Advanced Manufacturing of 2,6-Diethyl-4-Methyl Phenylacetate via Boronic Acid Coupling
The global demand for high-performance herbicides, particularly phenylpyrazoline compounds like pinoxaden, has necessitated the development of robust and scalable synthetic routes for their key intermediates. Patent CN113121341A introduces a groundbreaking methodology for synthesizing 2,6-diethyl-4-methyl phenylacetate, a critical building block in modern agrochemistry. This innovation addresses long-standing industrial pain points by replacing hazardous reagents and expensive catalysts with a streamlined, two-step process centered on boronic acid chemistry. For R&D directors and procurement specialists alike, this represents a significant shift towards safer, more cost-efficient manufacturing paradigms. The technology leverages the unique reactivity of 2,6-diethyl-4-methylphenylboronic acid, coupling it directly with glycine esters under mild conditions to achieve high yields without the baggage of complex purification protocols associated with legacy methods.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
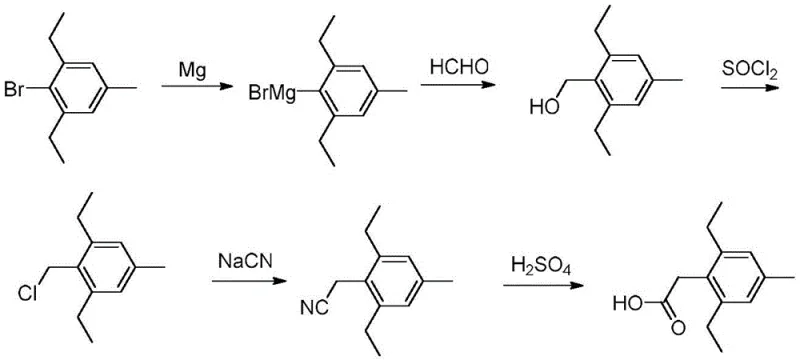
Historically, the synthesis of 2,6-diethyl-4-methyl phenylacetate has been plagued by severe safety and economic inefficiencies. Traditional routes often rely on Grignard reactions followed by cyanidation, a process that mandates the use of highly toxic sodium cyanide reagents, posing extreme risks to personnel and requiring costly waste management infrastructure. Furthermore, alternative pathways utilizing palladium catalysis, such as those described in DE102004053191A1, incur prohibitive costs due to the necessity of noble metals and specialized phosphine ligands. These methods also frequently suffer from strict anhydrous and anaerobic requirements, complicating equipment design and limiting scalability. The presence of chloro-substituted impurities in Meerwein arylation routes further degrades product quality, necessitating energy-intensive purification steps that erode profit margins and extend production lead times.
The Novel Approach
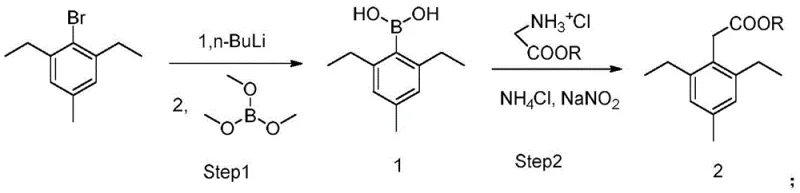
In stark contrast, the methodology disclosed in CN113121341A offers a transformative solution by utilizing a boronic acid intermediate coupled with glycine alkyl ester hydrochloride. This approach completely circumvents the need for toxic cyanides and expensive palladium catalysts, fundamentally altering the cost structure of production. The reaction proceeds in a biphasic toluene-water system at elevated temperatures, demonstrating remarkable tolerance to moisture compared to Grignard alternatives. By streamlining the synthesis into fewer steps and employing readily available reagents like n-butyllithium and trimethyl borane, the process achieves superior atom economy. This not only simplifies the operational workflow but also ensures a cleaner impurity profile, making it an ideal candidate for large-scale industrial adoption where safety and consistency are paramount.
Mechanistic Insights into Boronic Acid Coupling Strategy
The core of this technological advancement lies in the precise generation and utilization of the 2,6-diethyl-4-methylphenylboronic acid intermediate. The process initiates with the lithiation of 2,6-diethyl-4-methyl bromobenzene using n-butyllithium at cryogenic temperatures, typically around -65°C, to ensure regioselectivity and prevent side reactions. Subsequent quenching with trimethyl borane efficiently installs the boron functionality, creating a versatile coupling partner. This boronic acid species is then subjected to a unique oxidative coupling environment involving glycine ester hydrochloride, ammonium chloride, and sodium nitrite. The mechanistic pathway likely involves the in situ generation of a diazonium or nitroso species that facilitates the carbon-carbon bond formation between the aryl ring and the glycine moiety, bypassing the need for transition metal mediation entirely.
From an impurity control perspective, this mechanism offers distinct advantages over chloromethylation or Meerwein arylation strategies. By avoiding the introduction of chlorine atoms during the chain-extension phase, the process eliminates the formation of persistent chloro-substituted byproducts that are notoriously difficult to separate via standard distillation or crystallization. The use of a biphasic solvent system further aids in partitioning inorganic salts and polar impurities away from the organic product phase. This inherent selectivity reduces the burden on downstream purification units, allowing for the direct isolation of high-purity 2,6-diethyl-4-methyl phenylacetate. Such mechanistic elegance ensures that the final product meets the stringent specifications required for pharmaceutical and agrochemical applications without excessive processing.
How to Synthesize 2,6-Diethyl-4-Methyl Phenylacetate Efficiently
The practical implementation of this synthesis requires careful attention to stoichiometry and thermal management to maximize yield and safety. The initial lithiation step must be conducted under inert atmosphere to prevent premature quenching of the organolithium species, while the subsequent coupling reaction benefits from optimized molar ratios of the nitrite and ammonium salts to drive the conversion to completion. Detailed operational parameters, including specific temperature ramps and workup procedures, are critical for reproducing the high mass yields reported in the patent examples. For process chemists looking to implement this route, understanding the interplay between the aqueous and organic phases during the coupling step is essential for efficient product recovery.
- React 2,6-diethyl-4-methyl bromobenzene with n-butyllithium at -65°C, followed by trimethyl borane to form the boronic acid intermediate.
- Couple the resulting boronic acid with glycine alkyl ester hydrochloride using ammonium chloride and sodium nitrite in a toluene-water system at 120°C.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this novel synthetic route translates into tangible strategic benefits that extend beyond simple unit cost metrics. By eliminating the dependency on volatile and regulated toxic reagents like sodium cyanide, companies can significantly reduce their environmental compliance burdens and insurance liabilities. The removal of expensive palladium catalysts from the bill of materials directly lowers the variable cost of goods sold, enhancing margin resilience in fluctuating markets. Furthermore, the simplified process flow, characterized by fewer unit operations and milder reaction conditions, allows for faster batch turnover times and reduced equipment downtime. This operational agility enables manufacturers to respond more swiftly to market demand spikes, ensuring a reliable supply of critical agrochemical intermediates to downstream formulators.
- Cost Reduction in Manufacturing: The elimination of noble metal catalysts and toxic cyanide reagents results in substantial raw material savings and reduced waste disposal costs. Without the need for expensive ligand systems or specialized heavy metal scavenging processes, the overall production expenditure is drastically lowered. Additionally, the higher atom efficiency of the boronic acid coupling minimizes solvent usage and energy consumption per kilogram of product, contributing to a leaner and more profitable manufacturing model that withstands competitive pricing pressures.
- Enhanced Supply Chain Reliability: Sourcing stable and non-hazardous raw materials such as glycine esters and boron reagents mitigates the risk of supply disruptions often associated with controlled substances. The robustness of the reaction conditions reduces the likelihood of batch failures due to minor deviations in moisture or oxygen levels, ensuring consistent output quality. This reliability fosters stronger relationships with key customers who depend on uninterrupted delivery schedules for their own production lines, thereby strengthening the supplier's position as a preferred partner in the global value chain.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, utilizing common solvents like toluene and water which are easily managed in large reactors. The absence of highly toxic intermediates simplifies the permitting process for new manufacturing facilities and reduces the complexity of effluent treatment plants. This alignment with green chemistry principles not only future-proofs the operation against tightening environmental regulations but also enhances the corporate sustainability profile, appealing to eco-conscious stakeholders and investors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced synthesis technology. Understanding these details is crucial for stakeholders evaluating the feasibility of integrating this route into their existing production portfolios. The answers are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering a clear view of the operational realities and benefits.
Q: How does this new method improve safety compared to traditional cyanide routes?
A: The novel process completely eliminates the use of highly toxic sodium cyanide reagents required in conventional Grignard-cyanidation pathways, significantly reducing operational hazards and waste treatment costs.
Q: Does this synthesis require expensive noble metal catalysts?
A: No, unlike previous palladium-catalyzed methods disclosed in prior art, this route operates without expensive noble metals or phosphine ligands, drastically lowering raw material costs.
Q: What are the purity advantages of this boronic acid route?
A: By avoiding Meerwein arylation and chlorination steps, the process prevents the formation of difficult-to-remove chloro-substituted impurities, ensuring higher product quality for downstream applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,6-Diethyl-4-Methyl Phenylacetate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this boronic acid-based synthesis for the agrochemical industry. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are seamlessly translated into robust industrial processes. Our state-of-the-art facilities are equipped to handle the specific thermal and safety requirements of organolithium chemistry, while our rigorous QC labs guarantee that every batch meets stringent purity specifications required for high-value herbicide intermediates. We are committed to delivering consistent quality and supply continuity for our global clientele.
We invite you to collaborate with us to leverage this cost-effective and environmentally friendly technology for your supply chain needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this route can optimize your bottom line. Please contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments, and let us help you secure a competitive advantage in the market for 2,6-diethyl-4-methyl phenylacetate.
