Advanced Grignard Methodology for Scalable Sitagliptin Intermediate Production and Commercial Supply
Advanced Grignard Methodology for Scalable Sitagliptin Intermediate Production and Commercial Supply
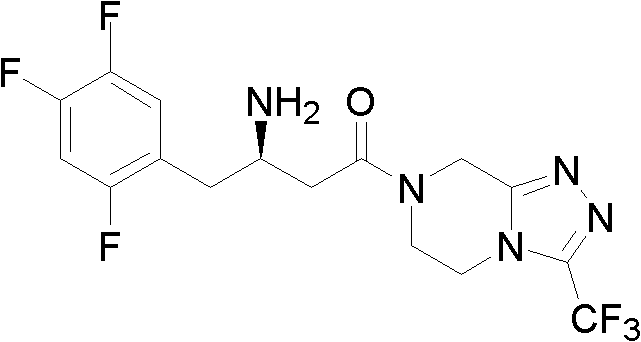
The pharmaceutical industry continuously seeks robust and scalable synthetic routes for high-value active pharmaceutical ingredients (APIs), particularly for blockbuster drugs like Sitagliptin, a premier dipeptidyl peptidase-IV (DPP-IV) inhibitor used in the treatment of type II diabetes. Patent CN102503829A discloses a groundbreaking preparation method for critical Sitagliptin intermediates, specifically 3-carbonyl-4-(2,4,5-trifluorophenyl)-butyrate (Intermediate I) and 1-(3-trifluoromethyl-5,6-dihydro-8H-[1,2,4]triazolo[4,3-a]pyrazine-7-)-4-(2,4,5-trifluorophenyl)-1,3-butanedione (Intermediate II). This technology represents a significant leap forward in process chemistry, utilizing a direct Grignard addition strategy that bypasses the cumbersome multi-step sequences of traditional methods. By leveraging the nucleophilic addition of organomagnesium reagents to nitriles, this patent offers a streamlined pathway that enhances both atomic economy and operational simplicity. For global procurement teams and R&D directors, understanding the nuances of this synthetic approach is vital for securing a reliable sitagliptin intermediate supplier capable of meeting stringent quality and volume demands.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
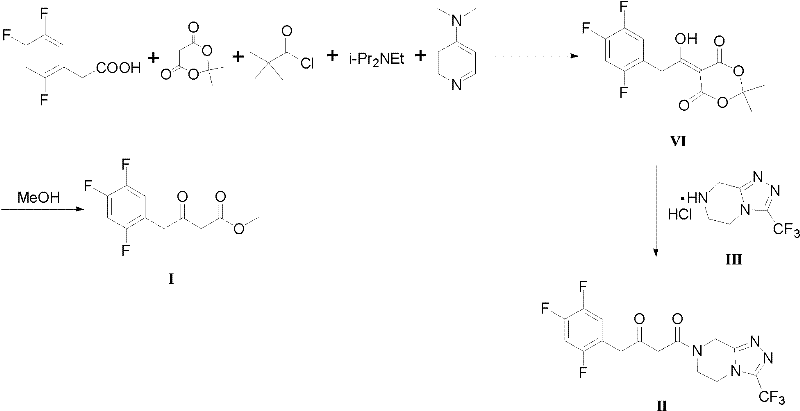
Prior art methodologies, such as those detailed in WO2004087650 and related literature, have historically relied on the use of Meldrum's acid and pivaloyl chloride to construct the beta-keto ester framework essential for Sitagliptin synthesis. While these routes can achieve acceptable yields, they suffer from significant operational drawbacks that hinder efficient large-scale manufacturing. The reliance on Meldrum's acid introduces additional synthetic steps, including condensation and subsequent alcoholysis, which not only increases the consumption of solvents and reagents but also generates substantial waste streams. Furthermore, the use of pivaloyl chloride and specialized bases like N,N-diisopropylethylamine adds to the raw material cost profile and complicates the purification process due to the formation of difficult-to-remove byproducts. These factors collectively result in a prolonged production cycle and elevated manufacturing costs, creating bottlenecks for supply chain managers aiming for cost reduction in API manufacturing.
The Novel Approach
In stark contrast, the novel approach outlined in CN102503829A revolutionizes the synthesis by employing a direct Grignard reaction between bromoacetate derivatives and 2,4,5-trifluorobenzeneacetonitrile. This strategy effectively collapses multiple synthetic steps into a single, highly efficient transformation. By generating the Grignard reagent in situ from readily available magnesium powder and bromoacetic esters, the process eliminates the need for expensive and hazardous activating agents like pivaloyl chloride. The subsequent addition to the nitrile group proceeds under mild thermal conditions, typically ranging from 0°C to 30°C, which minimizes thermal degradation and side reactions. This streamlined workflow not only drastically simplifies the operational procedure but also significantly improves the overall throughput of the manufacturing line. For a reliable agrochemical intermediate supplier or pharma partner, adopting this route translates to a more agile and responsive production capability.
Mechanistic Insights into Grignard Addition to Nitriles
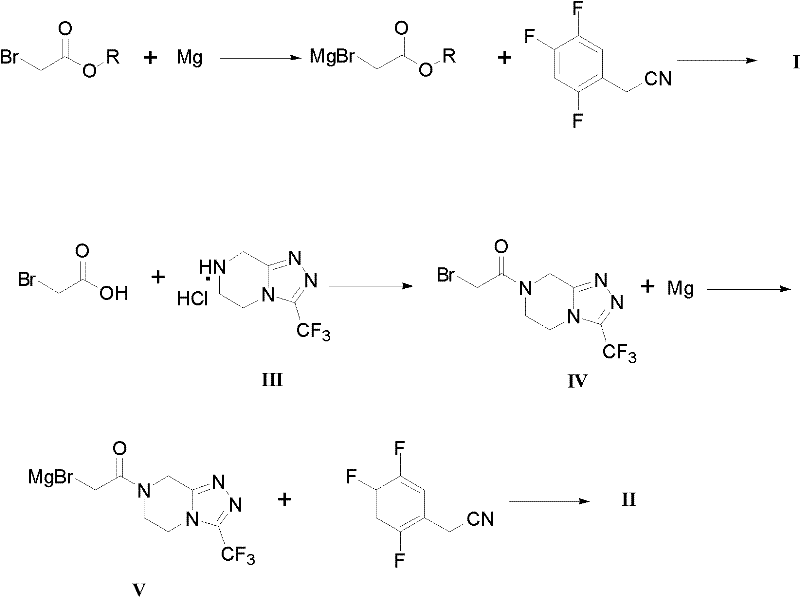
The core of this technological advancement lies in the precise control of the Grignard reagent formation and its subsequent nucleophilic attack on the nitrile functionality. The mechanism initiates with the oxidative insertion of magnesium into the carbon-bromine bond of the bromoacetate precursor, forming a highly reactive organomagnesium species. This step is critical and requires careful activation of the magnesium surface, often facilitated by iodine initiators or mechanical stirring in solvents like THF or diethyl ether. Once formed, the Grignard reagent acts as a potent nucleophile, attacking the electrophilic carbon of the 2,4,5-trifluorobenzeneacetonitrile. The resulting imine salt intermediate is then hydrolyzed under acidic conditions to yield the desired ketone product. The beauty of this mechanism is its tolerance for the electron-deficient aromatic ring, which remains intact throughout the process, ensuring the structural integrity of the final API scaffold.
From an impurity control perspective, the mild reaction conditions specified in the patent play a pivotal role in ensuring high-purity sitagliptin intermediate output. Traditional high-temperature condensations often lead to polymerization or decomposition of sensitive functional groups, generating complex impurity profiles that are challenging to purge. However, by maintaining the reaction temperature strictly between 0°C and 40°C during the Grignard formation and addition phases, the kinetic energy of the system is managed to favor the desired pathway over competing side reactions. Furthermore, the use of common organic solvents allows for straightforward work-up procedures involving aqueous acid quenching and extraction, which effectively removes magnesium salts and unreacted starting materials. This inherent cleanliness of the reaction profile reduces the burden on downstream purification units, thereby enhancing the overall yield and reducing the environmental footprint of the synthesis.
How to Synthesize Sitagliptin Intermediate Efficiently
The synthesis of these valuable intermediates involves a sequence of well-defined chemical transformations that prioritize safety and efficiency. The process begins with the preparation of the Grignard reagent, followed by its controlled addition to the nitrile substrate, and concludes with a standard acidic workup to isolate the product. Detailed operational parameters, such as solvent ratios,滴加 rates, and temperature gradients, are critical for reproducibility and must be strictly adhered to in a GMP environment. The following guide outlines the standardized protocol derived from the patent examples, providing a clear roadmap for technical teams looking to implement this superior methodology.
- Prepare the Grignard reagent by reacting bromoacetate or bromoacetic acid derivatives with magnesium powder in an organic solvent like THF or ether at 0-40°C.
- Perform an acylation reaction between bromoacetic acid and the triazolopyrazine hydrochloride (III) using a condensing agent like thionyl chloride to form intermediate IV.
- React the generated Grignard reagent with 2,4,5-trifluorobenzeneacetonitrile in an organic solvent at 0-30°C, followed by acidic quenching and purification to yield the final intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of the methodology described in CN102503829A offers profound strategic benefits that extend beyond mere chemical elegance. The primary advantage lies in the substantial cost savings achieved through the elimination of high-cost reagents and the reduction of processing time. By replacing expensive condensing agents and complex precursors with commodity chemicals like magnesium and bromoacetic acid, the raw material cost base is significantly lowered. Additionally, the simplified workflow reduces the demand on reactor occupancy time, allowing for higher batch turnover rates and improved asset utilization. These factors combine to create a more resilient and cost-effective supply chain, essential for maintaining competitiveness in the generic pharmaceutical market.
- Cost Reduction in Manufacturing: The novel Grignard-based route eliminates the dependency on costly reagents such as Meldrum's acid and pivaloyl chloride, which are not only expensive but also contribute to higher waste disposal costs. By utilizing magnesium powder and simple bromo-esters, the process leverages abundant and low-cost feedstocks, leading to a drastic reduction in the overall cost of goods sold (COGS). Furthermore, the reduction in synthetic steps means less solvent consumption and lower energy requirements for heating and cooling, compounding the financial benefits. This economic efficiency makes the process highly attractive for large-scale commercial production where margin optimization is critical.
- Enhanced Supply Chain Reliability: The reliance on readily available starting materials ensures a stable and continuous supply of intermediates, mitigating the risks associated with sourcing specialized or scarce reagents. Magnesium and bromoacetic acid are commodity chemicals with robust global supply networks, reducing the likelihood of production delays due to raw material shortages. Moreover, the operational simplicity of the process reduces the technical barriers to entry for manufacturing partners, allowing for easier technology transfer and broader geographic diversification of supply sources. This reliability is paramount for ensuring uninterrupted delivery of high-purity pharmaceutical intermediates to downstream API manufacturers.
- Scalability and Environmental Compliance: The mild reaction conditions and straightforward work-up procedures make this process inherently scalable from pilot plant to multi-ton commercial production. The absence of harsh reaction conditions minimizes safety risks, while the reduced generation of hazardous byproducts aligns with increasingly stringent environmental regulations. The ability to easily manage waste streams and the potential for solvent recovery further enhance the sustainability profile of the manufacturing process. For companies committed to green chemistry principles, this route offers a viable path to reducing the environmental impact of pharmaceutical manufacturing while maintaining high productivity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity for stakeholders evaluating this technology for potential adoption or licensing.
Q: What are the key advantages of the Grignard route over the Meldrum's acid method?
A: The Grignard route described in CN102503829A eliminates the need for complex precursors like Meldrum's acid and pivaloyl chloride, significantly simplifying the operation and reducing raw material costs compared to prior art methods.
Q: What are the optimal reaction conditions for the Grignard addition step?
A: The patent specifies maintaining the reaction temperature between 0°C and 30°C during the addition of the Grignard reagent to the nitrile, typically using THF or ether as the solvent to ensure high yield and purity.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process utilizes readily available raw materials like magnesium powder and bromoacetic acid, and operates under mild conditions (0-40°C), making it highly suitable for commercial scale-up and mass production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sitagliptin Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthetic routes in the modern pharmaceutical landscape. Our team of expert chemists has extensively analyzed the Grignard-based methodology disclosed in CN102503829A and possesses the technical capability to execute this process with precision and consistency. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent supply of high-quality intermediates. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced analytical instrumentation to guarantee stringent purity specifications, meeting the exacting standards required for FDA-regulated markets.
We invite global pharmaceutical partners to collaborate with us to leverage this advanced technology for their Sitagliptin supply chains. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical needs. We encourage interested parties to contact our technical procurement team to request specific COA data and comprehensive route feasibility assessments. Let us help you optimize your supply chain and reduce costs through the implementation of this superior synthetic strategy.
