Scalable Synthesis of High-Efficiency Carbonyl-Based OLED Materials for Next-Generation Display Manufacturing
Scalable Synthesis of High-Efficiency Carbonyl-Based OLED Materials for Next-Generation Display Manufacturing
The rapid evolution of the organic light-emitting diode (OLED) industry demands materials that transcend the limitations of traditional fluorescent and phosphorescent emitters. Patent CN107641117B introduces a groundbreaking series of carbonyl-containing organic electroluminescent materials that successfully integrate Aggregation-Induced Emission (AIE) and Thermally Activated Delayed Fluorescence (TADF) characteristics into a single molecular architecture. This dual-functionality addresses the critical bottleneck of triplet exciton utilization while simultaneously overcoming the efficiency roll-off typically observed in aggregated states. By utilizing a carbonyl group as the central core connected to distinct electron-donating groups, such as substituted phenyl, fluorenyl, carbazolyl, or spirobifluorenyl moieties, these materials achieve a highly twisted molecular conformation. This structural distortion is pivotal as it inhibits the formation of strong intermolecular π–π interactions, thereby preserving high quantum efficiency in the solid state. For R&D directors and procurement specialists seeking reliable OLED material suppliers, this technology represents a paradigm shift towards cost-effective, metal-free, high-performance emissive layers capable of supporting both doped and non-doped device architectures.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional OLED technologies have long struggled with fundamental efficiency and cost barriers that hinder widespread commercial adoption in large-area displays and solid-state lighting. First-generation fluorescent materials are inherently limited by spin statistics, capable of utilizing only 25% of singlet excitons for light emission while wasting 75% of triplet excitons through non-radiative decay pathways. To address this, second-generation phosphorescent materials incorporating heavy transition metals like iridium and platinum were developed to harvest both singlet and triplet excitons. However, these phosphorescent systems suffer from severe drawbacks including the prohibitive cost of precious metals, poor operational stability due to metal-ligand bond degradation, and significant efficiency roll-off at high brightness levels. Furthermore, third-generation pure organic TADF materials, while eliminating the need for heavy metals, often succumb to Aggregation-Caused Quenching (ACQ), where close packing of molecules in the solid film leads to detrimental intermolecular interactions that drastically reduce luminescence efficiency. These cumulative technical challenges necessitate a novel material design strategy that can simultaneously maximize exciton utilization and maintain structural integrity in the aggregated state.
The Novel Approach
The innovative approach detailed in the patent data overcomes these historical limitations by engineering a specific Donor-Acceptor (D-A) molecular topology centered around a carbonyl acceptor unit. By strategically attaching bulky and twisted electron-donating groups to the carbonyl core, the resulting molecules possess a rigid yet distorted three-dimensional geometry. This unique spatial arrangement physically separates the Highest Occupied Molecular Orbital (HOMO) and the Lowest Unoccupied Molecular Orbital (LUMO), which minimizes the exchange energy and results in a negligible singlet-triplet energy gap (ΔEST). This small energy gap facilitates efficient Reverse Intersystem Crossing (RISC), allowing triplet excitons to upconvert to singlet states and emit light, thus achieving theoretical 100% internal quantum efficiency similar to phosphorescent materials but without the metal cost. Crucially, the twisted structure prevents planar stacking, endowing the material with AIE properties where emission is enhanced rather than quenched upon aggregation. This synergy allows for the fabrication of high-efficiency, low roll-off OLED devices that can operate effectively even in simple non-doped configurations, offering a substantial reduction in manufacturing complexity and material costs for electronic chemical manufacturing.
Mechanistic Insights into Friedel-Crafts Acylation and Nucleophilic Substitution
The synthesis of these advanced electroluminescent materials relies on a robust two-step organic transformation sequence that ensures high purity and structural precision. The first critical stage involves a Friedel-Crafts acylation reaction, where a halogen-substituted benzoyl halide, preferably p-fluorobenzoyl chloride or p-bromobenzoyl chloride, acts as the electrophile. In the presence of a strong Lewis acid catalyst such as anhydrous aluminum chloride (AlCl3) within an ultra-dry dichloromethane (DCM) solvent system, the acyl group is selectively introduced onto the aromatic ring of the electron-donating precursor, such as N-phenylcarbazole. This step is meticulously controlled at moderate temperatures, typically around 40°C, to prevent polyacylation or decomposition of the sensitive carbazole moiety. The rigorous exclusion of moisture is paramount here, as water would instantly hydrolyze the acid chloride and deactivate the Lewis acid catalyst, leading to incomplete conversion and difficult-to-remove impurities. The result is a halogen-containing ketone intermediate that serves as the pivotal scaffold for the subsequent coupling reaction.
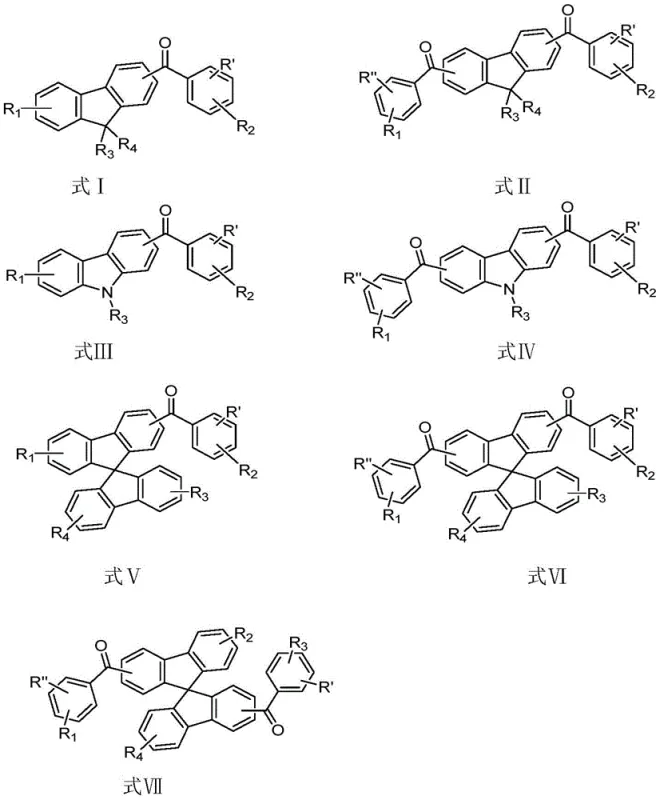
Following the formation of the ketone bridge, the second mechanistic phase employs a nucleophilic aromatic substitution (SnAr) to attach the second electron-donating unit. This step utilizes the halogen atom on the benzoyl ring as a leaving group, which is displaced by a nucleophilic aromatic amine, such as phenoxazine, phenothiazine, or dimethylacridine derivatives. The reaction is driven by a strong base, typically potassium tert-butoxide (t-BuOK), in a polar aprotic solvent like dimethylformamide (DMF) at elevated temperatures near 110°C. The mechanism proceeds through the formation of a Meisenheimer complex, where the electron-withdrawing nature of the carbonyl group activates the aromatic ring towards nucleophilic attack. This specific connectivity creates the desired D-A-D or D-A architecture with the necessary torsional angle between the donor and acceptor units. The choice of substituents, as illustrated in the diverse range of structures from formulas a to r, allows for fine-tuning of the emission color and energy levels, providing a versatile platform for developing full-color display materials with tailored photophysical properties.
How to Synthesize CP-BZ-PXZ Efficiently
The practical implementation of this technology is exemplified by the synthesis of the specific emitter CP-BZ-PXZ, which demonstrates the feasibility of scaling this chemistry for commercial production. The process begins with the precise stoichiometric mixing of p-fluorobenzoyl chloride and N-phenylcarbazole in ultra-dry DCM, followed by the slow addition of AlCl3 to manage the exothermic nature of the complexation. After reacting at 40°C for 3 hours, the mixture is quenched with ice hydrochloric acid and extracted to isolate the intermediate ketone with an impressive yield of 96%, indicating a highly efficient first step with minimal byproduct formation. The subsequent coupling with phenoxazine requires an inert nitrogen atmosphere to prevent oxidation of the sensitive amine species, utilizing t-BuOK in DMF at 110°C for 12 hours to ensure complete conversion. This standardized protocol highlights the reproducibility of the method, making it an attractive candidate for reliable OLED material suppliers looking to establish consistent supply chains. The detailed standardized synthesis steps are outlined below for technical reference.
- Perform Friedel-Crafts acylation by reacting p-fluorobenzoyl chloride with N-phenylcarbazole in ultra-dry DCM using AlCl3 catalyst at 40°C for 3 hours to obtain the halogen-containing intermediate.
- Conduct nucleophilic aromatic substitution by reacting the intermediate with phenoxazine in ultra-dry DMF using t-BuOK base under nitrogen protection at 110°C for 12 hours.
- Purify the final crude product via extraction with dichloromethane and water, followed by concentration and column chromatography to achieve the target electroluminescent material.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this carbonyl-based material platform offers compelling economic and logistical benefits that directly impact the bottom line of display manufacturing operations. The elimination of scarce and volatile transition metals like iridium from the supply chain removes a significant source of cost uncertainty and geopolitical risk. Since the synthesis relies on commodity chemicals such as benzoyl chlorides, carbazoles, and common bases, the raw material sourcing is robust and less susceptible to the market fluctuations that plague precious metal markets. Furthermore, the high yields reported in the patent examples, such as the 96% yield for the intermediate and roughly 80% for the final coupling step, suggest a process with excellent atom economy. This efficiency translates to reduced waste generation and lower disposal costs, aligning with increasingly stringent environmental regulations in chemical manufacturing zones.
- Cost Reduction in Manufacturing: The synthetic route avoids the use of expensive palladium-catalyzed cross-coupling reactions often required for similar conjugated systems, instead utilizing cost-effective Friedel-Crafts and nucleophilic substitution chemistries. By removing the need for costly transition metal catalysts and ligands, the overall bill of materials is significantly reduced. Additionally, the ability to fabricate high-performance non-doped devices simplifies the OLED stack architecture, potentially reducing the number of deposition steps and the quantity of host materials required, leading to substantial cost savings in the final panel assembly process.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, including p-fluorobenzoyl chloride and various carbazole derivatives, are widely available from multiple global chemical vendors, ensuring a diversified and secure supply base. Unlike proprietary phosphorescent dopants that may be locked behind exclusive licensing agreements with limited suppliers, this open chemical architecture allows for competitive sourcing strategies. The moderate reaction conditions (40°C to 110°C) and standard solvents (DCM, DMF) mean that production can be easily transferred to existing multipurpose chemical reactors without requiring specialized high-pressure or cryogenic equipment, thereby enhancing supply continuity and reducing lead time for high-purity organic electroluminescent materials.
- Scalability and Environmental Compliance: The process demonstrates excellent scalability potential, as evidenced by the straightforward workup procedures involving standard aqueous extractions and column chromatography purification. The high purity achievable through these standard methods ensures that the final material meets the stringent specifications required for electronic applications without needing complex sublimation cycles that can lower overall throughput. Moreover, the absence of heavy metals simplifies the wastewater treatment and waste solid disposal protocols, facilitating easier compliance with environmental standards and reducing the regulatory burden associated with hazardous metal waste management in large-scale production facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of these carbonyl-based TADF/AIE materials in industrial settings. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on performance metrics and processing requirements. Understanding these details is crucial for technical teams evaluating the feasibility of integrating these emitters into next-generation display backplanes or lighting panels.
Q: What distinguishes these carbonyl-based materials from conventional phosphorescent OLED emitters?
A: Unlike conventional phosphorescent materials that rely on expensive and unstable transition metals like iridium or platinum, these carbonyl-based materials are purely organic. They uniquely combine Aggregation-Induced Emission (AIE) and Thermally Activated Delayed Fluorescence (TADF) properties, allowing for 100% exciton utilization without the heavy metal cost burden or stability issues associated with phosphorescent dopants.
Q: How does the molecular structure contribute to the high efficiency of these materials?
A: The molecular design features a carbonyl core connecting different electron-donating groups, creating a distorted spatial structure. This distortion effectively suppresses strong π–π interactions in the aggregated state, preventing aggregation-caused quenching (ACQ). Furthermore, the separation of HOMO and LUMO distributions facilitates a small singlet-triplet energy gap (ΔEST), enabling efficient reverse intersystem crossing (RISC) characteristic of TADF materials.
Q: Are these materials suitable for non-doped OLED device architectures?
A: Yes, a significant advantage of this technology is its suitability for non-doped device structures. The AIE characteristics ensure high solid-state luminescence efficiency even without a host matrix, simplifying the device fabrication process and reducing material costs while maintaining low efficiency roll-off and high brightness performance.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable CP-BZ-PXZ Supplier
As the demand for high-efficiency, metal-free OLED materials accelerates, partnering with an experienced CDMO partner is essential for successful technology transfer and commercialization. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our state-of-the-art facilities are equipped to handle the moisture-sensitive and inert atmosphere conditions required for the synthesis of these advanced electroluminescent materials, guaranteeing stringent purity specifications through our rigorous QC labs. We understand that the performance of an OLED device is critically dependent on the purity of the emissive layer, and our dedicated analytical teams employ advanced chromatographic and spectroscopic techniques to verify the absence of trace impurities that could act as quenching sites.
We invite R&D directors and procurement leaders to collaborate with us to optimize this synthetic route for your specific application requirements. Our technical team is prepared to provide a Customized Cost-Saving Analysis that evaluates the economic viability of switching to this carbonyl-based platform for your product line. Please contact our technical procurement team today to request specific COA data for our pilot batches and to discuss route feasibility assessments tailored to your volume targets. Together, we can accelerate the deployment of next-generation display technologies that offer superior performance at a reduced total cost of ownership.
