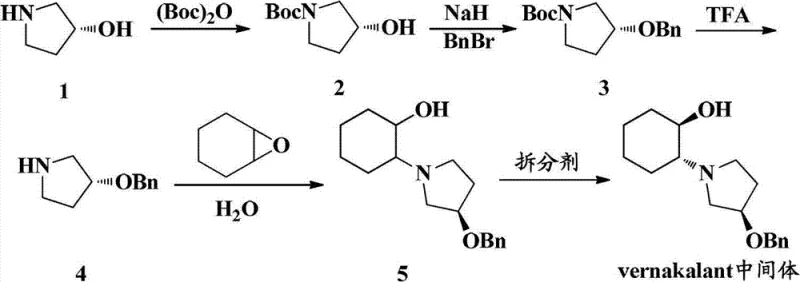
Optimizing Vernakalant Intermediate Production via Advanced Racemization Recovery Technology
The pharmaceutical landscape for anti-arrhythmic medications has seen significant advancements with the introduction of Vernakalant, a selective atrial sodium and potassium channel inhibitor approved in the European Union. As demand for this high-value therapeutic agent grows, the efficiency of its supply chain becomes paramount, particularly regarding the production of its chiral intermediates. Patent CN102702060A introduces a groundbreaking racemization recovery method specifically designed for the by-products found in the split mother liquor of Vernakalant intermediates, addressing a critical bottleneck in chiral synthesis. This technology transforms what was traditionally considered waste into valuable reaction substrates, thereby optimizing the entire manufacturing workflow. By focusing on the conversion of SSR configuration by-products back into usable racemic mixtures, this approach not only enhances yield coefficients but also aligns with modern green chemistry principles. For global procurement teams, understanding this technological shift is essential for securing a reliable Vernakalant intermediate supplier capable of meeting stringent volume requirements without compromising on purity or sustainability standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Vernakalant intermediates involved a chemical resolution step that inevitably generated a substantial amount of mother liquor containing the undesired SSR configuration. Traditional methods for recycling this material often relied on reagents such as methylsulfonyl chloride to induce racemization. However, methylsulfonyl chloride is known to be a strong and stimulating liquid, presenting significant challenges regarding storage, transportation, and operational safety within a manufacturing facility. Furthermore, reactions utilizing this reagent typically require extended periods, often spanning three to four days, which drastically reduces throughput and increases the capital tied up in work-in-progress inventory. The harsh nature of these conditions can also lead to the formation of unwanted impurities, complicating downstream purification and potentially impacting the final drug substance quality. These inefficiencies create a fragile supply chain where cost reduction in pharmaceutical intermediates manufacturing is hindered by safety protocols and slow reaction kinetics.
The Novel Approach
In contrast, the novel approach detailed in the patent utilizes dichloro triaryl phosphine compounds in conjunction with alkaline substances to achieve racemization under much milder conditions. This method operates effectively within a temperature range of 40 to 100 degrees Celsius and completes the reaction within a significantly shorter timeframe of 1 to 5 hours. The use of phosphine-based reagents eliminates the hazards associated with sulfonyl chlorides, thereby simplifying the engineering controls required for the reactor setup. Moreover, this pathway facilitates the recovery of triaryl phosphine oxides, which can be recycled, adding an additional layer of economic and environmental value to the process. By adopting this superior methodology, manufacturers can achieve a more robust and efficient production cycle that supports the commercial scale-up of complex pharmaceutical intermediates while maintaining high safety standards.
Mechanistic Insights into Phosphine-Mediated Racemization
The core of this technological advancement lies in the specific chemical mechanism where the SSR configuration of 2-((R)-3-(aromatic methoxyl)-1-pyrrolidyl)cyclohexanol acts as the reaction substrate. When treated with dichloro triaryl phosphines in an organic solvent, the stereochemical integrity at the reactive center is disrupted, allowing for the re-establishment of equilibrium between the RRR and SSR diastereoisomers. The reaction proceeds through a nucleophilic substitution pathway where the phosphine reagent activates the hydroxyl group, facilitating the inversion of configuration. Following the initial reaction phase, the addition of water triggers hydrolysis, releasing the racemized product which now contains approximately equal amounts of the desired RRR configuration and the SSR configuration. This precise control over stereochemistry ensures that the recovered material is suitable for re-entry into the chiral resolution loop, maximizing the utility of every gram of starting material processed.
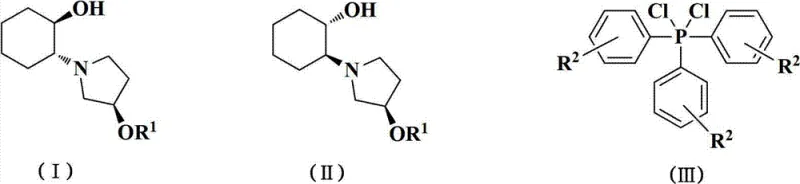
Impurity control is another critical aspect where this mechanism excels, as the mild reaction conditions minimize the degradation of the sensitive pyrrolidine and cyclohexanol moieties. Unlike harsher acidic or basic racemization methods that might cause ring-opening or elimination side reactions, the phosphine-mediated pathway preserves the structural integrity of the intermediate. The subsequent pH adjustment steps, ranging from acidic extraction to alkaline basification, are designed to selectively partition the organic product from inorganic salts and phosphine oxide by-products. This results in a crude racemic product with high HPLC purity, reducing the burden on final crystallization steps. For R&D directors, this level of mechanistic clarity provides confidence in the reproducibility of the process, ensuring that high-purity Vernakalant intermediates can be consistently delivered to meet regulatory specifications.
How to Synthesize Vernakalant Intermediate Efficiently
Implementing this synthesis route requires careful attention to the stoichiometry of the dichloro triaryl phosphine compounds and the selection of appropriate alkaline matter such as triethylamine or imidazole. The process begins with the purification of the mother liquor to isolate the SSR configuration, followed by the racemization reaction in solvents like acetonitrile or toluene. Detailed standard operating procedures regarding temperature ramps, addition rates, and workup protocols are critical for achieving the reported yields and purity levels. The following guide outlines the standardized synthesis steps derived from the patent data to ensure optimal process performance.
- Purify the mother liquor containing SSR configuration 2-((R)-3-(aromatic methoxyl)-1-pyrrolidyl)cyclohexanol obtained from initial chiral resolution.
- React the purified SSR substrate with dichloro triaryl phosphine compounds and alkaline matter in organic solvent at 40 to 100 degrees Celsius for 1 to 5 hours.
- Add water for hydrolysis, adjust pH to separate phases, and recover the racemic product containing both RRR and SSR configurations for re-resolution.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, the adoption of this racemization technology offers profound benefits that extend beyond simple chemical yield improvements. By converting waste mother liquor into valuable feedstock, the overall material efficiency of the plant is drastically enhanced, leading to substantial cost savings on raw material procurement. The elimination of hazardous reagents like methylsulfonyl chloride reduces the regulatory burden and insurance costs associated with handling dangerous goods, further contributing to the economic viability of the project. Additionally, the ability to recycle triaryl phosphine oxides creates a closed-loop system that minimizes waste disposal fees and aligns with corporate sustainability goals. These factors combined create a compelling value proposition for partners seeking a reliable Vernakalant intermediate supplier who prioritizes both cost-efficiency and environmental responsibility.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the significant increase in the overall yield coefficient achieved by recycling the SSR by-product. Instead of discarding nearly half of the material after the initial resolution, this method recovers it for re-processing, effectively doubling the utility of the expensive chiral starting materials. Furthermore, the recovery and potential reuse of the phosphine reagents reduce the recurring cost of consumables, creating a more lean manufacturing model. The milder reaction conditions also imply lower energy consumption for heating and cooling, contributing to reduced utility costs over the lifecycle of the product. These cumulative efficiencies allow for a more competitive pricing structure without sacrificing margin quality.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the availability of specialized or hazardous reagents, but this method utilizes readily available dichloro triaryl phosphines and common bases. The shorter reaction times, reducing the cycle from days to hours, significantly increase the throughput capacity of existing reactor trains, allowing for faster fulfillment of purchase orders. This agility enables the supply chain to respond more rapidly to fluctuations in market demand, reducing lead time for high-purity pharmaceutical intermediates. Moreover, the reduced safety risks associated with the reagents minimize the likelihood of unplanned shutdowns due to safety incidents, ensuring a steady and predictable flow of goods to downstream customers.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is facilitated by the use of standard unit operations such as liquid-liquid extraction and crystallization, which are well-understood in the industry. The generation of triaryl phosphine oxide as a solid by-product simplifies waste management compared to liquid hazardous waste streams, making it easier to comply with increasingly strict environmental regulations. The ability to treat waste mother liquor on-site transforms a liability into an asset, supporting a circular economy model within the manufacturing facility. This robustness ensures that the production of high-purity Vernakalant intermediates can be scaled up to meet global demand while maintaining a minimal environmental footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this racemization technology. These answers are derived directly from the patent specifications and are intended to clarify the operational benefits and chemical feasibility for potential partners. Understanding these details is crucial for evaluating the fit of this technology within your existing supply chain framework.
Q: What are the advantages of using dichloro triaryl phosphines over methylsulfonyl chloride for racemization?
A: Dichloro triaryl phosphines offer milder reaction conditions and eliminate the need for handling hazardous, strong stimulating liquids like methylsulfonyl chloride, thereby improving operational safety and reducing storage difficulties.
Q: Can the by-products from this racemization process be recycled?
A: Yes, the process generates triaryl phosphine oxides as by-products, which can be recovered through recrystallization and potentially converted back into the active phosphine reagents, enhancing the overall atom economy.
Q: How does this method impact the overall yield of Vernakalant intermediates?
A: By converting the wasted SSR configuration from the mother liquor back into a racemic mixture, this method allows for repeated splitting and salification, significantly improving the total yield coefficient compared to traditional discard methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Vernakalant Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent theory to commercial reality requires deep technical expertise and robust infrastructure. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the intricate details of this phosphine-mediated racemization are executed with precision. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of Vernakalant intermediate meets the highest international standards. Our commitment to process excellence means that we can deliver the cost and efficiency benefits described in this report reliably and consistently.
We invite you to engage with our technical procurement team to discuss how this advanced manufacturing route can optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain specific insights into how implementing this technology can impact your bottom line. We encourage you to contact us for specific COA data and route feasibility assessments tailored to your project requirements. Let us partner with you to drive innovation and efficiency in the production of life-saving anti-arrhythmic medications.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
