allows for the commercial scale-up of complex bisazo compounds with greater confidence and reduced engineering controls. From an environmental standpoint, the wastewater generated is primarily saline and can be treated using standard neutralization and filtration protocols, avoiding the need for specialized hazardous waste incineration. This alignment with green chemistry principles future-proofs the manufacturing process against tightening environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 4,4'-dihydroxy-diazobenzophenone. These insights are derived directly from the experimental data and claims within the patent documentation, providing clarity for technical teams evaluating this synthesis route for potential adoption or outsourcing.
Q: What are the critical temperature controls for this synthesis?
A: The patent specifies maintaining temperatures between 0-5°C during both the diazotization of the amine salt and the subsequent coupling with phenol to prevent decomposition of the unstable diazonium intermediate.
Q: How is the final product purified?
A: The crude solid is purified via recrystallization using ethanol with a specific solid-to-liquid ratio of 1g to 70ml, ensuring high purity suitable for optical applications.
Q: What is the theoretical yield potential?
A: While specific examples in the patent demonstrated yields around 40%, the disclosure indicates that optimization of the process parameters can potentially achieve yields reaching up to 60%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4,4'-Dihydroxy-diazobenzophenone Supplier
As the demand for advanced photochromic and optical storage materials accelerates, having a manufacturing partner with deep expertise in azo chemistry is essential. NINGBO INNO PHARMCHEM stands at the forefront of this sector, combining rigorous scientific inquiry with industrial pragmatism. Our facilities are equipped to handle the precise temperature controls and safety protocols required for diazotization reactions, ensuring that every batch meets stringent purity specifications. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, leveraging our rigorous QC labs to verify structural integrity through NMR, IR, and UV-Vis spectroscopy, just as validated in the patent literature.
We invite R&D directors and procurement leaders to collaborate with us to optimize this synthesis for your specific application needs. Whether you require custom particle sizes for polymer dispersion or specific purity grades for electronic applications, our technical team is ready to assist. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can enhance your supply chain resilience and product performance.
